Purification of Benzaldehyde Week 2 Benzaldehyde Most aldehydes

Purification of Benzaldehyde Week 2
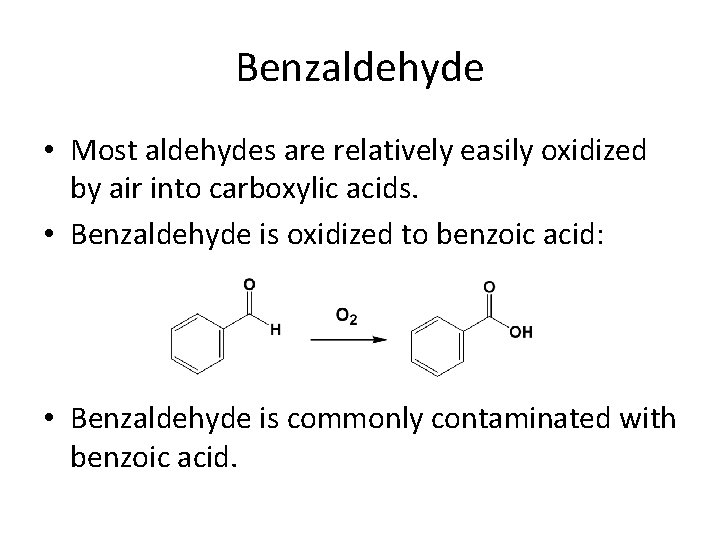
Benzaldehyde • Most aldehydes are relatively easily oxidized by air into carboxylic acids. • Benzaldehyde is oxidized to benzoic acid: • Benzaldehyde is commonly contaminated with benzoic acid.
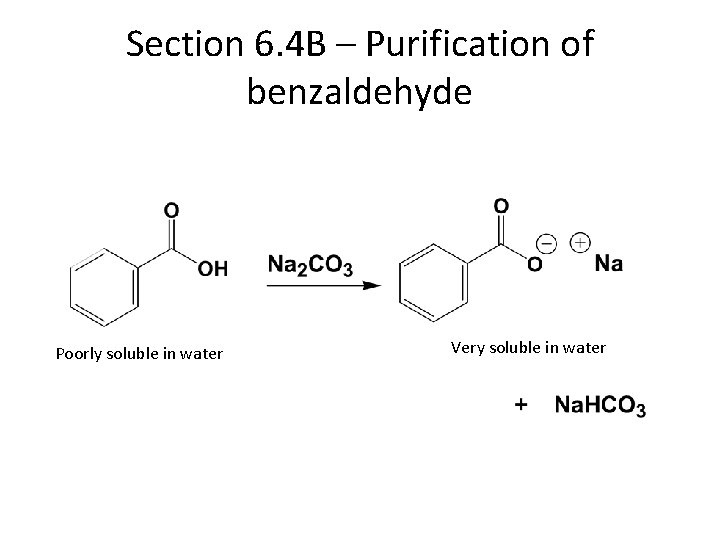
Section 6. 4 B – Purification of benzaldehyde Poorly soluble in water Very soluble in water
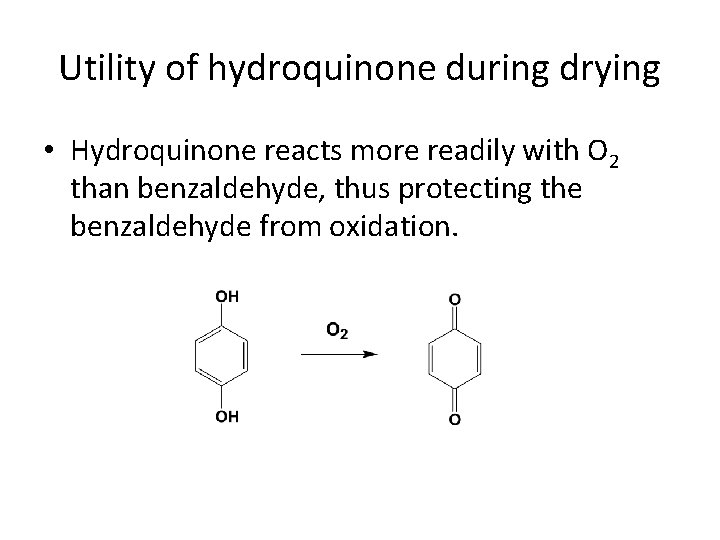
Utility of hydroquinone during drying • Hydroquinone reacts more readily with O 2 than benzaldehyde, thus protecting the benzaldehyde from oxidation.

