PULMONARY DISEASE TREATMENT CONCERNING COPD BY MELISSA JAKUBOWSKI

PULMONARY DISEASE TREATMENT CONCERNING COPD BY MELISSA JAKUBOWSKI

AGENDA üIntroduce the journal article üReview of preliminary background üDiscuss the details of the study üEvaluation of the study üDiscuss how the implications of the study translate into practice üQuestions

JOURNAL ARTICLE • Efficacy of corticosteroid therapy in patients with an acute exacerbation of chronic obstructive pulmonary disease receiving ventilatory support

THIS STUDY ASSESSED: • The effect of systemic corticosteroids on ICU patients who were mechanically ventilated (invasive or noninvasive) due to COPD exacerbations

IMPORTANCE • Previous randomized trials assessing the effect of systemic corticosteroid therapy on COPD exacerbations have excluded patients with respiratory failure who required mechanical ventilation or ICU admission • Critically ill patients in the ICU are more prone to develop complications potentially associated with corticosteroid therapy

HOSPITAL TREATMENT FOR COPD • Bronchodilators • Antibiotics • Oxygen • Corticosteroids

COPD AND CORTICOSTEROID THERAPY • COPD: airways damaged interfering with the exchange of oxygen and carbon dioxide in the lungs • Function: reduce lung and bronchial tube inflammation related to tissue damage and improve respiratory function
SHORT-TERM SIDE EFFECTS • Increased appetite • Fluid retention • Weight gain • Increased blood pressure • Hyperglycemia
SYSTEMIC STEROIDS, CORTICOSTEROIDS • Prednisone • Prednisolone • Methylprednisolone • • • Betamethasone Beclamethasone Dexamethasone Flurocortisone Hydrocortisone Triamcinolone

SCREENING FOR SUBJECTS • Screening: 354 patients from 8 hospitals in 4 countries from July 2005 – July 2009 • Criteria: hospitalization because of exacerbation of COPD requiring ventilatory support in the ICU • Controls: diagnosis of “exacerbation of COPD” was explicitly defined; patients with conditions that would alter integrity of study were excluded

