Purification of phosphoric acid by liquidliquid extraction Mariana

Purification of phosphoric acid by liquid-liquid extraction Mariana Campos Assuncao 1, 2, Alexandre Chagnes 1, Hubert Halleux 2, Gérard Cote 1 1, PSL Research University, Chimie Paris. Tech - CNRS, Institut de Recherche de Chimie Paris, 11 rue Pierre et Marie Curie, 75005 Paris, France 2 Prayon, Gansbroekstraat 31, B 2870 Ruisbroek, Belgium Institut de recherche de Chimie Paris 2 nd International Conference on Separation Techniques (26 -28 September 2016) 1
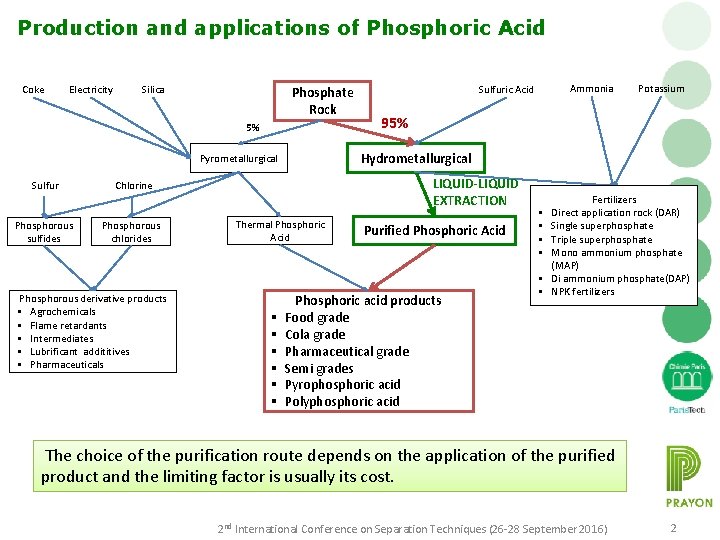
Production and applications of Phosphoric Acid Coke Electricity Silica Phosphate Rock 5% Phosphorous sulfides Phosphorous derivative products § Agrochemicals § Flame retardants § Intermediates § Lubrificant addititives § Pharmaceuticals 95% LIQUID-LIQUID EXTRACTION Chlorine Phosphorous chlorides Thermal Phosphoric Acid § § § Potassium Hydrometallurgical Pyrometallurgical Sulfur Ammonia Sulfuric Acid Purified Phosphoric Acid Phosphoric acid products Food grade Cola grade Pharmaceutical grade Semi grades Pyrophosphoric acid Polyphosphoric acid § § § Fertilizers Direct application rock (DAR) Single superphosphate Triple superphosphate Mono ammonium phosphate (MAP) Di ammonium phosphate(DAP) NPK fertilizers The choice of the purification route depends on the application of the purified product and the limiting factor is usually its cost. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 2