We will not distill • Benzaldehyde must be distilled under low pressure and at low temperature, or it will oxidize quickly. • The present organic chemistry teaching lab is not equipped for low pressure distillations. • Extracting out the benzoic acid with base, and protecting with hydroquinone is usually sufficient to render benzaldehyde pure for most chemistry.

Benzoin Condensation Week 2

Benzoin Condensation • Traditional approach involves cyanide ion (-C≡N) as the catalyst. – The cyanide mechanism is outlined in detail in you book. – Cyanide is highly toxic and not used in our organic chemistry labs. • We will be using thiamine (vitamin B 1) as an alternative catalyst.
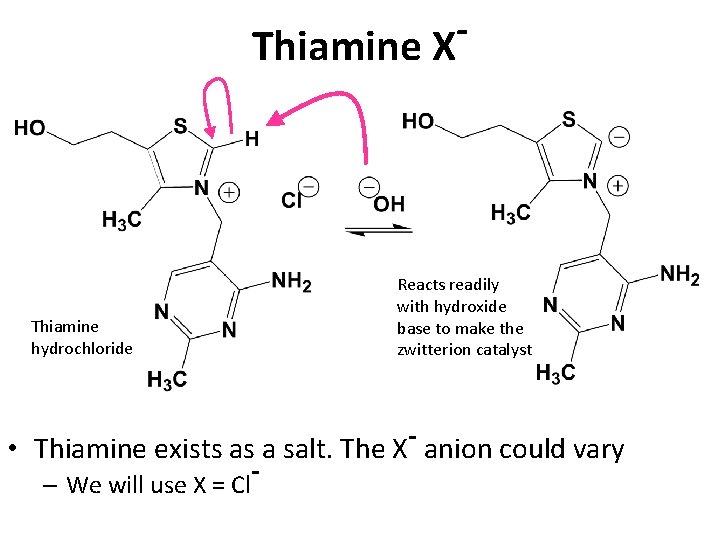
