Putting Brewing Water in Perspective John Palmer Brewing

Putting Brewing Water in Perspective John Palmer Brewing Solutions, Inc. Palmer Brewin Solutions, Inc.

The Dogma of Virgin Water Brewers don’t brew with water as-is. Classic beer styles may start out as a marriage of local ingredients and local water, but the beer/process evolves. Water has been adjusted for hundreds of years: Pre-boiled to precipitate alkalinity (aka. temporary hardness). Acid additions, sauermalz additions, salt additions. Even changing the water source. 2 Palmer Brewin Solutions, Inc.

The BIG Picture Why do we adjust brewing water? Improve Beer Yield. Improve Beer Flavor. What do we adjust? Mineral composition How do we adjust it? Salt additions Acid additions Ion Removal processes 3 Palmer Brewin Solutions, Inc.

Know Your Source Water Surface Water Low in Minerals, High in Organics Often requires Carbon Filtration to remove odors Ground Water High in Minerals, Low in Organics Often requires ion-exchange or RO to reduce Alkalinity Does your Source change during the year? 4 Palmer Brewin Solutions, Inc.

Order of Precedence Filter the source water Particle filtration for Silt Carbon filtration for Organics Adjust Source water Add Calcium salts (Ca. SO 4, Ca. Cl 2) Adjust Flavor Balance (Sulfate, Chloride, Sodium) Adjust Alkalinity (Na. HCO 3, Acidification) 5 Palmer Brewin Solutions, Inc.

Water Affects Beer Flavor Water Residual Alkalinity drives Mash p. H, Mash p. H drives Beer p. H, Beer p. H drives beer flavor expression. Seasoning Balance: Sulfate to Chloride Ratio More Sulfate = drier, more assertive hops More Chloride = rounder, fuller, sweeter malt Amount of Seasoning (Total Dissolved Solids) Palmer Brewin Solutions, Inc.

What is Water Hardness? Hardness = The sum of Calcium and Magnesium measured as calcium carbonate We WANT calcium and magnesium in our mash/beer. Water Hardness helps lower mash p. H Permanent Hardness = Ca/Mg Sulfates & Chlorides Temporary Hardness = Ca/Mg Bicarbonate, Carbonate SOFT = Not Hard. 7 Palmer Brewin Solutions, Inc.

What is Alkalinity? Total Alkalinity = the sum of carbonate species in water (from limestone) Alkalinity ≅ Temporary Hardness This is why we say we want to get rid of temporary hardness. Alkalinity raises Mash p. H, makes beer less acidic, but duller. 8 Palmer Brewin Solutions, Inc.

What is “As Ca. CO 3”? Calcium Carbonate is limestone. It is the major source of hardness, alkalinity, and scale on plumbing. Therefore, Hardness (Ca+2, Mg+2) and Alkalinity (HCO 3 -1, CO 3 -2) are measured in terms of their equivalents “as Ca. CO 3” 9 Palmer Brewin Solutions, Inc.

Measuring Concentration as Ca. CO 3 Calcium, magnesium, and bicarbonate can be measured as themselves, i. e. , Ca+2 40 ppm Or they can be measured as their calcium carbonate potential, i. e. , “as Ca. CO 3. The equivalent weight of Ca. CO 3 is 50 g/eq. 40 ppm Ca = 40/20 x 50 = 100 ppm Calcium Hardness as Ca. CO 3 10 Palmer Brewin Solutions, Inc.

Know What’s in Your Water Measure the Calcium, Magnesium, Total Alkalinity, Sodium, Chloride, Sulfate, and p. H with the Brew. Lab by the La. Motte Company. 11 Palmer Brewin Solutions, Inc.

Water, in general: Mineral Concentrations 0 -50 ppm is Low 50 -100 ppm is Medium 100 -150 ppm is High >150 ppm is a Problem Palmer Brewin Solutions, Inc.