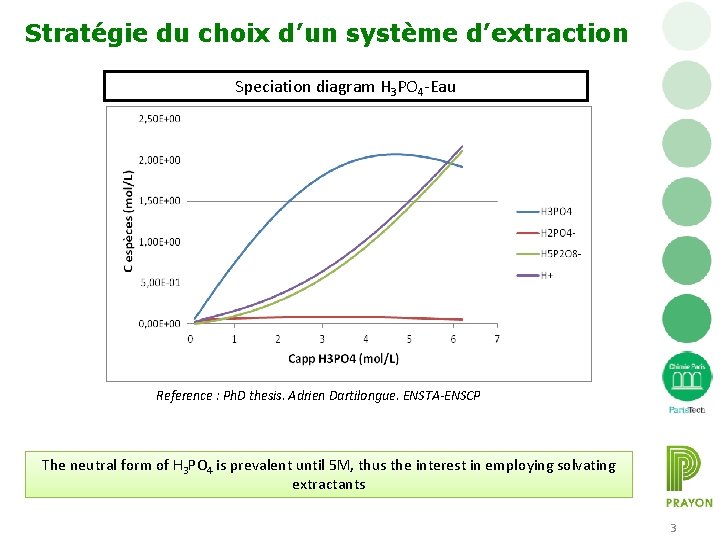
Stratégie du choix d’un système d’extraction Speciation diagram H 3 PO 4 -Eau Reference : Ph. D thesis. Adrien Dartilongue. ENSTA-ENSCP The neutral form of H 3 PO 4 is prevalent until 5 M, thus the interest in employing solvating extractants 3
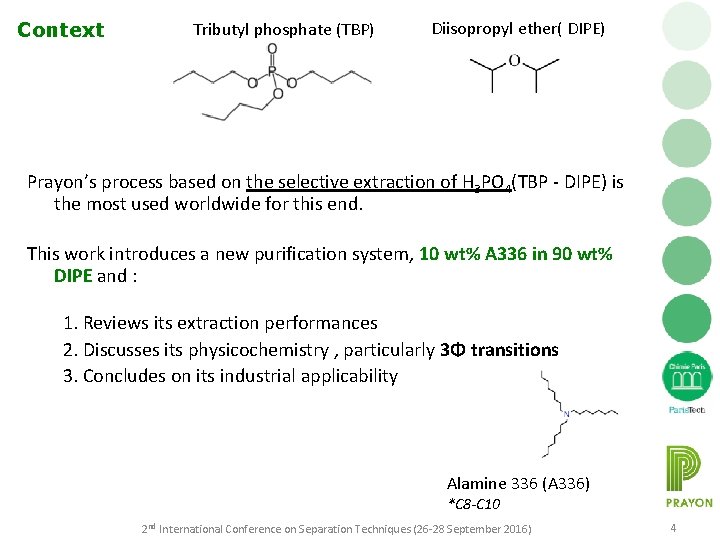
Context Tributyl phosphate (TBP) Diisopropyl ether( DIPE) Prayon’s process based on the selective extraction of H 3 PO 4(TBP - DIPE) is the most used worldwide for this end. This work introduces a new purification system, 10 wt% A 336 in 90 wt% DIPE and : 1. Reviews its extraction performances 2. Discusses its physicochemistry , particularly 3Φ transitions 3. Concludes on its industrial applicability Alamine 336 (A 336) *C 8 -C 10 2 nd International Conference on Separation Techniques (26 -28 September 2016) 4

Purification of phosphoric acid Typical composition of feed solution Element Concentration (%) Al 2 O 3 0, 79 F 0, 19 Mg. O 0, 36 SO 3 0, 39 As 0, 40 Fe 2 O 3 0, 57 H 3 PO 4 13. 4 M Ti 1380 ppm Ca. O 0, 03 TOC 78, 9 K 2 O 0, 12 Si. O 2 0, 01 Particularities of the system: High acid content Important volume variation during extraction Extraction of H 3 PO 4 Organic Aqueous Expansion Organic Contraction 2 nd International Conference on Separation Techniques (26 -28 September 2016) Aqueous 5
![Extraction yield and selectivity Feed solution [H 3 PO 4]=14 M DH 3 PO Extraction yield and selectivity Feed solution [H 3 PO 4]=14 M DH 3 PO](http://slidetodoc.com/presentation_image/142f0a41d7de9e0828255e9c07a68c4d/image-6.jpg)