Thiamine X Thiamine hydrochloride Reacts readily with hydroxide base to make the zwitterion catalyst • Thiamine exists as a salt. The X- anion could vary – We will use X = Cl
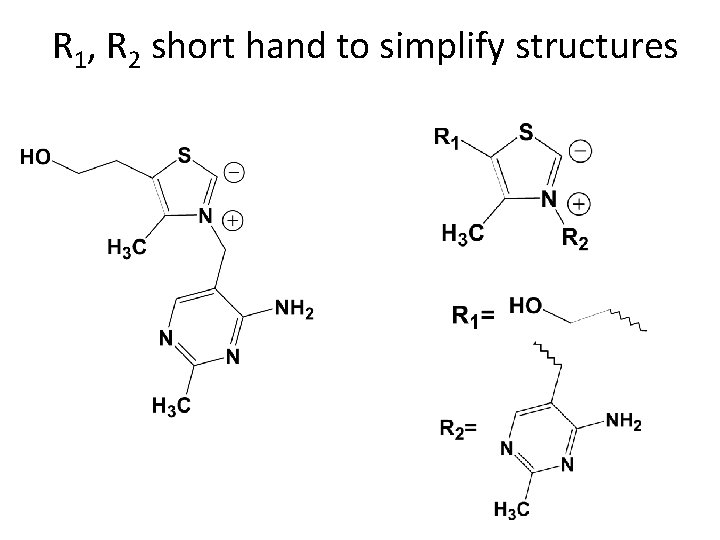
R 1, R 2 short hand to simplify structures
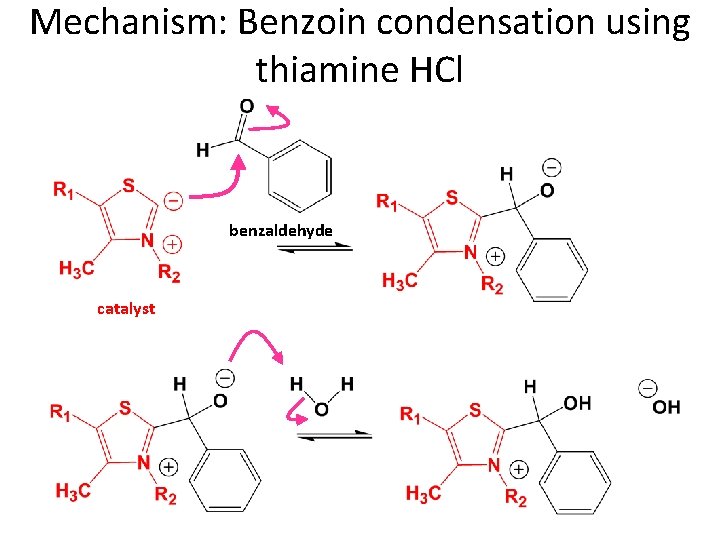
Mechanism: Benzoin condensation using thiamine HCl benzaldehyde catalyst
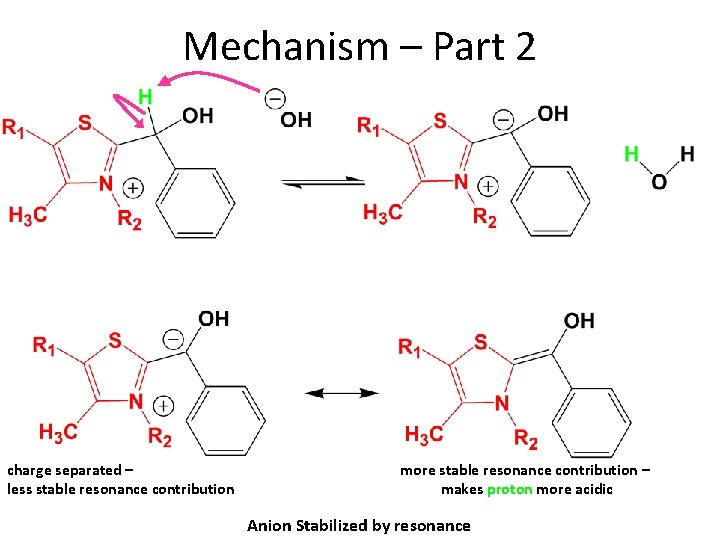
Mechanism – Part 2 charge separated – less stable resonance contribution more stable resonance contribution – makes proton more acidic Anion Stabilized by resonance
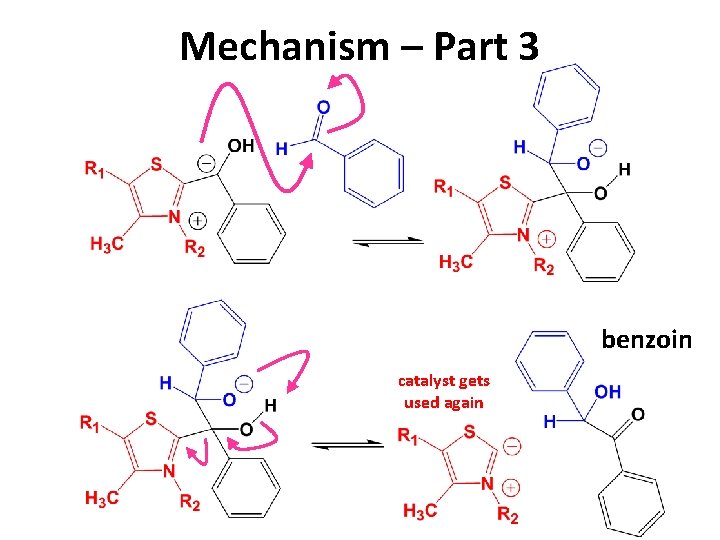
Mechanism – Part 3 benzoin catalyst gets used again

Hazards • You will be working with a Na. OH solution. This can cause chemical burns.