Two Groups of Ions Affect p. H: Calcium Magnesium Total Alkalinity Affect Flavor: Sulfate Chloride Sodium 13 Palmer Brewin Solutions, Inc.

Calcium The most important ion in brewing. Cofactor for: Mash p. H via Residual Alkalinity Protein coagulation, trub formation. Yeast metabolism, flocculation, and beer clarity Oxalate Precipitation 50 ppm minimum in the wort for beer clarity 100 -200 ppm in mash and sparge liquor for stable p. H during lautering and better clarity. >200 ppm tends to taste minerally. 14 Palmer Brewin Solutions, Inc.

Magnesium The sidekick to calcium, but half as effective due to higher solubility of magnesium phosphates. A vital yeast nutrient. 5 ppm minimum in the wort needed for yeast. All malt wort (1. 040, 10°P) typically has 70 ppm Recommended level in water is 0 -40 ppm >80 ppm tastes sour/bitter in beer (maybe >40? ) Dark beers seem to benefit from ~30 ppm in the mash 15 Palmer Brewin Solutions, Inc.

Total Alkalinity as Ca. CO 3 The Villain Keeps the Hero in check Generally should be low, but RA is the key. Bicarbonate is generally 95% of Total Alkalinity drives mash p. H up Causes more tannin and silicate extraction Results in coarser bitterness from hops But, provides balance for darker, more acidic grain bills Recommended range 0 -100 ppm as Ca. CO 3, 150 ppm max. 16 Palmer Brewin Solutions, Inc.

Sulfate The Dark Hero. Accentuates hop character, helps dry the beer finish. Tastes minerally with high concentrations of sodium, chloride, and bicarbonate. 200 -400 ppm in Pale Ales and IPAs improves hop character and finish. Sulfur is generally not desired for German lagers such as Pilsner or Helles, conflicts with soft noble hop character. (≤ 50 ppm) Recommended range is 50 -400 ppm for most styles. 17 Palmer Brewin Solutions, Inc.

Chloride Imparts a rounder, sweeter, fuller quality to the beer. Minimum effective level for accentuating beer mouthfeel and flavor is probably 50 ppm. High Concentrations can hurt the beer: >300 ppm can hurt clarity and stability >400 ppm can hurt beer flavor >500 ppm can hurt fermentation Recommended range is 50 -150 ppm for most styles. 18 Palmer Brewin Solutions, Inc.

Sodium The bastard stepchild of brewing minerals. It’s everywhere: sodium chloride, sodium bicarbonate, sodium hydroxide… Can be difficult to remove from water; reverse osmosis removes up to 97%. Recommended level is <100 ppm Acts to improve mouthfeel and sweetness of malt. >150 ppm tends to taste salty, especially with significant chloride (<100 ppm). People’s sensitivity varies. 19 Palmer Brewin Solutions, Inc.

Sulfate to Chloride Ratio The Ratio can affect the balance of the beer – Dryness vs. Fullness It is not magic – 40: 10 ≠ 400: 100 Useful range is 9: 1 to 0. 5: 1 Maximum suggested sulfate is 500 ppm Maximum suggested chloride is 200 ppm Recommend to not exceed combined sum of 500 ppm. (Tastes Minerally) 20 Palmer Brewin Solutions, Inc.

TDS Effect –Mineral Structure Light vs Heavy Seasoning Bohemian Pilsner Rich, malty beer backed by large soft bitterness. Smooth finish balanced between malt and hops. No sharp edges. German Pils Crisp and bitter hop forward character, followed by clean malt and dry finish. This is a beer defined by clean edges. Dortmunder Export Balanced rich malt and firm dry bitterness. A “castle” of beer 21 structure. Lower alcohol but doesn’t taste like it. Palmer Brewin Solutions, Inc.