Extraction yield and selectivity Feed solution [H 3 PO 4]=14 M DH 3 PO 4 % Extraction 0. 4 % Stripping 100 90 0. 35 80 70 0. 25 60 0. 2 50 0. 15 40 Great selectivity 30 0. 1 20 0. 05 0 % Yield DH 3 PO 4 0. 3 10 TBP A 336 0 The new system have a better yield an interesting selectivity towards Ti. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 6
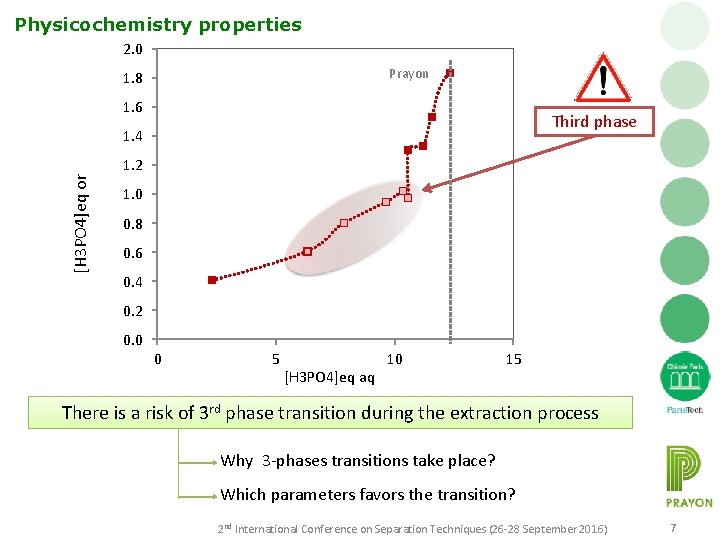
Physicochemistry properties 2. 0 Prayon 1. 8 1. 6 Third phase [H 3 PO 4]eq or 1. 4 1. 2 1. 0 0. 8 0. 6 0. 4 0. 2 0. 0 0 5 [H 3 PO 4]eq aq 10 15 There is a risk of 3 rd phase transition during the extraction process Why 3 -phases transitions take place? Which parameters favors the transition? 2 nd International Conference on Separation Techniques (26 -28 September 2016) 7
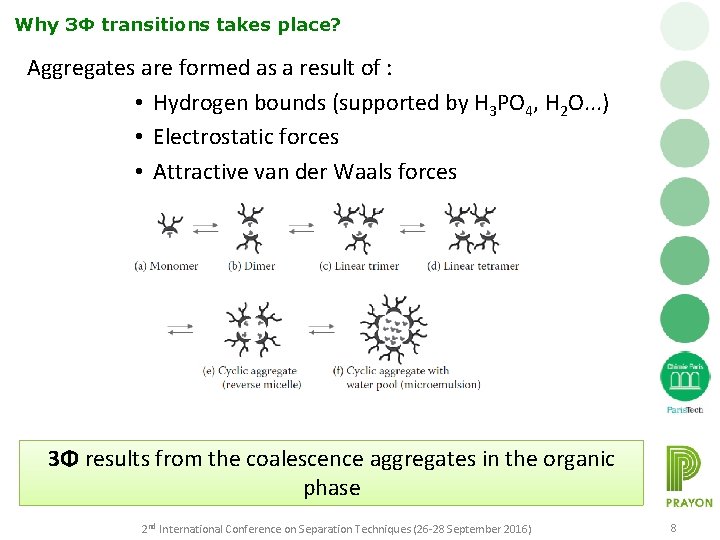
Why 3Φ transitions takes place? Aggregates are formed as a result of : • Hydrogen bounds (supported by H 3 PO 4, H 2 O. . . ) • Electrostatic forces • Attractive van der Waals forces 3Φ results from the coalescence aggregates in the organic phase 2 nd International Conference on Separation Techniques (26 -28 September 2016) 8
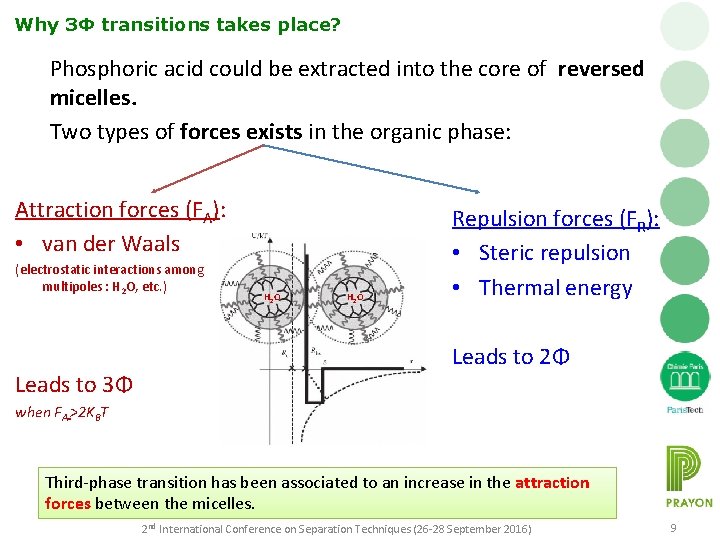
Why 3Φ transitions takes place? Phosphoric acid could be extracted into the core of reversed micelles. Two types of forces exists in the organic phase: Attraction forces (FA): • van der Waals (electrostatic interactions among