Stilbene by Wittig reaction Week 2
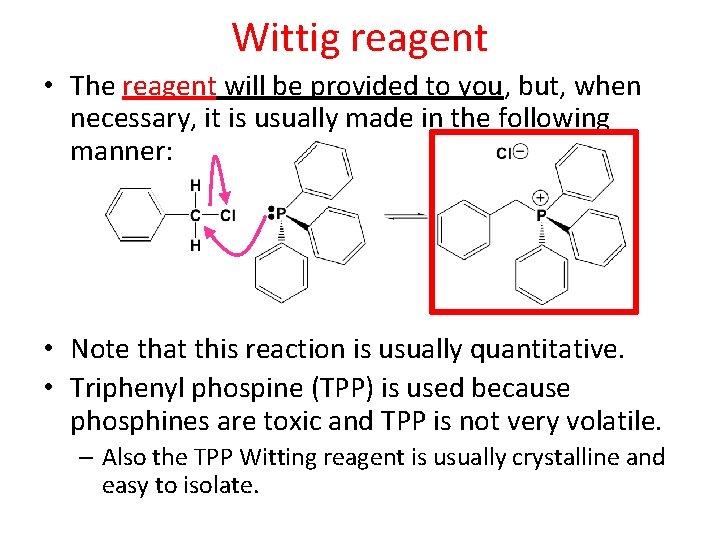
Wittig reagent • The reagent will be provided to you, but, when necessary, it is usually made in the following manner: • Note that this reaction is usually quantitative. • Triphenyl phospine (TPP) is used because phosphines are toxic and TPP is not very volatile. – Also the TPP Witting reagent is usually crystalline and easy to isolate.
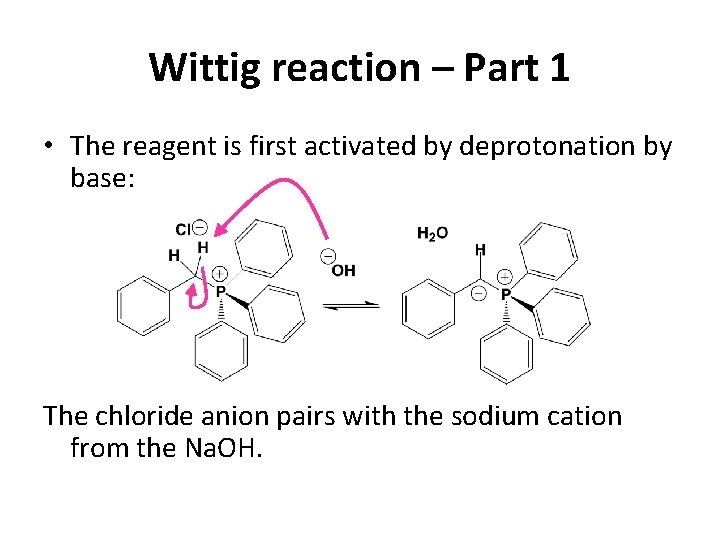
Wittig reaction – Part 1 • The reagent is first activated by deprotonation by base: The chloride anion pairs with the sodium cation from the Na. OH.
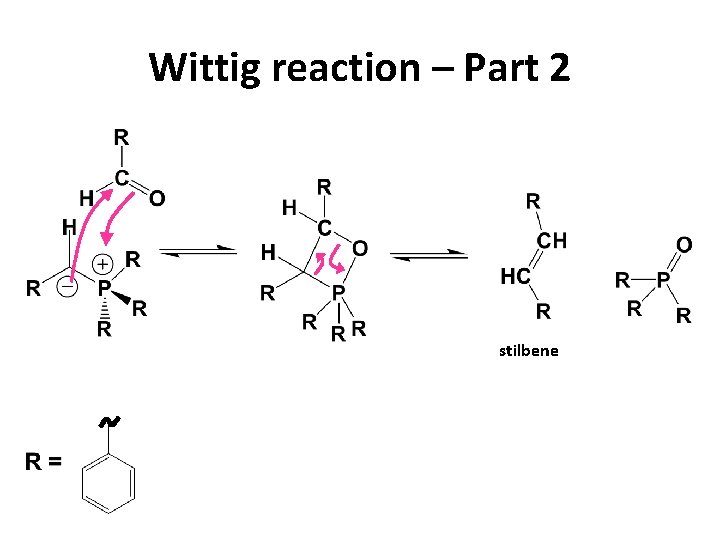
Wittig reaction – Part 2 stilbene
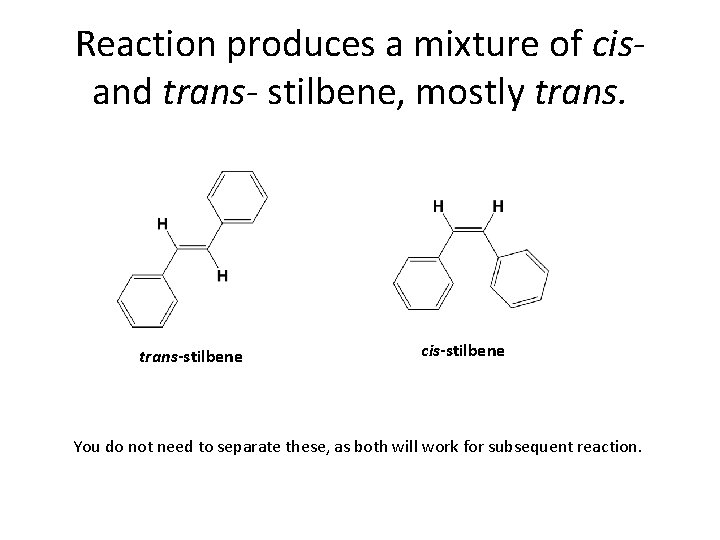
Reaction produces a mixture of cisand trans- stilbene, mostly trans-stilbene cis-stilbene You do not need to separate these, as both will work for subsequent reaction.