ETHICS • Approved by the ethics committee at each hospital • Written informed consent obtained from patients or their surrogates
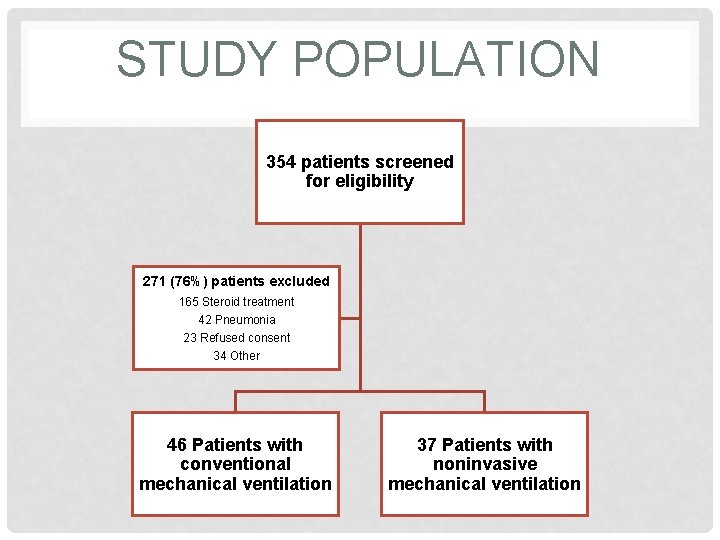
STUDY POPULATION 354 patients screened for eligibility 271 (76%) patients excluded 165 Steroid treatment 42 Pneumonia 23 Refused consent 34 Other 46 Patients with conventional mechanical ventilation 37 Patients with noninvasive mechanical ventilation
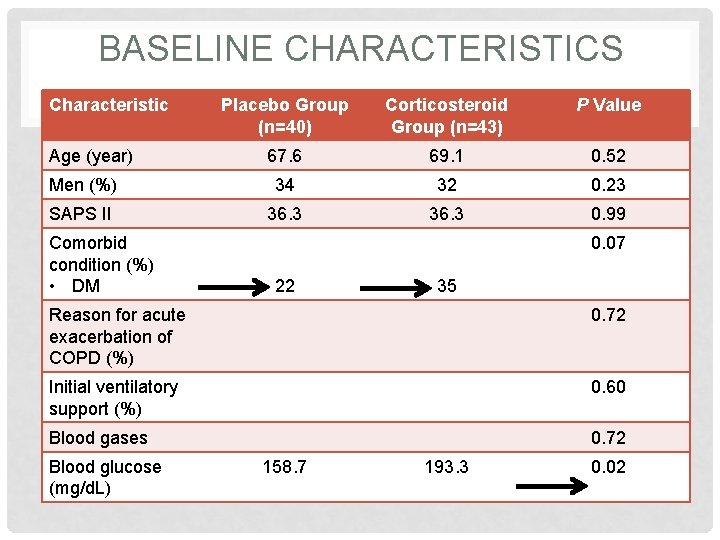
BASELINE CHARACTERISTICS Characteristic Placebo Group (n=40) Corticosteroid Group (n=43) P Value 67. 6 69. 1 0. 52 Men (%) 34 32 0. 23 SAPS II 36. 3 0. 99 Age (year) Comorbid condition (%) • DM 0. 07 22 35 Reason for acute exacerbation of COPD (%) 0. 72 Initial ventilatory support (%) 0. 60 Blood gases 0. 72 Blood glucose (mg/d. L) 158. 7 193. 3 0. 02
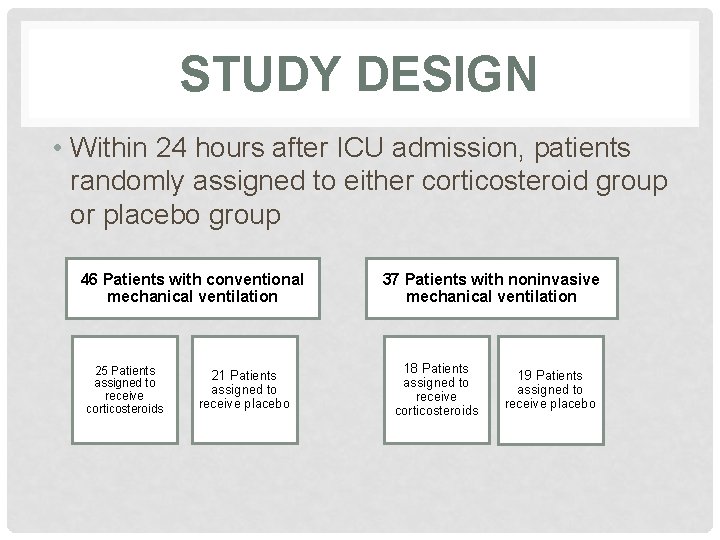
STUDY DESIGN • Within 24 hours after ICU admission, patients randomly assigned to either corticosteroid group or placebo group 46 Patients with conventional mechanical ventilation 25 Patients assigned to receive corticosteroids 21 Patients assigned to receive placebo 37 Patients with noninvasive mechanical ventilation 18 Patients assigned to receive corticosteroids 19 Patients assigned to receive placebo

LENGTH OF ADMINISTRATION: 10 DAYS Treatment Group • Methylprednisolone Placebo Group • Normal saline solution

ADMINISTRATION REGIMEN Days 1 -3 Day 4 -6 Days 7 -10 • 0. 5 mg/kg every 6 hours • 0. 5 mg/kg every 12 hours • 0. 5 mg/kg per day

OUTCOME MEASURES • Duration of mechanical ventilation, d • Length of ICU stay, d • Length of hospital stay, d • In-ICU mortality, % • Failure of NIMV, % • Reintubation within 48 hours of planned extubation and received CMV either initially or after failure of NIMV, %

FREQUENCY OF ADVERSE EVENTS • Superinfection • Gastrointestinal bleeding • Arterial hypertension • Hyperglycemia • Ventilator-associated pneumonia • Delirium • ICU-acquired paresis

