PURE COMPONENT CONSTANTS Week 3 Method of Marrero




![An example: CHF=CF-CF 3 is denoted by 1 pair of [=]CH- & [=]C< 1 An example: CHF=CF-CF 3 is denoted by 1 pair of [=]CH- & [=]C< 1](https://slidetodoc.com/presentation_image_h2/0422bdc45d0b49dec6dfffb1c93353f2/image-8.jpg)
![The location of bonding in esters is distinguished by the use of [ ] The location of bonding in esters is distinguished by the use of [ ]](https://slidetodoc.com/presentation_image_h2/0422bdc45d0b49dec6dfffb1c93353f2/image-9.jpg)
















- Slides: 32

PURE COMPONENT CONSTANTS Week 3

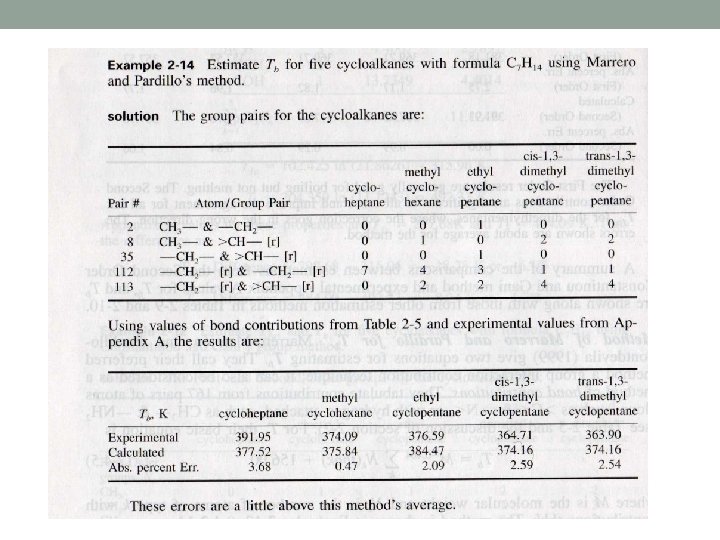
Method of Marrero and Pardillo • Bond contribution method




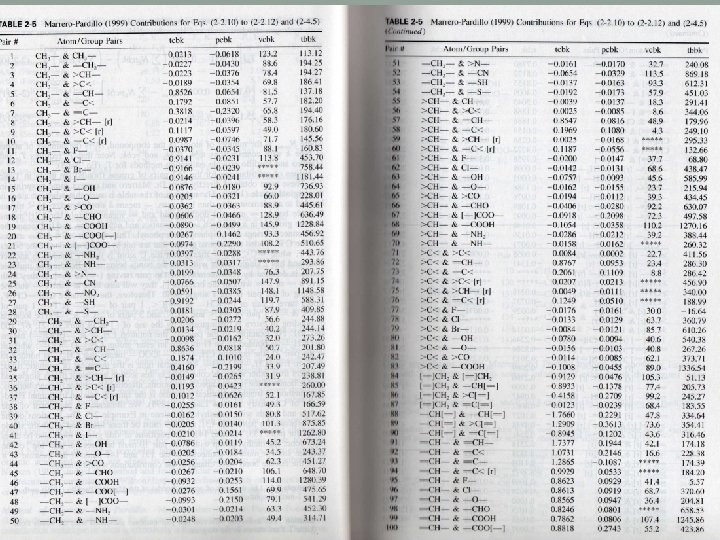
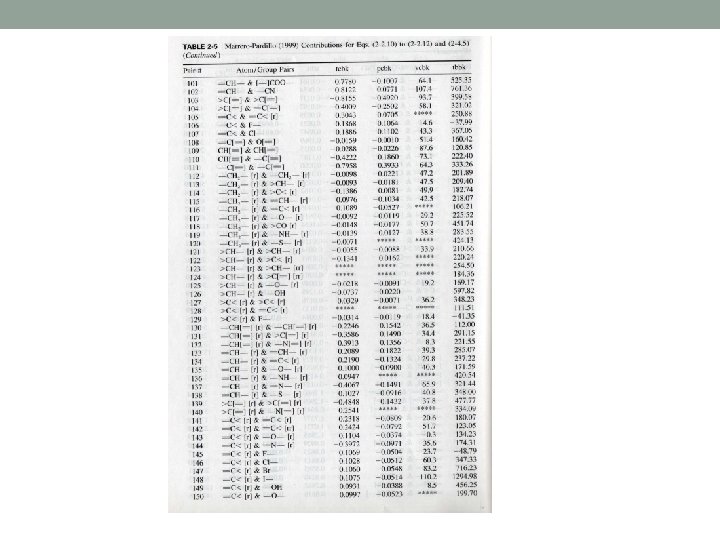
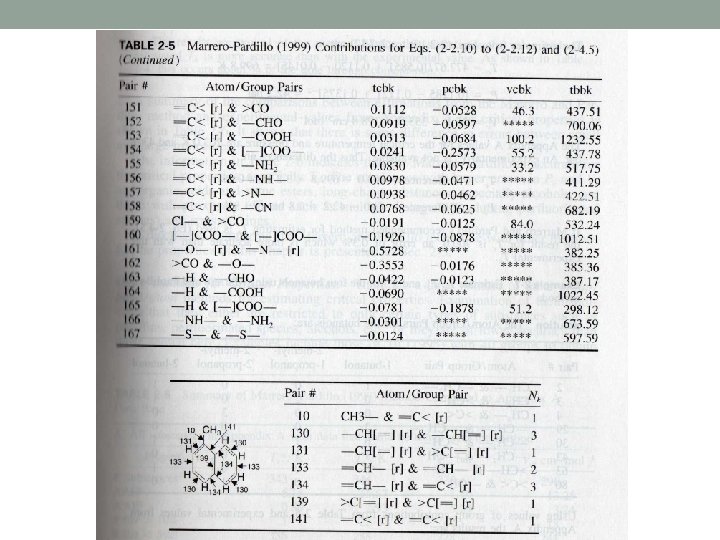
About table 2 -5 I. III. IV. V. VI. - means a single bond > or < mean two single bonds = means a double bond means a triple bond r means a ring such as aromatics and naphthenics rr means the pair of two connecting rings

