Published Data Randomized Doubleblind Placebocontrolled Trial Remdesivir for

Published Data — Randomized, Double-blind, Placebo-controlled Trial Remdesivir for the Treatment of Covid-19 ACTT-1—Final Report (Multinational) Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19 (ACTT-1): Study Design • Background: A multicenter, double-blind, randomized, placebo-controlled trial of intravenous remdesivir in adults hospitalized with Covid-19 with evidence of lower respiratory tract involvement during February 21 -April 19, 2020. • Location: 73 sites in the United States, Europe, Asia, and Mexico • Inclusion Criteria (n = 1062) - Age ≥ 18 years - Hospitalized - Lab confirmed SARS-Co. V-2 (PCR or other public health assay) <72 hours of enrollment - One or more of the following: - Pulmonary infiltrates on chest imaging - Rales or crackles on exam AND Sp. O 2 ≤ 94% on room air - Requiring mechanical ventilation or supplementary oxygen • Exclusion Criteria - Pregnant or breastfeeding - AST/ALT >5 x ULN - e. GFR <30 Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19 (ACTT-1): Study Design • Primary Outcome: Time to recovery (discharge or no longer requiring supplemental O 2)* • Secondary outcomes: - Mortality at days 14 and 28 - Grade 3 or 4 adverse events, or severe adverse events * This primary outcome was changed from an earlier primary outcome of recovery at day 15 as evolving clinical information showed the prolonged course of COVID-19 disease. None of the preliminary data was known at the time of this decision Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19 (ACTT-1): Study Design Arms and Interventions (1: 1 randomization stratified by disease severity and site) Remdesivir 200 mg loading dose on day 1, then 100 mg maintenance dose daily on days 2 -10 (n = 541) or Placebo Volume equivalent loading and maintenance doses (n = 521) *If the hospital had a written policy or guideline, participants could receive other experimental or off-label treatments for COVID-19. Otherwise, other specific treatments were prohibited from study day 1 through day 29. Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19 (ACTT-1): Safety • In the remdesivir arm, 98. 2% received the drug as assigned - 36 patients discontinued due to an adverse event • In the placebo arm, 99. 2% completed infusions as assigned - 36 patients discontinued due to an adverse event Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19 (ACTT-1): Baseline Characteristics Remdesivir (n = 541) Placebo (n = 521) Age, years (mean ± SD) 58. 6 ± 14. 6 59. 2 ± 15. 4 Male, n (%) 352 (65. 1) 332 (63. 7) Hypertension 269/532 (50. 6) 264/519 (50. 9) Obesity 242/531 (45. 6) 234/518 (45. 2) Type 2 Diabetes 164/532 (30. 8) 158/519 (30. 4) 9 (6 - 12) 9 (7 - 13) Coexisting conditions, n/total (%) Duration of symptoms prior to randomization, days (median, IQR) Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]