Comparison of Three Beers and Water Profiles City Ca+2 Mg+2 Na+1 Cl-1 SO 4 -2 HCO 3 -1 TDS Pilsen Munich* 7 12 180 2 17 15 2 4 40 6 8 130 8 18 330 16 95 80 50 150 775 Dortmund* *Numbers after pre-boil decarbonation treatment. Orders of magnitude increase in total mineral levels (TDS). Transition of beer character from soft, to assertive, to bold. 22 Palmer Brewin Solutions, Inc.

Adjusting Water for Style To Adjust water for style, use the cube: Define the style by Flavor, Color, & Structure. Read water profile by Calcium, Total Alkalinity, and Sulfate to Chloride Ratio. Palmer Brewin Solutions, Inc.

Adjusting Water for Style Beer Color => Residual Alkalinity Flavor Balance => Sulfate to Chloride Beer Structure => Calcium level Palmer Brewin Solutions, Inc.

What is Residual Alkalinity? Residual alkalinity is the difference between effects of the Total Alkalinity and Hardness on mash p. H. RA = Total Alkalinity – (Ca + ½ Mg)/3. 5 Note: Units must be m. Eq/L or ppm as Ca. CO 3 3. 5 factor is dependent on Mash Ratio and Crush Calcium and Magnesium react with malt phosphates to produce protons and lower mash p. H. 10 Ca+2 + 12 HCO 3− 1 + 6 H 2 PO 4− 1 + 2 H 2 O → Ca 10(PO 4)6(OH)2 + 12 CO 2 + 12 H 2 O + 2 H+1 Magnesium also reacts, but about half as much. 25 Palmer Brewin Solutions, Inc.

Effect of RA on baseline Mash p. H The distilled water mash p. H of a base malt is typically 5. 6 -6. 0. (Distilled water is the baseline. ) Therefore, achieving the optimum mash p. H range (5. 25. 6) with base malt alone is problematic. The RA of water can raise the mash p. H by 0. 1 p. H unit per 50 -75 ppm as Ca. CO 3 RA (or 1 -1. 5 m. Eq/L). Conversely, negative RA values can lower the mash p. H by the same amount. This is why we typically want to add hardness and remove alkalinity from our brewing water. 26 Palmer Brewin Solutions, Inc.

Increasing Effects R=8 R=6 R=4 R=2 Palmer Brewin Solutions, Inc.

∆RA inversely proportional to R ∆RA ≈ 200/R Palmer Brewin Solutions, Inc.

Mash ph is the Equilibrium between Water Chemistry and Malt Chemistry The effect of water chemistry in the mash is summarized by Residual Alkalinity. But this is only half the story. Each Malt contributes (poly)peptide buffers and organic acids. Quantifiable by titration and distilled water mash p. H Malt acidity is variable between maltsters and crop year, even for the same type. 29 Palmer Brewin Solutions, Inc.

Malt Chemistry Specialty malts have been kilned and/or roasted to produce color (melanoidins) and flavor compounds. These Maillard reactions also produce organic acids, such as acetic acid. There is a transition from yellow-red melanoidins to brown melanoidins at 325 -350°F (165 -175°C). This is also the transition for kilning to roasting. 30 Palmer Brewin Solutions, Inc.

Kilned vs. Roasted Acidity In general, distilled water mash p. H for a specialty malt is proportional to malt color Darker = lower p. H However, Caramel malts have higher buffering power than Roast malts. Roasting causes pyrolyzation and breakdown of buffers and acids. 31 Palmer Brewin Solutions, Inc.
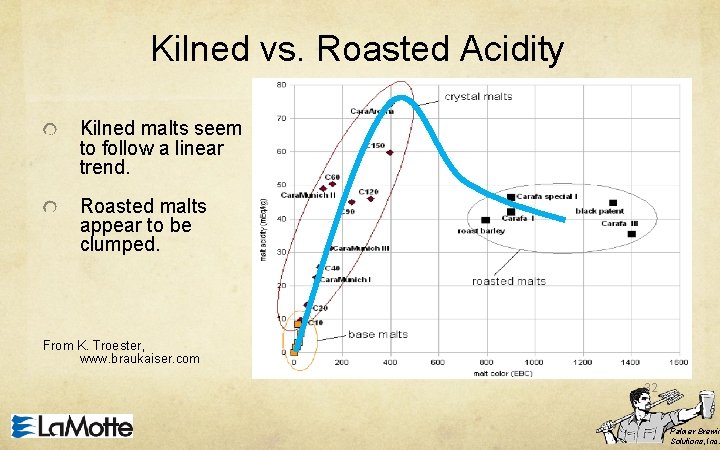
Kilned vs. Roasted Acidity Kilned malts seem to follow a linear trend. Roasted malts appear to be clumped. From K. Troester, www. braukaiser. com 32 Palmer Brewin Solutions, Inc.

