PURE SUBSTANCE A pure substance is a type

PURE SUBSTANCE A pure substance is a type of matter with a fixed makeup of atoms. Elements are always a pure substance. H 2 O and Na. Cl, is a pure substance, it is always the same. But NOT when mixed together in a solution! Substances can be either elements or compounds.
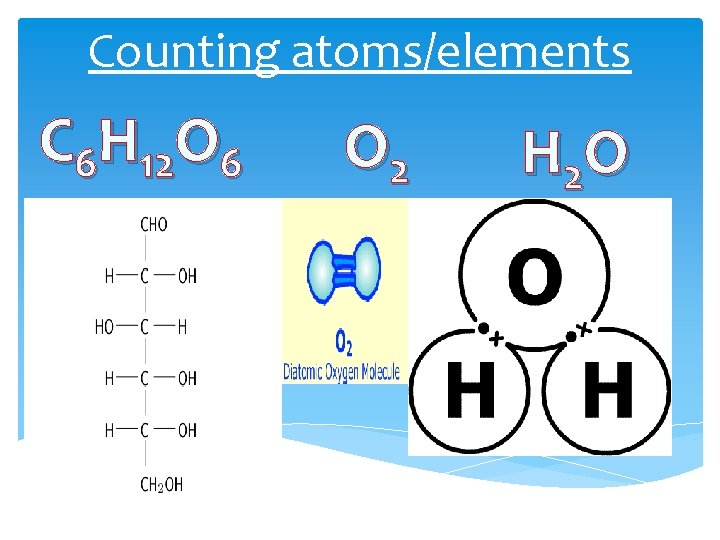
Counting atoms/elements C 6 H 12 O 6 O 2 H 2 O
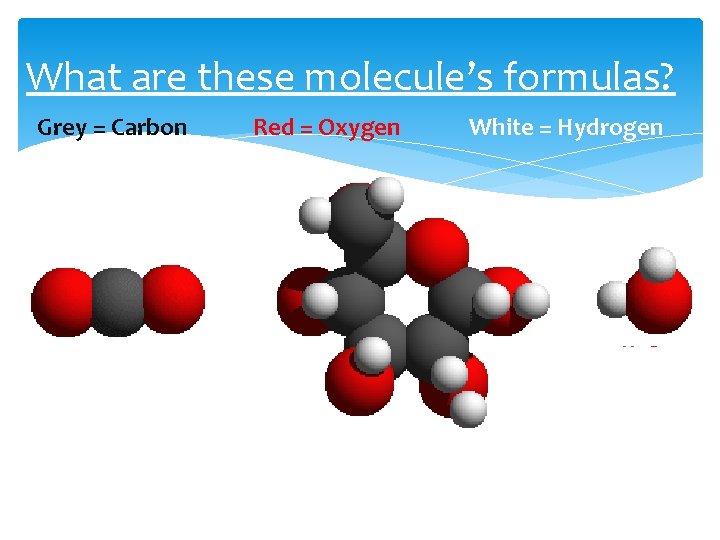
What are these molecule’s formulas? Grey = Carbon Red = Oxygen White = Hydrogen

Law of Conservation of Mass* Matter can neither be created nor destroyed, it can only change form.
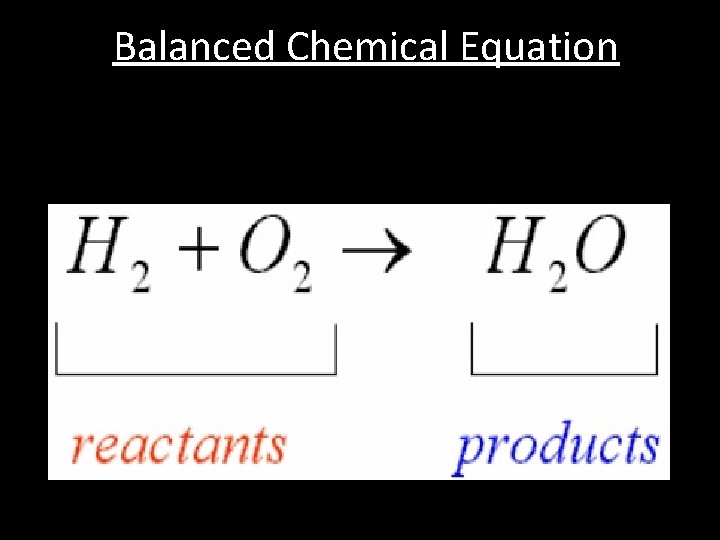
Balanced Chemical Equation
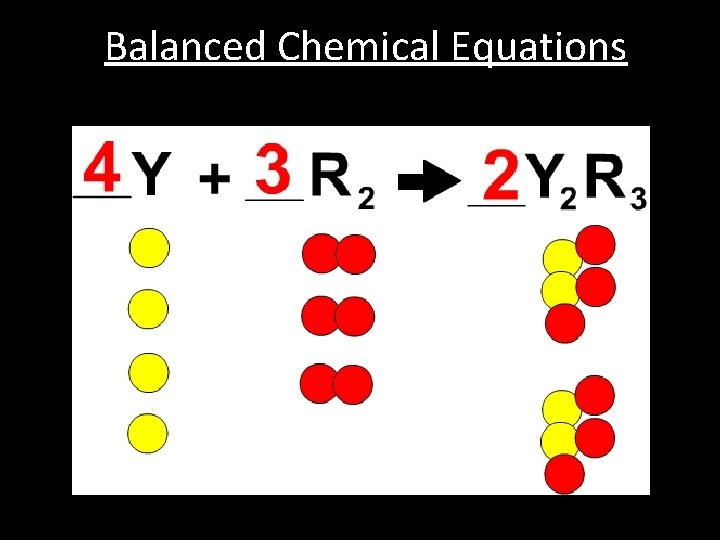
