Pure Substances vs Mixtures Using the characteristic properties

Pure Substances vs. Mixtures Using the characteristic properties of substances to separate mixtures

Characteristic Properties l What are some characteristic properties of substances? l l l density melting point boiling point heat capacity solubility

Mixtures l A mixture is comprised of two or more pure substances. l The substances composing a mixture can be combined in any amount l l For example, salt-water can be made of one grain of salt in a gallon of water or a cup of salt in a gallon of water. The properties of a mixture are between those of the constituent substances l A mixture of ethanol and water will have a boiling point between that of water and that of ethanol

Mixtures l l The properties of mixtures are intermediate between those of the components of the mixture because each component retains its own properties. This is rather significant! It means that we can exploit individual substances’ properties in order to separate a mixture.

Ethanol and Water l Let’s say we had a mixture of ethanol and water. l l l Ethanol has a boiling point of 78. 4 o. C and water has a boiling point of 100. 0 o. C. Together, their boiling point will be appear to be somewhere between those two numbers depending on how much there was of each substance. The different boiling points can be used to separate the mixture by using distillation.
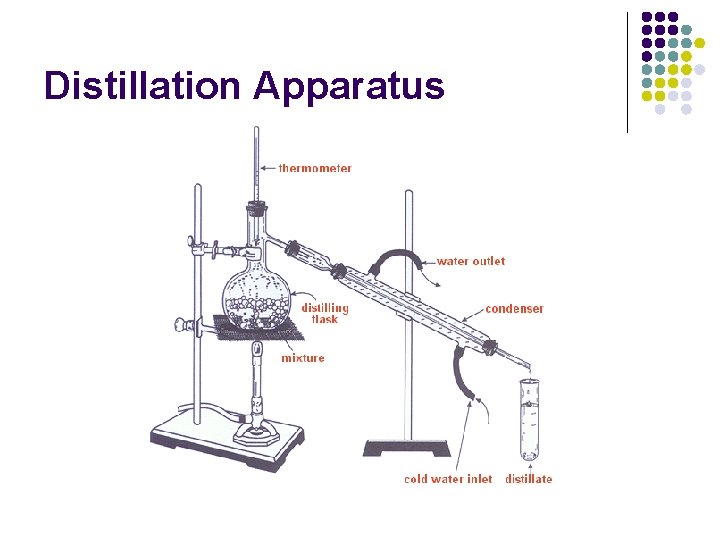
Distillation Apparatus
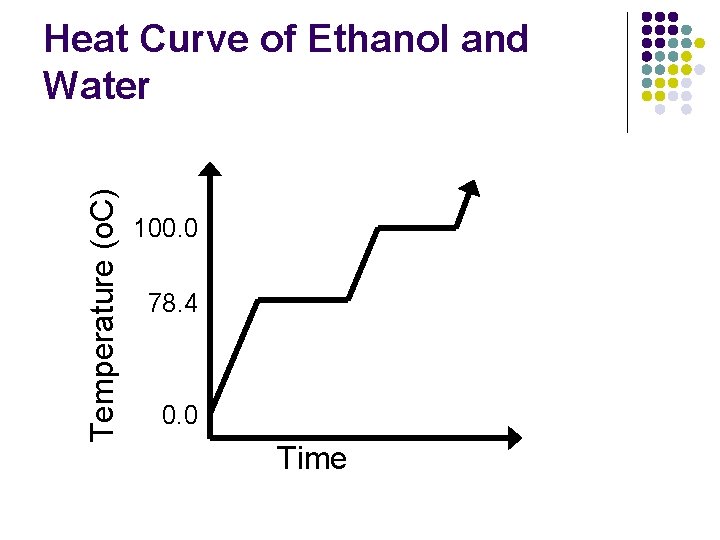
Temperature (o. C) Heat Curve of Ethanol and Water 100. 0 78. 4 0. 0 Time

Fractional Distillation l An animation of fractional distillation as a means of refining oil can be viewed at the following website: http: //science. howstuffworks. com/oilrefining 4. htm

Other Methods of Separation l How could you separate a mixture of salt and sand? l l l Start by listing some properties of salt and of sand. Differences in this list can be exploited to separate the mixture. Is salt water-soluble? Is sand? Could water be used to separate the two? How could the water be recovered from the salt?

Other Methods of Separation l Chromatography is another method of separation. l l The components of ink can be separated using this method. Homework: Bring in a marker from home in order to do a chromatography experiment.
- Slides: 10