multipoles : H 2 O, etc. ) Leads to 3Φ H 2 O Repulsion forces (FR): • Steric repulsion • Thermal energy Leads to 2Φ when FA >2 KBT Third-phase transition has been associated to an increase in the attraction forces between the micelles. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 9
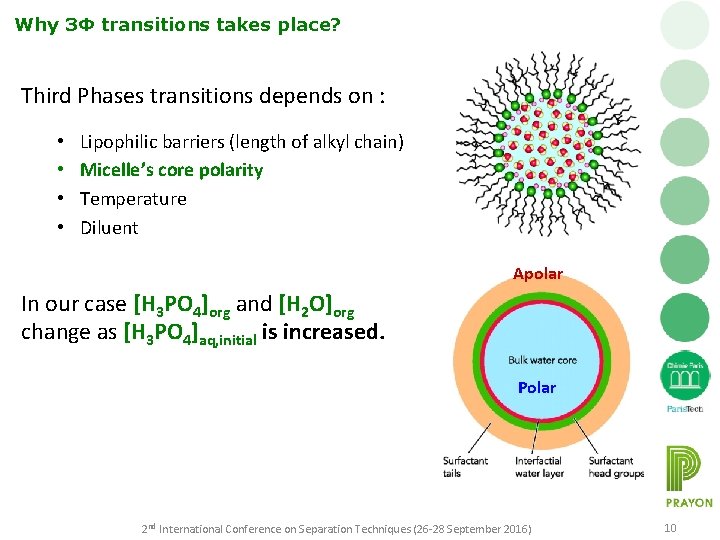
Why 3Φ transitions takes place? Third Phases transitions depends on : • • Lipophilic barriers (length of alkyl chain) Micelle’s core polarity Temperature Diluent Apolar In our case [H 3 PO 4]org and [H 2 O]org change as [H 3 PO 4]aq, initial is increased. Polar 2 nd International Conference on Separation Techniques (26 -28 September 2016) 10

Why 3Φ transitions takes place? Reverse micelles in the organic phase have strong intermolecular attraction when their cores includes high H 2 O concentration H 2 O exists in reverse micelles as : • • Free water Bulky water Anion bounded water Cation bounded water The avarage radius of aggregation increases when the acid and/or H 2 O concentration increases. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 11
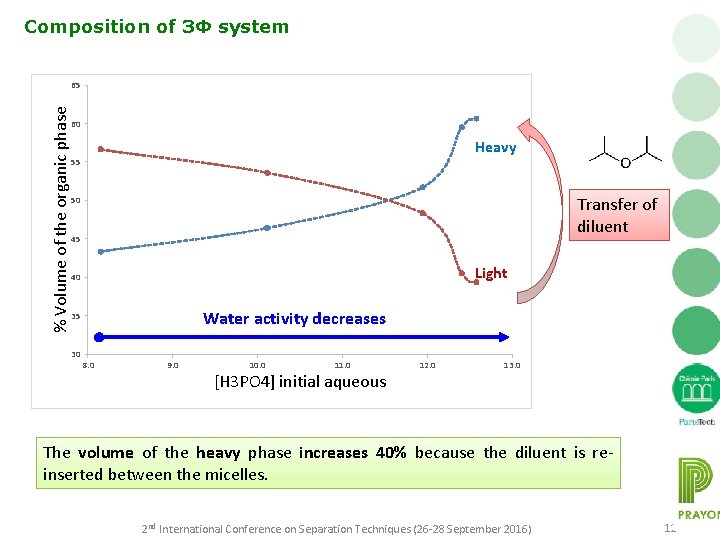
Composition of 3Φ system % Volume of the organic phase 65 60 Heavy 55 Transfer of diluent 50 45 Light 40 Water activity decreases 35 30 8. 0 9. 0 10. 0 11. 0 [H 3 PO 4] initial aqueous 12. 0 13. 0 The volume of the heavy phase increases 40% because the diluent is reinserted between the micelles. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 12