Balanced Chemical Equations
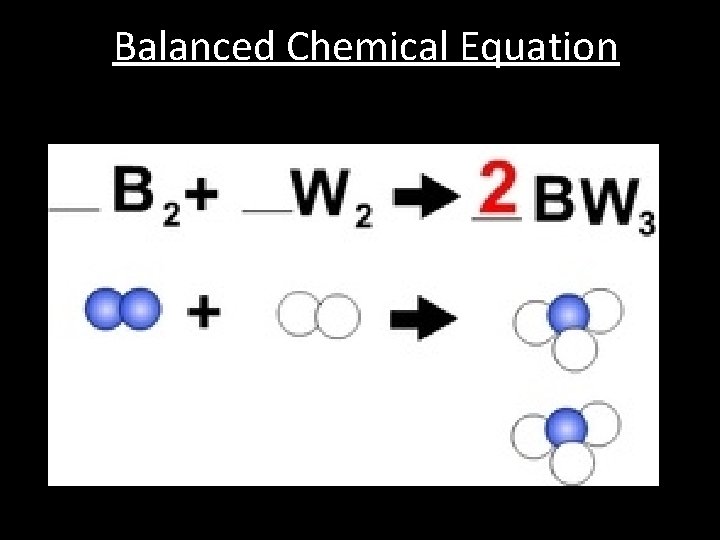
Balanced Chemical Equation
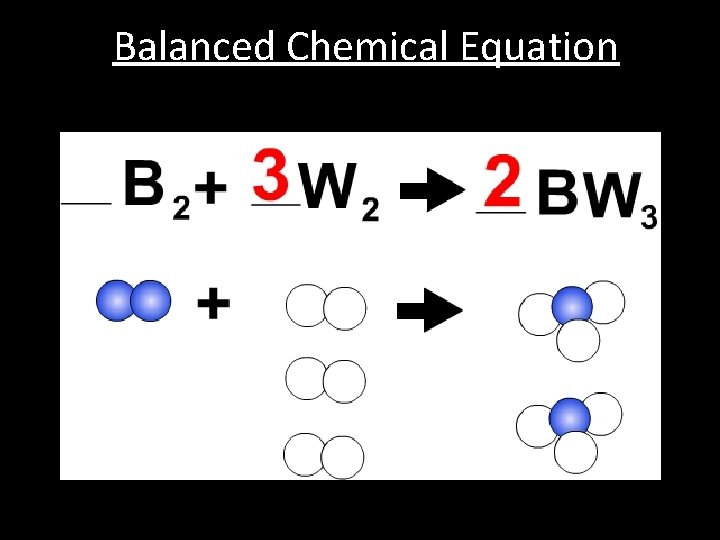
Balanced Chemical Equation
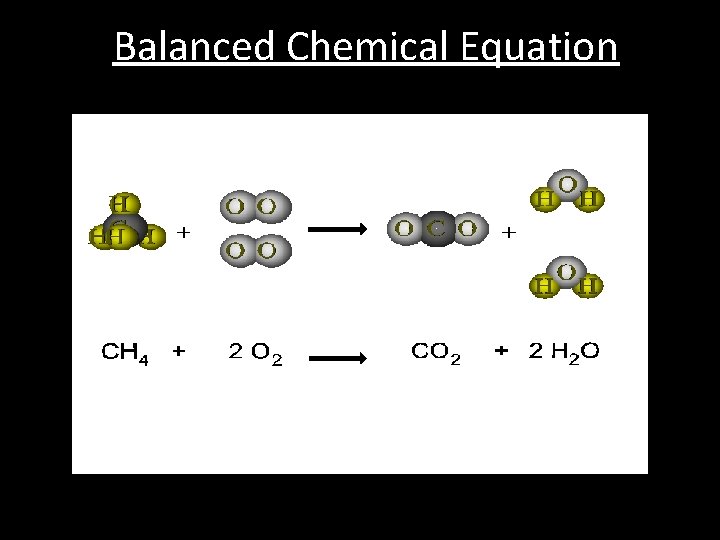
Balanced Chemical Equation
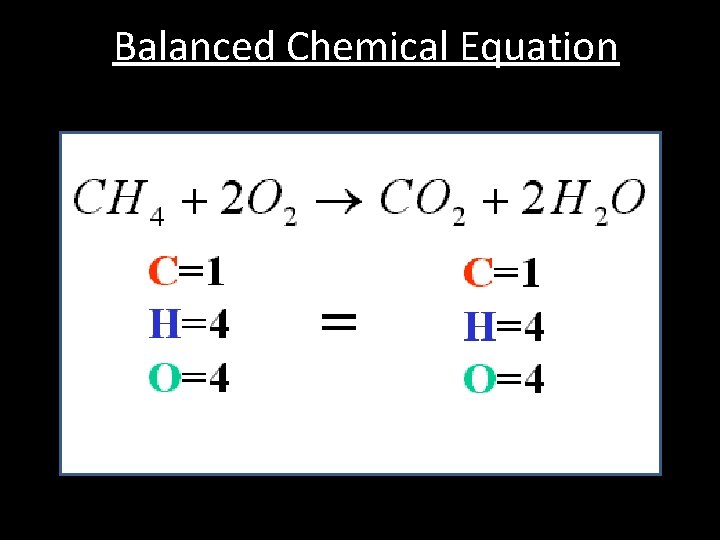
Balanced Chemical Equation

It is balanced? Potassium Permanganate + Glycerin + Oxygen Potassium Carbonate + Carbon Dioxide + Water 14 KMn. O 4 + 4 C 3 H 5(OH) 3 7 K 2 CO 3 + CO 2 + 16 H 2 O

Balancing Chemical Equations Write and balance (coefficients) the following: 1) __ H 2 + __O 2 __H 2 O 2) __Fe + __Cl 2 __Fe. Cl 3 3) __Li + __H 2 O __Li. OH + __H 2 4) __Al + __S 8 __Al 2 S 3 5) __Al(OH)3 __Al 2 O 3 + __H 2 O
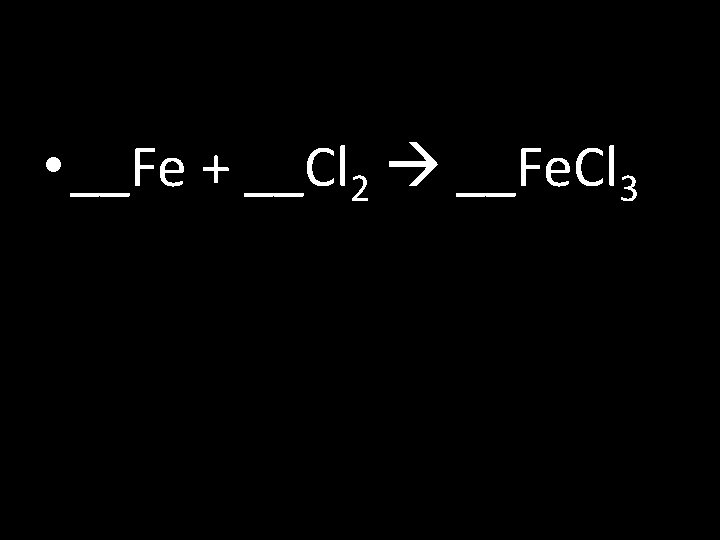