Coming Together in the Mash Let’s assume our target mash p. H is 5. 4. The base malt, at a DI water p. H of 5. 8, is alkaline and needs to come down. The specialty malt(s), at a DI water p. H of (4. 6), is acidic and needs to come up. The residual alkalinity of the water can be positive or negative, and may help or hinder reaching the target. 33 Palmer Brewin Solutions, Inc.

Bottom Line Water Chemistry + Malt Chemistry = Mash Chemistry (measured as p. H) 34 Palmer Brewin Solutions, Inc.

p. H 1. Residual Alkalinity is the cornerstone of Mash p. H. 2. The Mash p. H is the Equilibrium between the Water Chemistry and the Malt Chemistry. 3. The Mash p. H sets up the wort p. H. 4. The wort p. H sets up the beer p. H. 5. The beer p. H controls how the beer flavors are experienced by your palate. 35 Palmer Brewin Solutions, Inc.
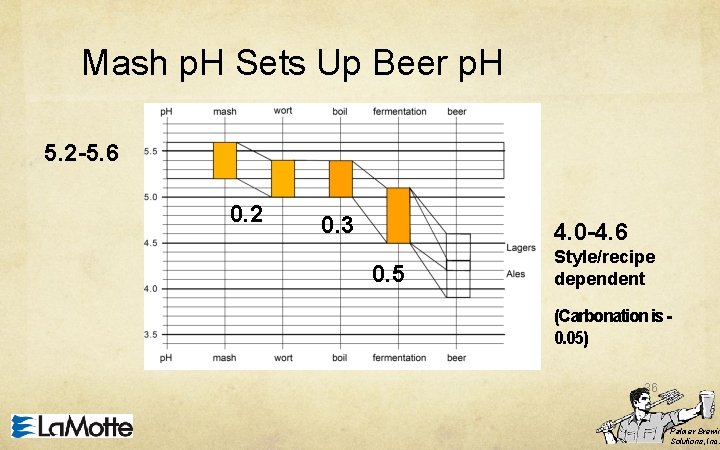
Mash p. H Sets Up Beer p. H 5. 2 -5. 6 0. 2 0. 3 4. 0 -4. 6 0. 5 Style/recipe dependent (Carbonation is 0. 05) 36 Palmer Brewin Solutions, Inc.

What Is the Optimum Mash p. H? It Depends: There are many enzymatic processes occurring in the mash, each with it’s own optimum p. H range. The p. H optima for proteolysis is lower than for saccharification. Therefore optimum mash p. H represents a compromise between priorities: conversion, FAN, lauterability, etc. “Optimum” Mash p. H is generally based on optimum yield. However, published opinions differ on what the optimum range is… Palmer Brewin Solutions, Inc.

p. H for Best Yield, by Author Briggs et al. : 5. 2 -5. 4 (5. 45 -5. 65 at 20°C) Bamforth et al. : 5. 3 -5. 8 (5. 55 -6. 05 at 20°C) Kunze: (5. 25 -5. 35) 5. 5 -5. 6 at 20°C Best Yield is therefore 5. 4 -5. 8 Anecdotally, many brewers report better beer flavor with lower mash p. H, 5. 2 -5. 6 at 20°C. Parentheses indicate p. H/temperature conversion Palmer Brewin Solutions, Inc.