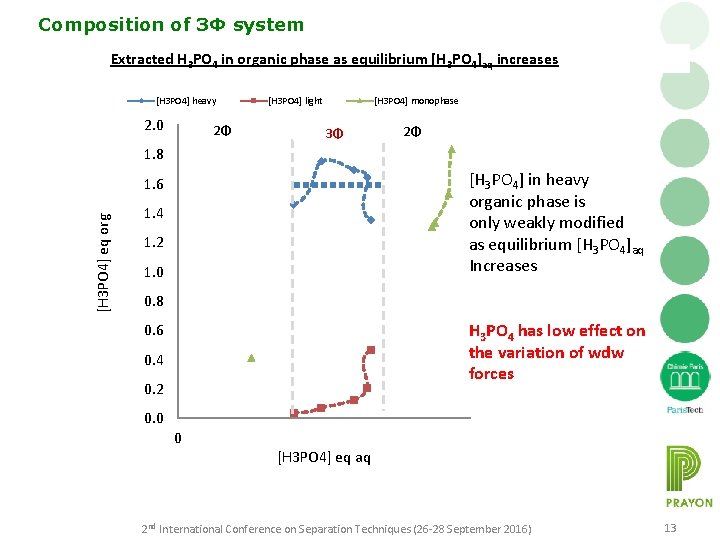
Composition of 3Φ system Extracted H 3 PO 4 in organic phase as equilibrium [H 3 PO 4]aq increases [H 3 PO 4] heavy 2. 0 2Φ [H 3 PO 4] light [H 3 PO 4] monophase 3Φ 2Φ 1. 8 [H 3 PO 4] in heavy organic phase is only weakly modified as equilibrium [H 3 PO 4]aq Increases [H 3 PO 4] eq org 1. 6 1. 4 1. 2 1. 0 0. 8 H 3 PO 4 has low effect on the variation of wdw forces 0. 6 0. 4 0. 2 0. 0 0 [H 3 PO 4] eq aq 2 nd International Conference on Separation Techniques (26 -28 September 2016) 13
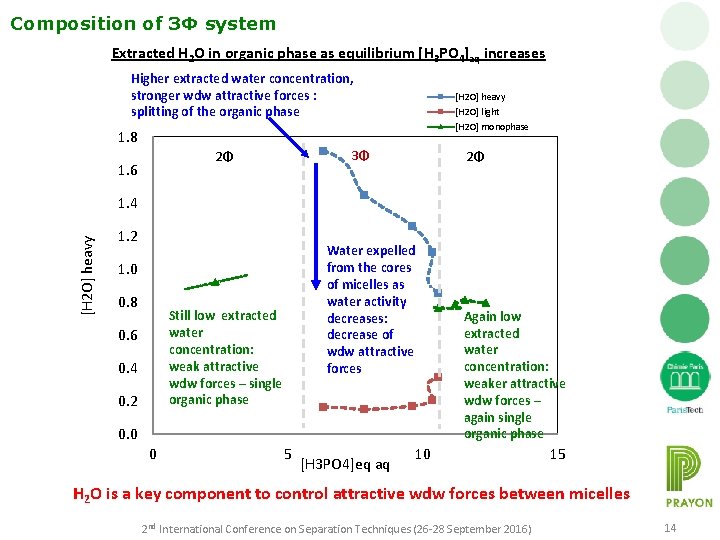
Composition of 3Φ system Extracted H 2 O in organic phase as equilibrium [H 3 PO 4]aq increases Higher extracted water concentration, stronger wdw attractive forces : splitting of the organic phase [H 2 O] heavy [H 2 O] light [H 2 O] monophase 1. 8 3Φ 2Φ 1. 6 2Φ [H 2 O] heavy 1. 4 1. 2 Water expelled from the cores of micelles as water activity decreases: decrease of wdw attractive forces 1. 0 0. 8 Still low extracted water concentration: weak attractive wdw forces – single organic phase 0. 6 0. 4 0. 2 0. 0 0 5 [H 3 PO 4]eq aq Again low extracted water concentration: weaker attractive wdw forces – again single organic phase 10 15 H 2 O is a key component to control attractive wdw forces between micelles 2 nd International Conference on Separation Techniques (26 -28 September 2016) 14
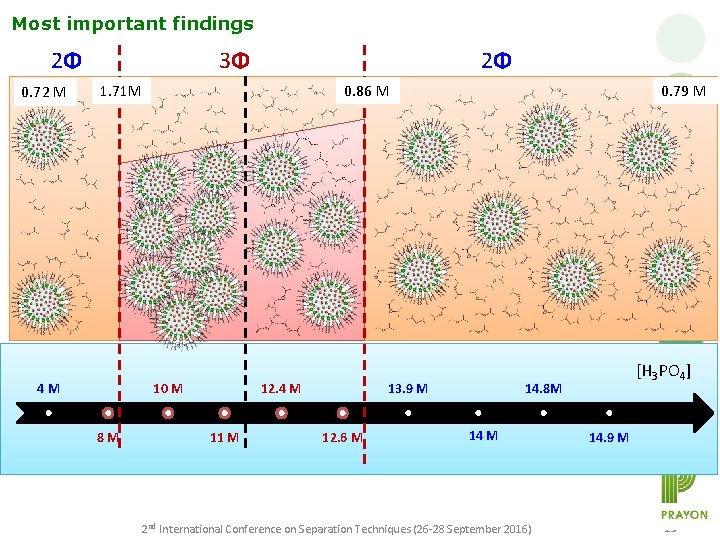
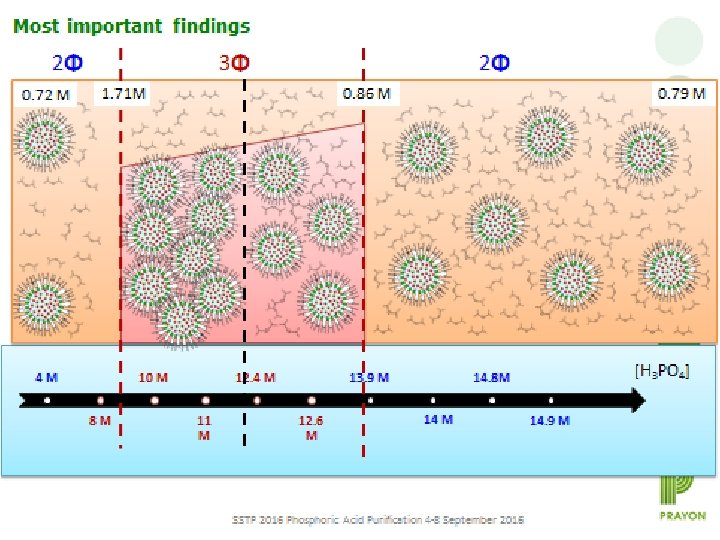
