Pure Substances and Mixtures Categorizing Matter All matter

Pure Substances and Mixtures Categorizing Matter

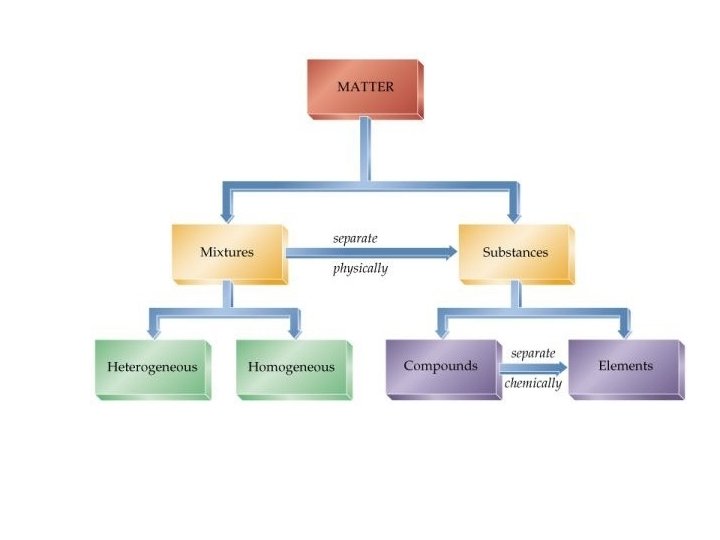
• All matter in the universe is either a pure substance or a mixture.

Pure Substance = 1) Made of either a single element or one particular kind of compound 2) Has a definite composition 1) H 2 O, Na. Cl, C, etc. 3) Can’t be broken down into simpler substances through physical changes Two Types of Pure Substances • • Elements Compounds

Element = • Consists of only one type of atom • Au, Ag, C, Na, Br, K, H, etc. Compound = • 2 or more different atoms chemically combined together 1) Composition is always the same 2) Cannot be broken down using physical changes • H 20, Na. Cl, CO 2, etc.

Mixtures = • Substances that have been combined physically (no chemical reaction) 1) Homogeneous Mixtures = solutions • • “Homo” = same Mixture that looks the same everywhere – Air, salt water, sugar water 2) Heterogeneous Mixtures = suspensions • • “Hetero” = different Looks different throughout – Mud puddle, blood, salad dressing, chocolate chip cookie, bird seed

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Element, Compound, or Mixture?

Exam Review and ET: #9 -12

9. A heterogeneous mixture is one that is not a. uniform throughout. b. made of two or more liquids. c. easily mixed together. d. edible by humans.

10. Which of the following is a homogeneous mixture? a. salad dressing b. rubbing alcohol c. gelatin d. orange juice with pulp

11. A compound differs from a mixture because it a. always remains frozen even at high temperatures. b. is formed from two cations. c. always contains the same elements in the same proportion. d. can form only in the presence of heat energy.
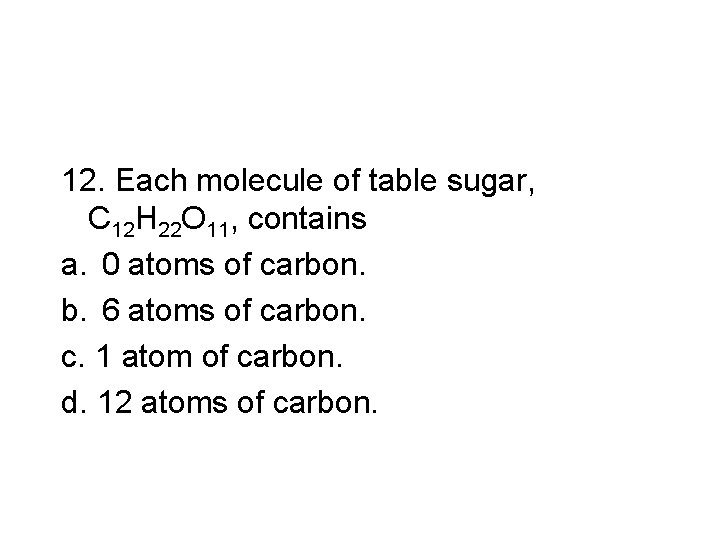
12. Each molecule of table sugar, C 12 H 22 O 11, contains a. 0 atoms of carbon. b. 6 atoms of carbon. c. 1 atom of carbon. d. 12 atoms of carbon.
- Slides: 19