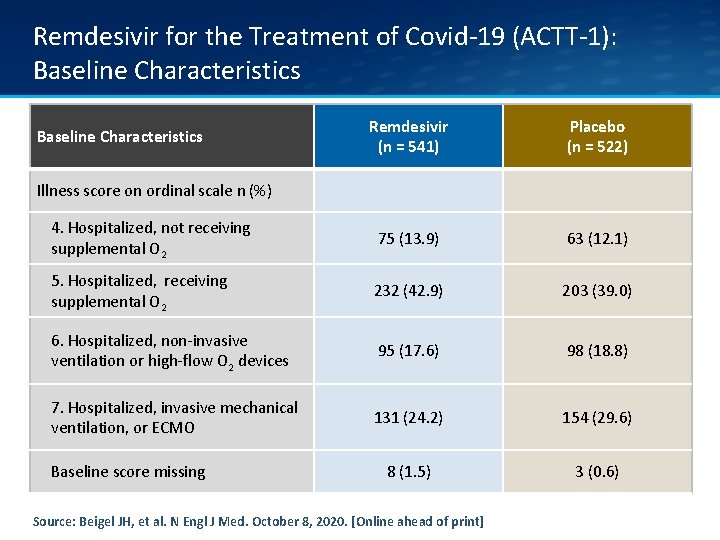
Remdesivir for the Treatment of Covid-19 (ACTT-1): Baseline Characteristics Remdesivir (n = 541) Placebo (n = 522) 4. Hospitalized, not receiving supplemental O 2 75 (13. 9) 63 (12. 1) 5. Hospitalized, receiving supplemental O 2 232 (42. 9) 203 (39. 0) 6. Hospitalized, non-invasive ventilation or high-flow O 2 devices 95 (17. 6) 98 (18. 8) 7. Hospitalized, invasive mechanical ventilation, or ECMO 131 (24. 2) 154 (29. 6) 8 (1. 5) 3 (0. 6) Baseline Characteristics Illness score on ordinal scale n (%) Baseline score missing Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19 (ACTT-1): Results • Median time to recovery was significantly different: - Remdesivir 10 days (95% confidence interval [CI], 9 to 11) - Placebo 15 days (95% CI, 13 to 18) in those who received placebo - Rate ratio for recovery, 1. 29; 95% CI, 1. 12 to 1. 49; P<0. 001) Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]
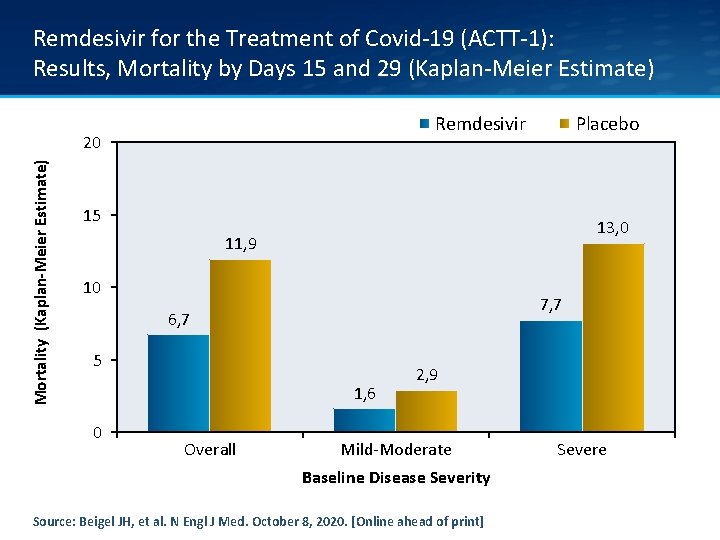
Remdesivir for the Treatment of Covid-19 (ACTT-1): Results, Mortality by Days 15 and 29 (Kaplan-Meier Estimate) Remdesivir Mortality (Kaplan-Meier Estimate) 20 Placebo 15 13, 0 11, 9 10 7, 7 6, 7 5 1, 6 0 Overall 2, 9 Mild-Moderate Baseline Disease Severity Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print] Severe
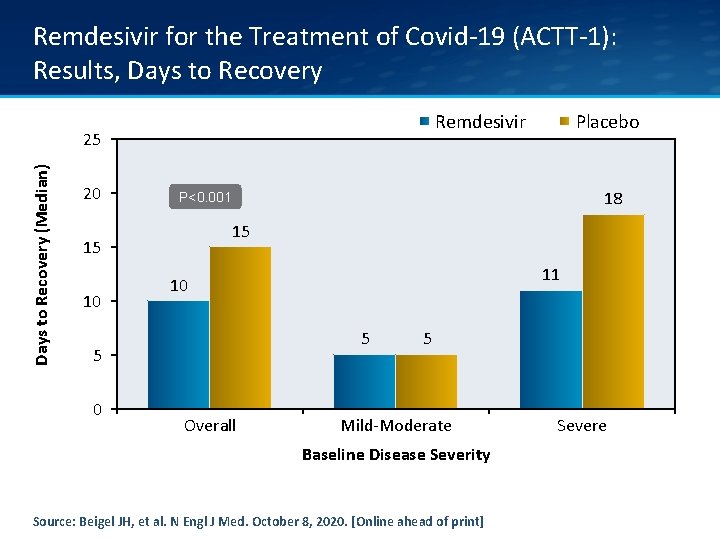
Remdesivir for the Treatment of Covid-19 (ACTT-1): Results, Days to Recovery Remdesivir Days to Recovery (Median) 25 20 18 P<0. 001 15 15 10 11 10 5 5 0 Placebo Overall 5 Mild-Moderate Baseline Disease Severity Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print] Severe
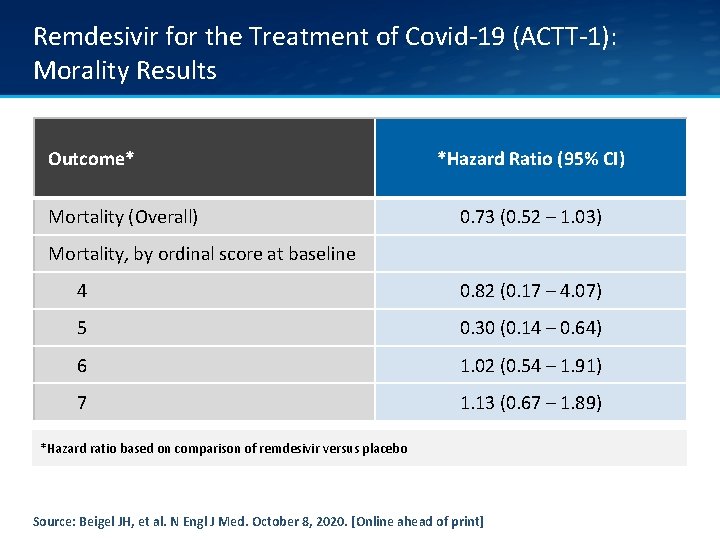
Remdesivir for the Treatment of Covid-19 (ACTT-1): Morality Results Outcome* Mortality (Overall) *Hazard Ratio (95% CI) 0. 73 (0. 52 – 1. 03) Mortality, by ordinal score at baseline 4 0. 82 (0. 17 – 4. 07) 5 0. 30 (0. 14 – 0. 64) 6 1. 02 (0. 54 – 1. 91) 7 1. 13 (0. 67 – 1. 89) *Hazard ratio based on comparison of remdesivir versus placebo Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]

Remdesivir for the Treatment of Covid-19: Authors’ Conclusions: “Our data show that remdesivir was superior to placebo in shortening the time to recovery in adults who were hospitalized with Covid-19 and had evidence of lower respiratory tract infection. ” Source: Beigel JH, et al. N Engl J Med. October 8, 2020. [Online ahead of print]
- Slides: 12