>C< & F- means that the C atom is bonded to 4 atoms which are not hydrogen and one of them is F =C< & F- means that the C atom is doubly bonded to another atoms and has two single bonds which are not hydrogen and one of them is F. Bonding of multiple bonds is denoted by both members of the pair having [=] and [ ].
![An example CHFCFCF 3 is denoted by 1 pair of CH C 1 An example: CHF=CF-CF 3 is denoted by 1 pair of [=]CH- & [=]C< 1](https://slidetodoc.com/presentation_image_h2/0422bdc45d0b49dec6dfffb1c93353f2/image-8.jpg)
An example: CHF=CF-CF 3 is denoted by 1 pair of [=]CH- & [=]C< 1 pair of =CH- and F 1 pair of =C< & >C< 3 pairs of >C< & -F
![The location of bonding in esters is distinguished by the use of The location of bonding in esters is distinguished by the use of [ ]](https://slidetodoc.com/presentation_image_h2/0422bdc45d0b49dec6dfffb1c93353f2/image-9.jpg)
The location of bonding in esters is distinguished by the use of [ ] For example CH 3 - & -COO[-] means that CH 3 is bonded to a O CH 3 - & [-] COO- means that CH 3 is bonded to a C The treatment of the aromatic ring is different from other methods and the exact location of the double and single bonds are considered.



An overview of Marrero & Pardillo method This method is generally accurate, specially for critical temperature. The substances with larger errors with VC and PC are organic acids and some esters, long chain substances, especially alcohols, and substances containing multiple halogen atoms.

Comparison of the Methods for critical temperature § The methods are all broadly applicable. The average. percent error is around 1%. § If there is an experimental value for normal boiling point, method of Marrero and Pardillo is more accurate than the method of Wilson and Jasperson. § At the other hand the latter method is much simpler and covers more substances such as inorganic substances. § Joback’s method covers more substances but it is somehow more complex and less accurate. § If there is not any experimental Tb Marrero and Pardillo and Wilson and Jasperson would be more suitable for smaller compounds with 3 or less carbon atoms, while Joback’s and Constantinou and Gani are more reliable for larger molecules.

Comparison of the Methods for critical pressure and volume § All of the methods are usually reliable, but not as accurate for critical temperature. § Wilson/Jasperson method is most accurate when there is an experimental value for Tb. § All of the methods are broadly applicable Joback’s method is the most accurate (3%) and Constantiou/Gani is less accurate (4. 5%).

Acentric Factor This factor which is normally related to critical constants was originally introduced by Pitzer in 1955. He used this property to estimate thermodynamic properties of normal fluids. The following equation is used to find estimates of ZC

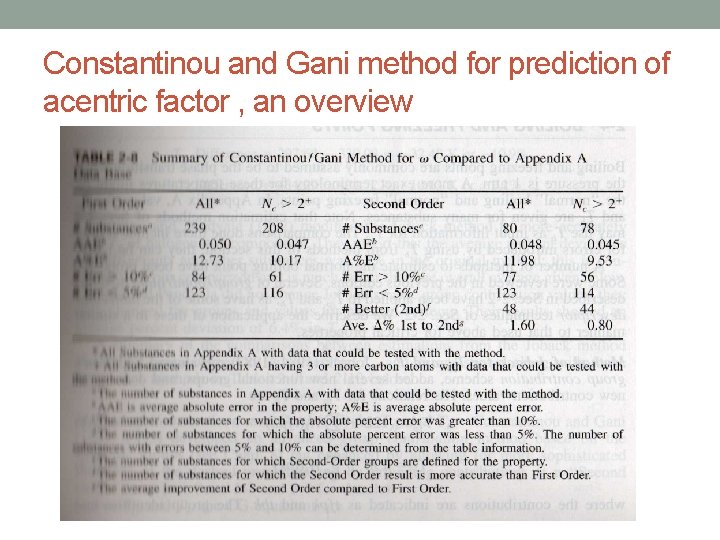
Constantinou and Gani method for prediction of acentric factor