OTHER DAILY MEASUREMENTS • ABG analysis • C-reactive protein • Maximal blood glucose level • Daily dose of insulin • PEEP (positive end-expiratory pressure): only in patients who were intubated
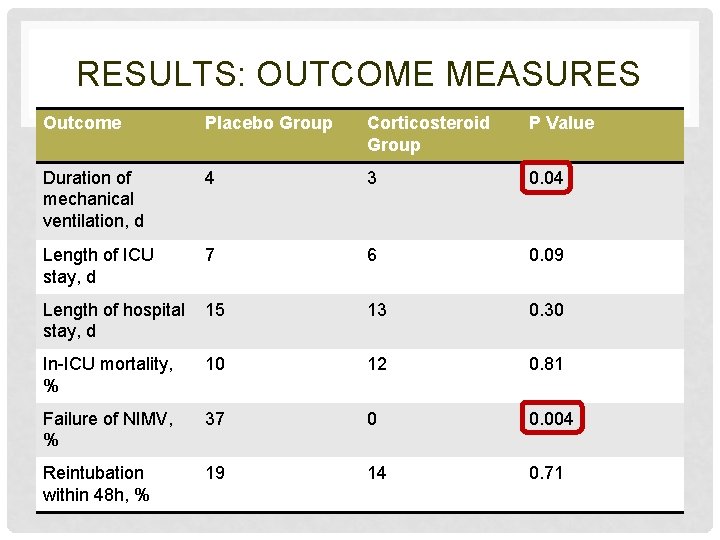
RESULTS: OUTCOME MEASURES Outcome Placebo Group Corticosteroid Group P Value Duration of mechanical ventilation, d 4 3 0. 04 Length of ICU stay, d 7 6 0. 09 Length of hospital stay, d 15 13 0. 30 In-ICU mortality, % 10 12 0. 81 Failure of NIMV, % 37 0 0. 004 Reintubation within 48 h, % 19 14 0. 71
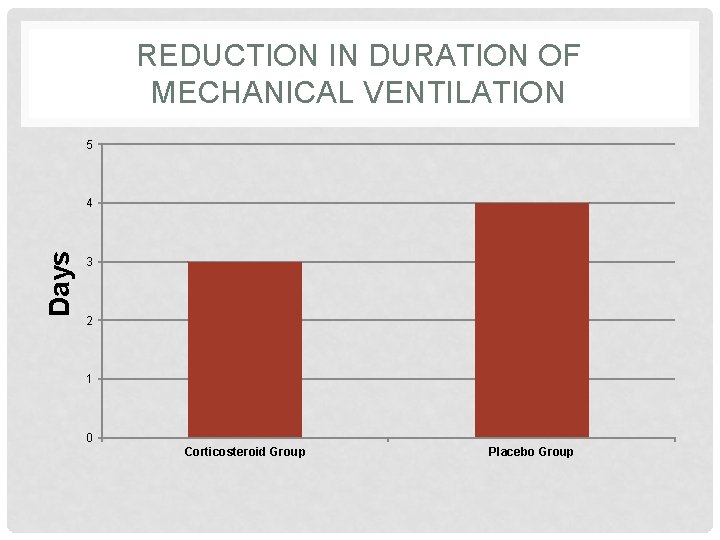
REDUCTION IN DURATION OF MECHANICAL VENTILATION 5 Days 4 3 2 1 0 Corticosteroid Group Placebo Group
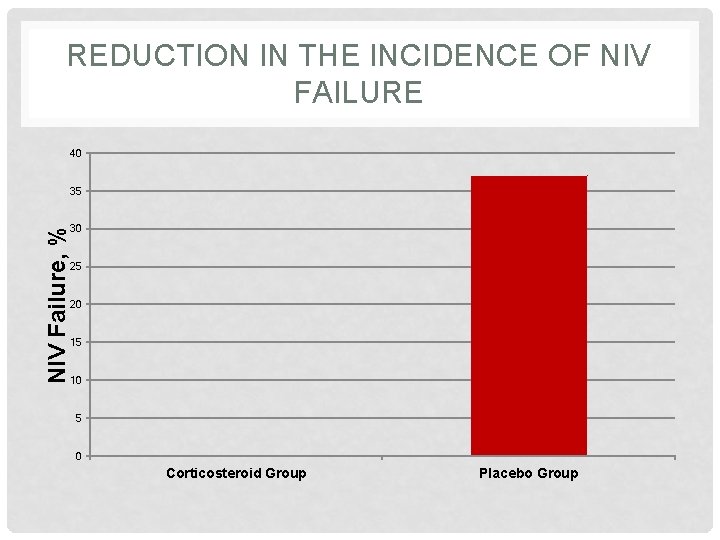
REDUCTION IN THE INCIDENCE OF NIV FAILURE 40 NIV Failure, % 35 30 25 20 15 10 5 0 Corticosteroid Group Placebo Group
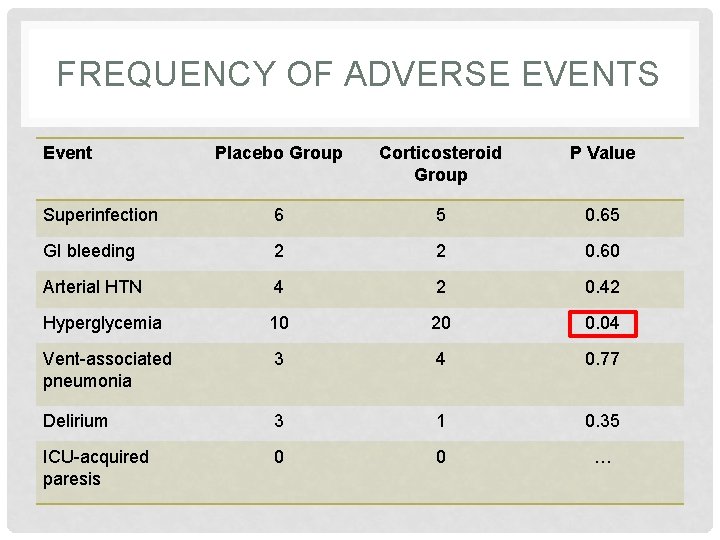
FREQUENCY OF ADVERSE EVENTS Event Placebo Group Corticosteroid Group P Value Superinfection 6 5 0. 65 GI bleeding 2 2 0. 60 Arterial HTN 4 2 0. 42 Hyperglycemia 10 20 0. 04 Vent-associated pneumonia 3 4 0. 77 Delirium 3 1 0. 35 ICU-acquired paresis 0 0 …

