Pure Substances and Mixtures Pure Substances any substance

Pure Substances and Mixtures

Pure Substances ● any substance that has a fixed composition and definite properties o a chemical formula can be written ● Chemical Formula: chemical symbols and numbers indicating the atoms contained in a substance. o glucose sugar: C 6 H 12 O 6 o table salt: Na. Cl

Pure Substances ● Elements: pure substances that can not be broken down by chemical means. o Elements can be broken down physically into smaller parts known as an atom. o Each element is made of one kind of atom. o Examples: Carbon (C) or Hydrogen (H)

Pure Substances ● Compounds are pure substances. o Made up of two or more atoms of different elements. o If a compound is broken down into its smallest pieces, it is known as a molecule. o Ex: water (H 2 O) or sugar (C 6 H 12 O 6)

Mixtures ● a combination of more than one pure substance that are only physically combined. ● you cannot write a chemical formula for a mixture.
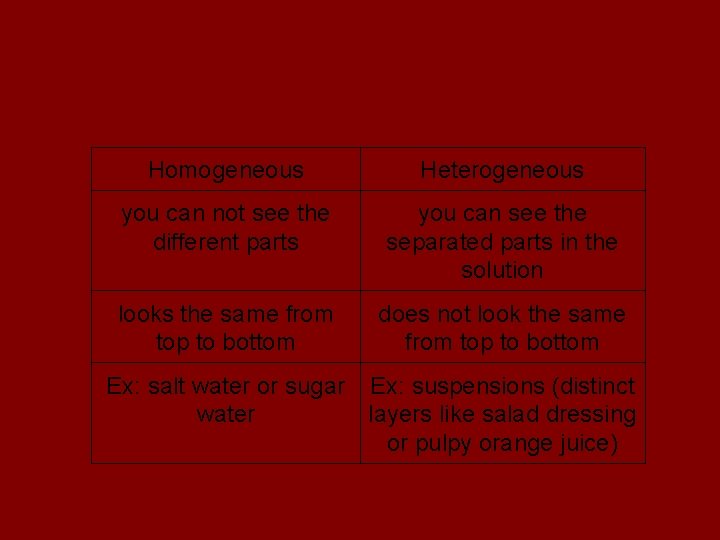
Mixtures Homogeneous Heterogeneous you can not see the different parts you can see the separated parts in the solution looks the same from top to bottom does not look the same from top to bottom Ex: salt water or sugar Ex: suspensions (distinct water layers like salad dressing or pulpy orange juice)

Pure Substance or Mixture? Gold (Au) Pure Substance Element or Compound?

Pure Substance or Mixture? Chicken Noodle Soup Mixture Homogeneous or Heterogeneous ?

Pure Substance or Mixture? Water (H 2 O) Pure Substance Element or Compound ?

Pure Substance or Mixture? Apple Juice Mixture Homogeneous or Heterogeneous ?
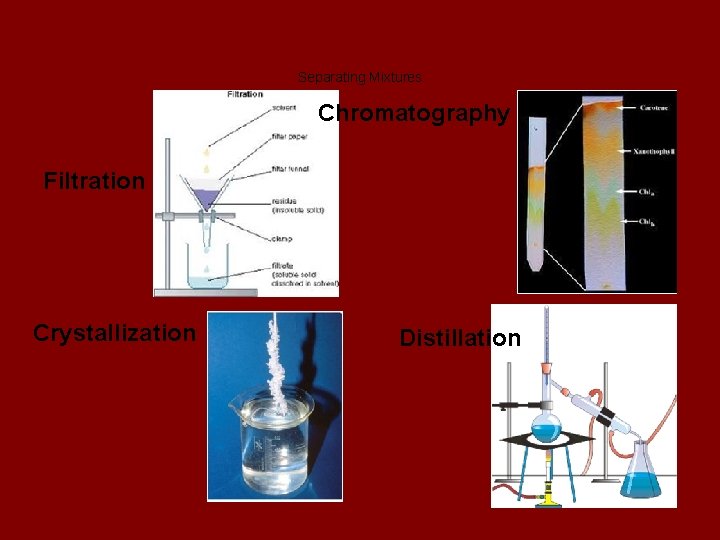
Separating Mixtures Chromatography Filtration Crystallization Distillation

Filtration ● Filtration is when the solid substance (filtrate) is removed from the liquid of a solution. ● Example: If you mix water and chalk, you can remove the chalk by filtering it out. The chalk left in the filter paper is called the filtrate.

Distillation ● Distillation is a process in which one substance is boiled away from another and then collected. The boiled off vapor is cooled and "condensed". o Scientists often use lab equipment such as a distillation flask and a condenser.

Chromatography ● The separation of a mixture into its individual components. o Used to separate the different components of inks and dyes
- Slides: 14