Constantinou and Gani method for prediction of acentric factor , an overview

Boiling and Freezing points Method of Joback


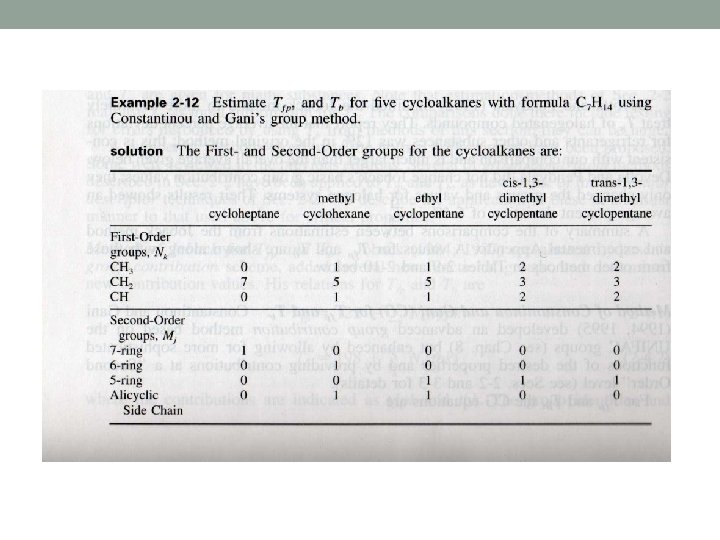
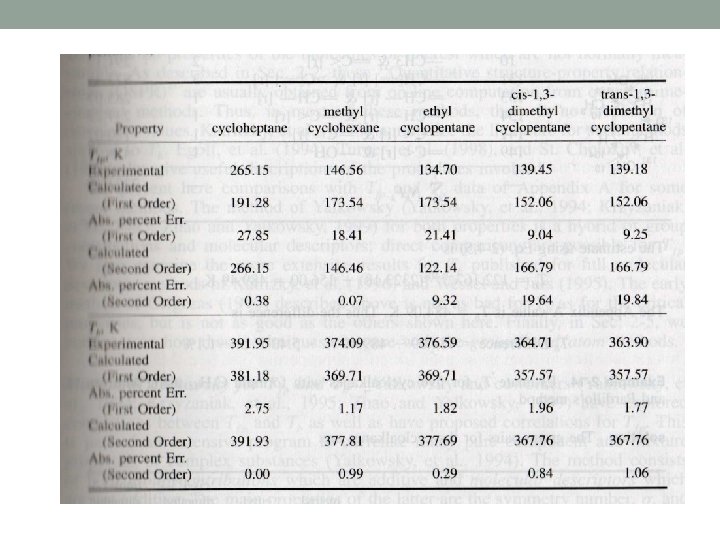
Constantinou and Gani method




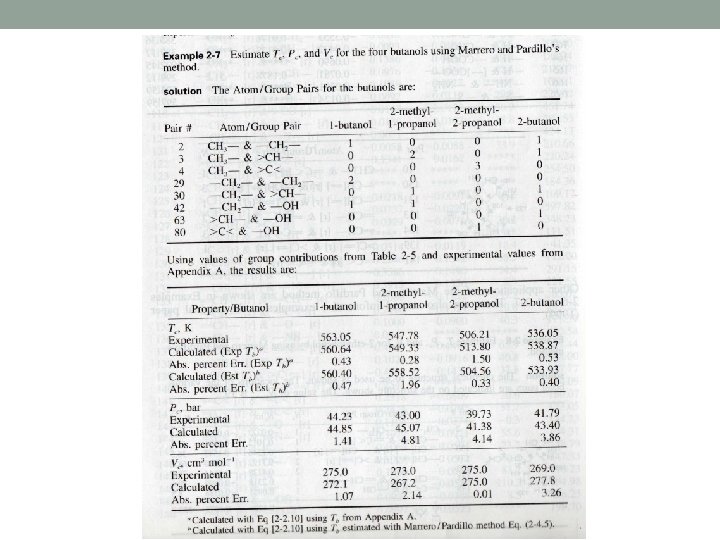
Method of Marrero and Pardillo Estimate Tc, Pc and Vc for 2 -ethylphenol by using Marrero and Pardillo method.