PROS VS. CONS Pros • Modest reduction in the duration of mechanical ventilation • Increased success of NIMV • Trend towards shorter ICU stay Cons • Hyperglycemia

EVALUATION OF THE STUDY: PROS • 1 st study to confirm the benefits of systemic corticosteroid therapy for ICU patients receiving MV for COPD exacerbation • Validates its usage in clinical practice today • Double-blinded experiment • Source of funding

PROS (CONTINUED) • Funded by University of Vall d’Hebron Hospital, Laboratory of Experimental Cardiology (LEC) • LEC mission statement: “to contribute to lessen the impact of cardiovascular diseases on survival capacity and quality of life of the general population by elucidating the mechanisms of disease and proposing new treatments”

EVALUATION OF THE STUDY: CONS • Results will not have an impact on current clinical treatment practice guidelines • Sample size was small (83) • Length of study (5 years) • Does not mention the effect of the drug on eating behavior

RESOURCE • Alia I, de la Cal MA, Esteban A, et al. Efficacy of corticosteroid therapy in patients with an acute exacerbation of chronic obstructive pulmonary disease receiving ventilatory support. Arch Intern Med. 2011; 171(21): 1939 -1946. Accessed April 13, 2012.

ADDITIONAL THOUGHTS • What about corticosteroid long-term side-effects? • Optimal dose and length of treatment? • How do the findings translate into practice?

QUESTIONS
- Slides: 30