• __Fe + __Cl 2 __Fe. Cl 3
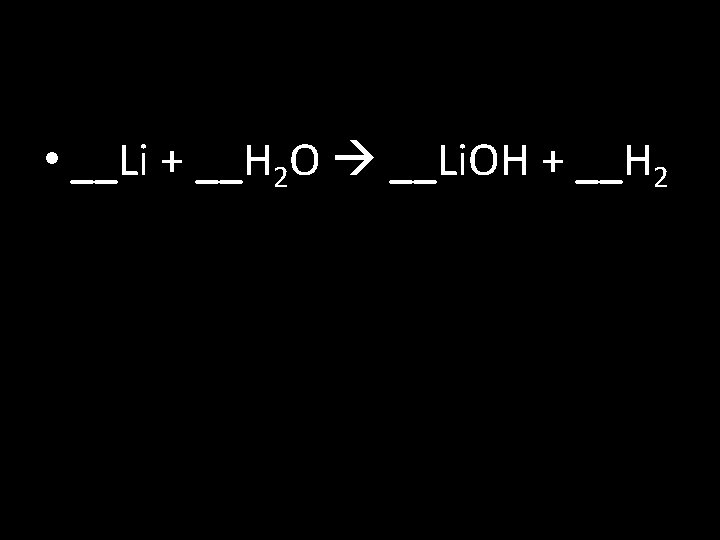
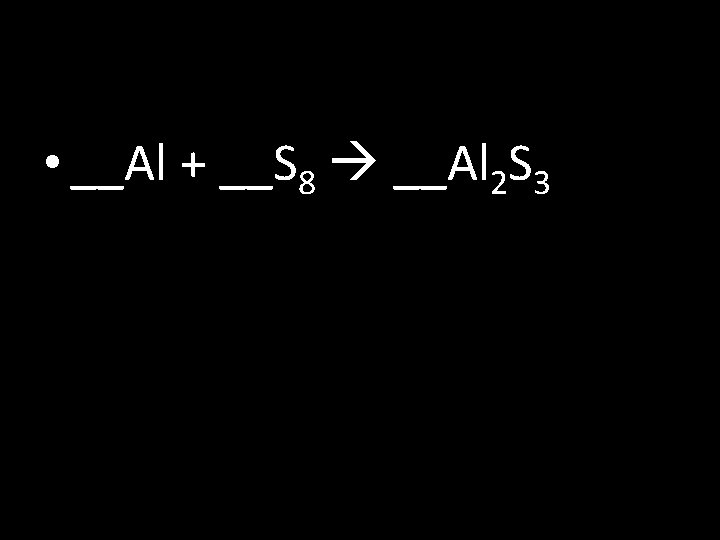
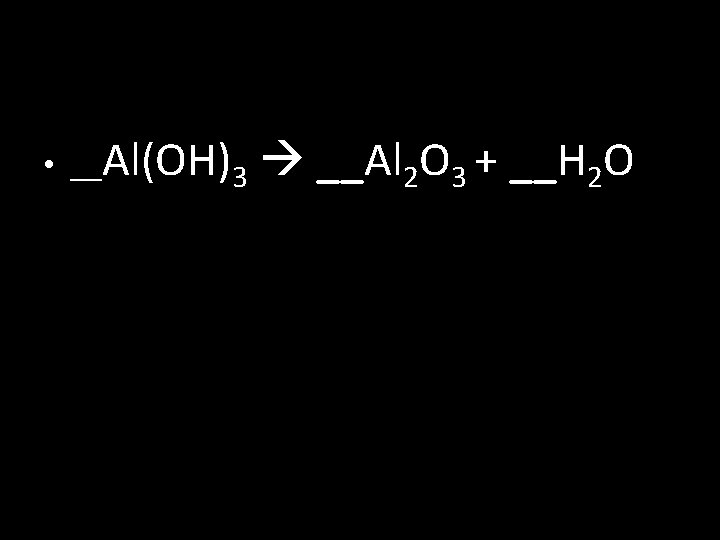
• __Al(OH)3 __Al 2 O 3 + __H 2 O

Atomic Structure List and explain all three sub atomic particles. Explain how those particles are arranged in an atom. Periodic Table Name three different ways the periodic table is organized. Explain why Hydrogen is on the left side of the Periodic Table and Helium is on the far right. Estimate how many atoms there are in the following: A penny, an apple, a bottle of water, and your body. (Try and use x 10 x exponents. ) Draw an atom of Boron, Chlorine, or Sodium. Label each part including the Compounds and Molecules nucleus and the Explain the difference between a electron cloud. molecule and a compound. Write the chemical formula for the following and label them as a Draw Lavoisier Sodium compound, molecule, or both: water, carbon dioxide, oxygen (in the air), and sugar (if you know it). Atomic # Chemical Symbol Chemical Name Atomic Mass Complete the box on the left for the element Gold. Chemical and Physical Give 5 physical properties of a nail. Give 2 chemical properties of a nail. Explain the difference between Draw Mendeleev a physical change and a chemical change. Give an example of each.

Elephant Toothpaste Write and balance (coefficients) the following: 2 H 2 O 2 → 2 H 2 O + O 2 Why is there only 1 reactor when there are four ingredients?
- Slides: 18