Bottom Line on p. H Optima There is no single correct answer, it is a range where your target will depend on several factors, including water profile, grain bill, yield targets, and perceived beer flavor. I recommend picking a value between 5. 2 -5. 6 (measured at room temperature) that best suits your beer recipe. Mash p. H should be 5. 2 -5. 6 (paler = lower) Don’t go over 5. 8 or under 5. 2! Beer p. H should be 4. 0 -4. 6 (paler = lower) Palmer Brewin Solutions, Inc.

WHEN & HOW to Measure p. H? You are looking for a mash p. H of 5. 2 -5. 6 @ room temp, for saccharification. Measure the p. H about 10 minutes into the mash. Cool the mash wort sample to room temp on a shallow dish, then measure with a p. H meter. Note that mash p. H decreases throughout the mash. If at first you don’t succeed, brew again. (you can try to fix it now, but conversion happens quickly, so…) 40 Palmer Brewin Solutions, Inc.

Effect of Beer p. H on Flavor In general, a lower beer p. H focuses and brightens the malt and hop flavors. Best for single malt pale beers. In general, a higher beer p. H broadens and opens up malt and hop flavors. Best for multiple malt dark beers. Palmer Brewin Solutions, Inc.

Every Beer has a p. H Every beer recipe has an optimum beer p. H, generally in the range of 4. 0 -4. 6. Every beer should have a flavor portfolio that includes: malt flavors and aromas, hop flavors and aromas, yeast flavors and aromas. If you can’t taste or smell everything, you are probably not at the optimum beer p. H. 42 Palmer Brewin Solutions, Inc.

p. H Guidelines Mash p. H is important Pale beers: 5. 2 -5. 4 Amber beers: 5. 3 -5. 5 Dark beers: 5. 4 -5. 6 Always cool a sample and measure at room temperature. Wort p. H is important Beer p. H is important Water p. H is NOT! Palmer Brewin Solutions, Inc.

The Water p. H is Not Important. The water p. H is not important. The water p. H is the chemical equilibrium of the water, i. e. , the balance of hardness and alkalinity. Higher p. H = more alkalinity than hardness 2 different waters can have the same water p. H Brewers are interested in the chemical equilibrium of the mash. 44 Palmer Brewin Solutions, Inc.

Acidification of Water is Easy Acidification to reduce Total Alkalinity is easy. Divide Total Alkalinity by 50 to get m. Eq/liter. Ex. TA = 150 ppm as Ca. CO 3 ÷ 50 = 3 m. Eq/liter. Use 3 m. Eq/Liter of 1 N Acid per liter of water to neutralize the alkalinity. 1 N solution is 85 m. L of 88% Lactic Acid added to water to make 1 liter (total). Mix acid solution with water in HLT and stir to 45 vent the CO 2. Palmer Brewin Solutions, Inc.

When to Add Salts? Mash p. H is the thing! Add to HLT, or Mash, but get mash p. H right. You can always add more at the kettle if you want to tweak total Calcium or Sulfate: Chloride. You can do post adjustments, but more is needed than if done earlier in process. 46 Palmer Brewin Solutions, Inc.

Capping– Dark Grain Last? Mash Roast malt separately or together? Depends; the important thing is your mash p. H. How much roast flavor do you want? Shorter time = more grain Longer time = less grain p. H determines flavor profile 47 Palmer Brewin Solutions, Inc.

Palmer’s Water App 48 Palmer Brewin Solutions, Inc.

Why Do We Adjust Water? To Improve Beer Yield Better mash = better wort = better beer To Improve Beer Flavor Balance Flavor Structure Flavor expression (Sulfate: Chloride) (TDS) (p. H) 49 Palmer Brewin Solutions, Inc.

Questions? 50 Palmer Brewin Solutions, Inc.
- Slides: 50