Most important findings 2Φ 0. 72 M 3Φ 2Φ 1. 71 M 4 M 0. 86 M 10 M 8 M 12. 4 M 11 M 0. 79 M 13. 9 M 12. 6 M [H 3 PO 4] 14. 8 M 14 M 2 nd International Conference on Separation Techniques (26 -28 September 2016) 14. 9 M 15
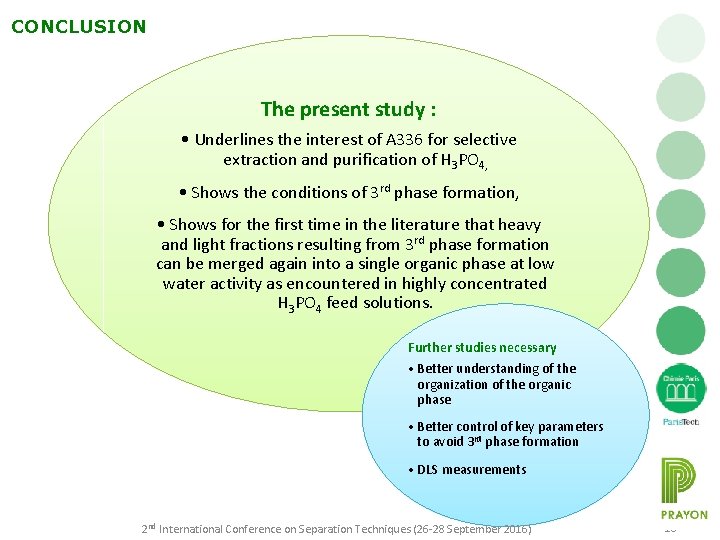
CONCLUSION The present study : • Underlines the interest of A 336 for selective extraction and purification of H 3 PO 4, • Shows the conditions of 3 rd phase formation, • Shows for the first time in the literature that heavy and light fractions resulting from 3 rd phase formation can be merged again into a single organic phase at low water activity as encountered in highly concentrated H 3 PO 4 feed solutions. Further studies necessary • Better understanding of the organization of the organic phase • Better control of key parameters to avoid 3 rd phase formation • DLS measurements 2 nd International Conference on Separation Techniques (26 -28 September 2016) 16

Thank you for the attention Questions ? 2 nd International Conference on Separation Techniques (26 -28 September 2016) 17
Comité de suivi de thèse(20/11/2015) 18
![[H 3 PO 4] eq aq Heavy phase Light Phase Sum Monophase n H [H 3 PO 4] eq aq Heavy phase Light Phase Sum Monophase n H](http://slidetodoc.com/presentation_image/142f0a41d7de9e0828255e9c07a68c4d/image-19.jpg)