Cautions • Treat the phosphorus containing compounds as toxic. • You are using a strong base, Na. OH which can cause serious chemical burns. • Dichloromethane (DCM) is a cancer suspect agent – use in the hood. • Distill off the solvent cautiously. – Do not distill to dryness • Distilling any solvent to dryness can cause an explosion hazard. – Reduce the volume, then pour the last 5 m. L onto a watch glass, and allow to evaporate to dryness. Rinse out any remaining solid from the flask onto the watch glass with a few m. L of DCM. • Use all other cautions provided to you about working with organic compounds and working in this lab.

Trituration with warm hexanes • Hexane is a nonpolar solvent. – The polar triphenylphosphine oxide will not be extracted, while the nonpolar stilbenes will extracted. • We do not have a steam bath. Gently warm the hexanes in a flask on your hotplate. – Caution: fire hazard! Hexanes vapor can ignite on your hotplate. • This will not explode, but may cause a flame to appear at the top of your flask. If this happens, notify your TA for help.

Trituration – part 2 • Swirl your solid mixture in the warm hexanes. – Again, the salt (Na. Cl) and phosphine oxide will not dissolve, while your stilbenes will dissolve. • Pour the suspension through a filter paper. • Pour additional aliquots of hexanes through to ensure you have washed through the stilbenes. – Discard the solid on the filter paper. – Distill off (not to dryness – caution – explosion hazard!) the hexanes in the flask to obtain your pure stilbenes. • Pour off the remaining few m. L onto a watch glass as done earlier.

Convenient places to stop if you are running out of time • Carrying out reaction • Distilling of dicholormethane – You could stop after this step, if necessary, and complete the hexanes trituration the following week. • Hexanes trituration – You could stop after this step, if necessary, and complete the TLC the following week. • TLC analysis of final product

Do not recrystallize • Your product comes out reasonably pure. • Recrystallization is time consuming, and one often does not recover enough product for the subsequent steps in the synthesis. • Also, the two stilbene isomers act as impurities to each other in recrystallization, so the process eliminates one of the isomers, though both isomers work in the bromination chemistry.

END
- Slides: 24