[H 3 PO 4] eq aq Heavy phase Light Phase Sum Monophase n H 2 O n H 3 PO 4 2. 33 / / 0. 30 0. 13 6. 32 0. 24 0. 20 0. 03 0. 01 / / 0. 27 0. 21 7. 85 0. 21 0. 26 0. 03 0. 01 / / 0. 24 0. 27 9. 62 0. 21 0. 28 0. 03 0. 02 / / 0. 24 0. 30 10. 35 0. 22 0. 33 0. 03 / / 0. 25 0. 36 10. 57 0. 16 0. 26 0. 05 0. 06 / / 0. 21 0. 32 10. 58 / / 0. 24 0. 41 11. 21 / / 0. 26 0. 44 11. 57 / / 0. 26 0. 48 12. 34 / / 0. 26 0. 60 The total of moles of acid increases consistently but total moles of water remains the same. As we move towards the border of 3Φ→ 2Φ 19
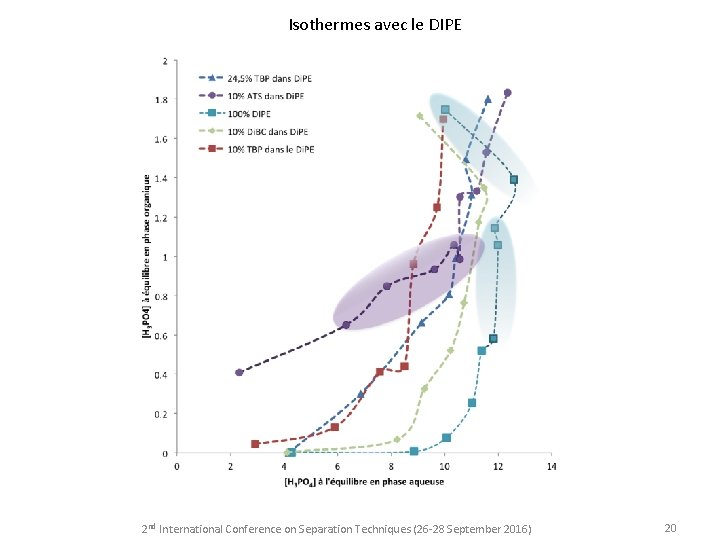
Isothermes avec le DIPE 2 nd International Conference on Separation Techniques (26 -28 September 2016) 20
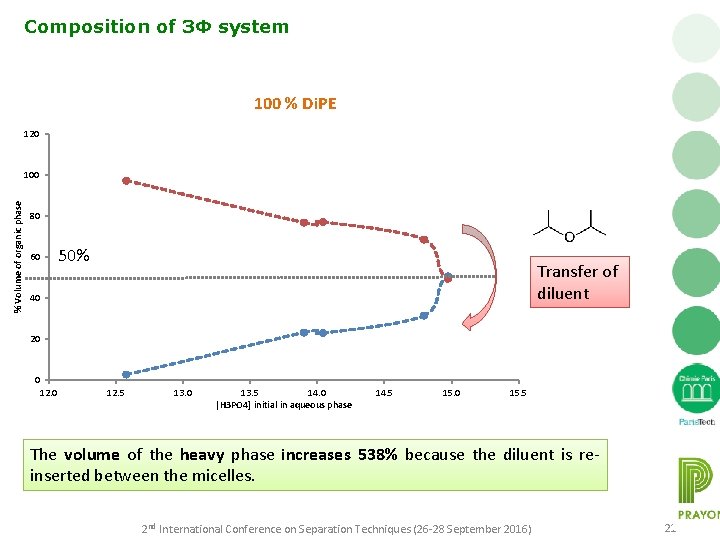
Composition of 3Φ system 100 % Di. PE 120 % Volume of organic phase 100 80 50% 60 Transfer of diluent 40 20 0 12. 5 13. 0 13. 5 14. 0 |H 3 PO 4] initial in aqueous phase 14. 5 15. 0 15. 5 The volume of the heavy phase increases 538% because the diluent is reinserted between the micelles. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 21
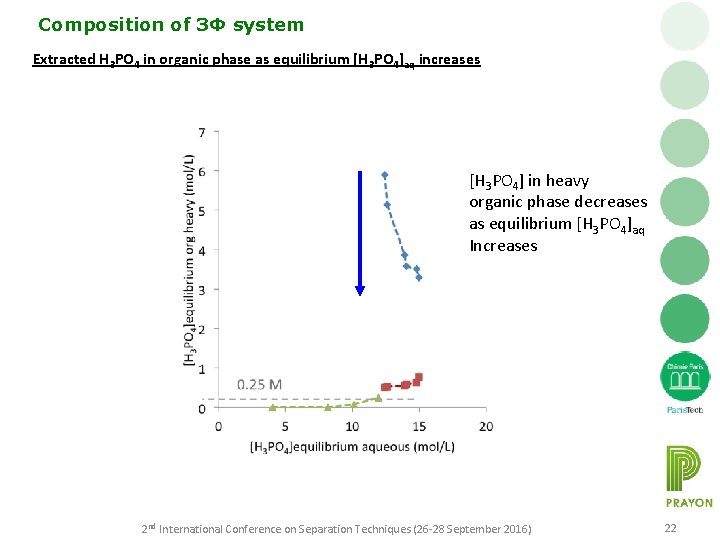
Composition of 3Φ system Extracted H 3 PO 4 in organic phase as equilibrium [H 3 PO 4]aq increases [H 3 PO 4] in heavy organic phase decreases as equilibrium [H 3 PO 4]aq Increases 2 nd International Conference on Separation Techniques (26 -28 September 2016) 22
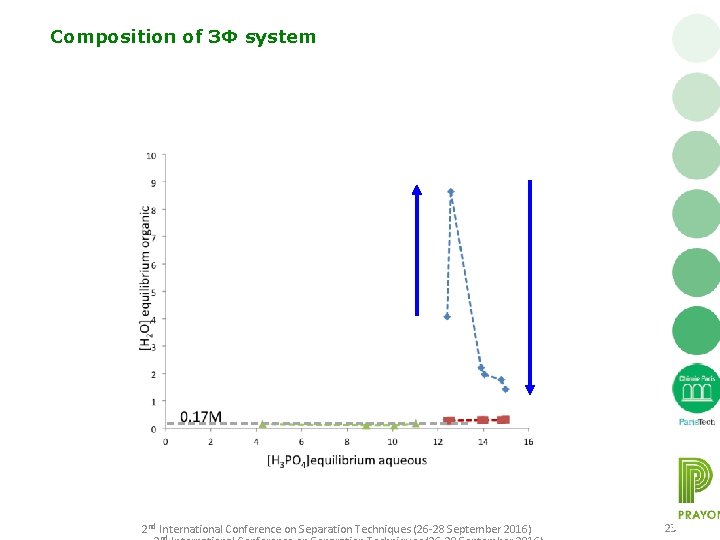
Composition of 3Φ system 2 nd International Conference on Separation Techniques (26 -28 September 2016) 23
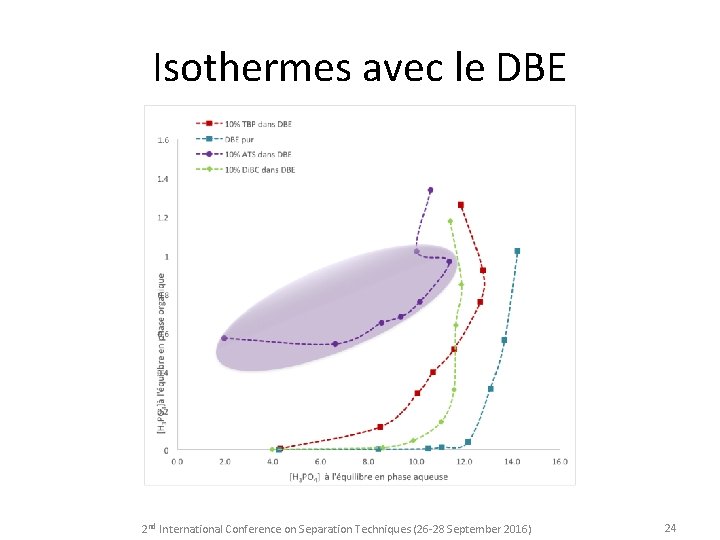
Isothermes avec le DBE 2 nd International Conference on Separation Techniques (26 -28 September 2016) 24
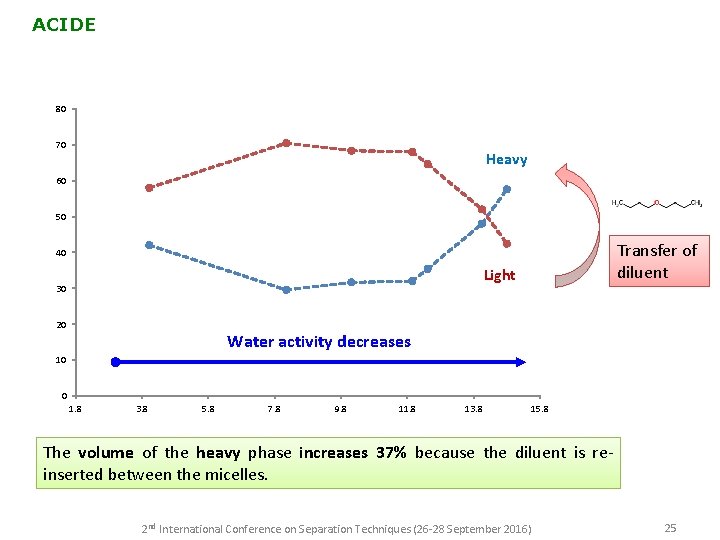
ACIDE 80 70 Heavy 60 50 Transfer of diluent 40 Light 30 20 Water activity decreases 10 0 1. 8 3. 8 5. 8 7. 8 9. 8 11. 8 13. 8 15. 8 The volume of the heavy phase increases 37% because the diluent is reinserted between the micelles. 2 nd International Conference on Separation Techniques (26 -28 September 2016) 25
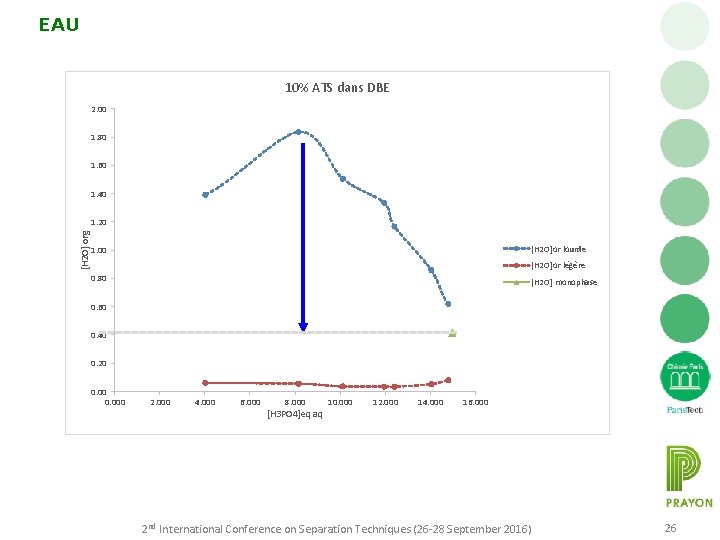
EAU 10% ATS dans DBE 2. 00 1. 80 1. 60 1. 40 [H 2 O] org 1. 20 [H 2 O]or lourde 1. 00 [H 2 O]or légère 0. 80 [H 2 O] monophase 0. 60 0. 40 0. 20 0. 000 2. 000 4. 000 6. 000 8. 000 [H 3 PO 4]eq aq 10. 000 12. 000 14. 000 16. 000 2 nd International Conference on Separation Techniques (26 -28 September 2016) 26
![2. 5 [H 3 PO 4] heavy [H 3 PO 4] light 2 [H 2. 5 [H 3 PO 4] heavy [H 3 PO 4] light 2 [H](http://slidetodoc.com/presentation_image/142f0a41d7de9e0828255e9c07a68c4d/image-27.jpg)
2. 5 [H 3 PO 4] heavy [H 3 PO 4] light 2 [H 3 PO 4] monophase 1. 5 1 0. 5 0 0 2 4 6 8 10 12 14 2 nd International Conference on Separation Techniques (26 -28 September 2016) 16 27
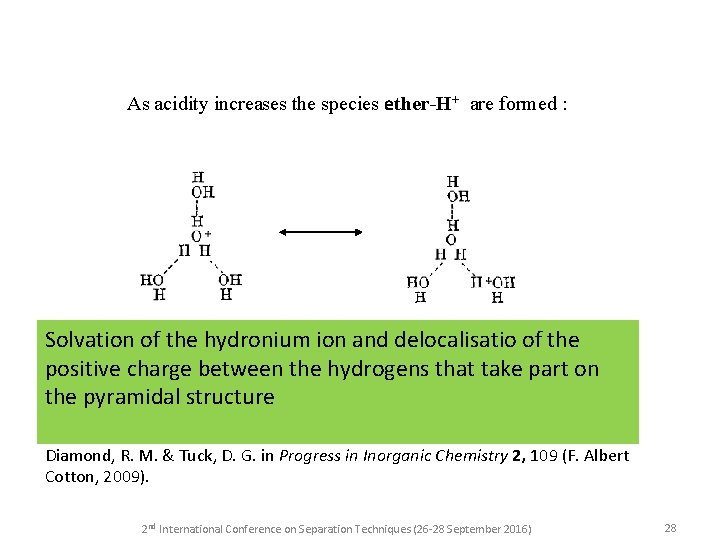
As acidity increases the species ether-H+ are formed : Solvation of the hydronium ion and delocalisatio of the positive charge between the hydrogens that take part on the pyramidal structure Diamond, R. M. & Tuck, D. G. in Progress in Inorganic Chemistry 2, 109 (F. Albert Cotton, 2009). 2 nd International Conference on Separation Techniques (26 -28 September 2016) 28
- Slides: 28