PublicPrivate Product Development Partnerships for Global Health RD

Public-Private Product Development Partnerships for Global Health R&D: Issues & Challenges Suerie Moon Giorgio Ruffolo Doctoral Fellow in Sustainability Science & Doctoral Candidate in Public Policy Center for International Development, Harvard Kennedy School of Government suerie_moon@hksphd. harvard. edu Presentation to: Designing Strategies for Neglected Disease Research Spring 2009 UC Berkeley: Law 284. 26/Public Policy 290, 190 Professors Stephen Maurer & Amy Kapczynski 10 March 2009

Overview 1. Introduction: History, Organizational Form, and Funding 2. Strengths and Weaknesses 3. Targeted Diseases 4. Access Provisions: Policies & IP Management 5. PDPs in the broader R&D Ecosystem 6. Governance Issues 7. Conclusions

1. 1 Introduction: History of global health product development 1900 s-1950 s: National R&D efforts led to ‘trickle-down’ approach 1960 s-80 s: International bifurcated public/private system n Public: e. g. UNICEF-UNDP-World Bank-WHO Special Programme for Research and Training in Tropical Diseases (TDR) n Private: e. g. Industry, globalizing intellectual property rights system 1990 s: Global health & neglected diseases n Linkages: research, health, development n Medicines as global public good: access to AIDS treatment n Global health needs: 10/90 Gap and neglected diseases 2000 s: Global health for all? n New system for Neglected Diseases (Type III): n n PDPs “a fad and a sacred cow”? Trust and networks built; low-hanging fruit harvested Honeymoon period over? Diseases of global incidence? (Type I)
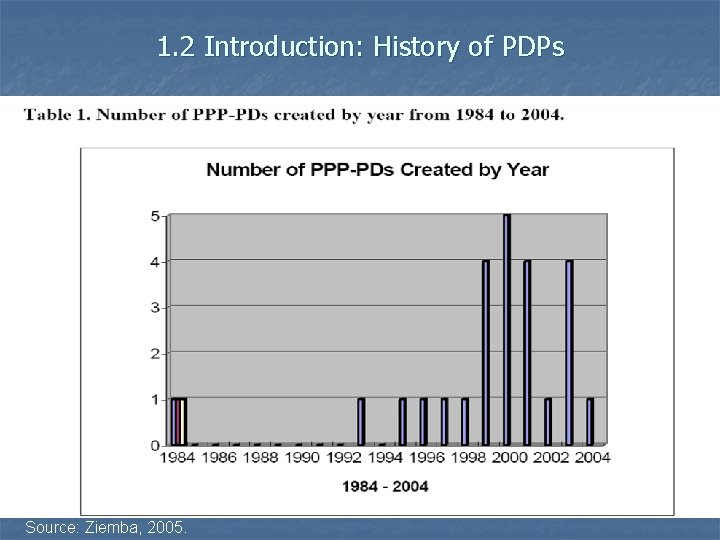
1. 2 Introduction: History of PDPs Source: Ziemba, 2005.

1. 3 Introduction: Definition & Examples of PDPs Definition: Public health driven not-for-profit organisations that drive neglected disease drug [product] development in conjunction with industry groups (Moran et al. , 2005) n n n Aeras: Global Tuberculosis Vaccine Foundation BVGH: BIO Ventures for Global Health CONRAD: Contraceptive Research and Development Program CICCR: Consortium for Industry Collaboration in Contraceptive Research DNDI: Drugs for Neglected Diseases initiative EMVI: European Malaria Vaccine Initiative FIND: Foundation for Innovative New Diagnostics Gates/UNC: Gates Foundation/University of North Carolina Partnership for the Development of New Drugs GMP: Global Microbicide Project HHVI: Human Hookworm Vaccine Initiative IAVI: International AIDS Vaccine Initiative Source: Ziemba 2005 n n n n IDRI: Infectious Disease Research Institute IOWH: Institute for One. World Health IPM: International Partnership for Microbicides LAPDAP: LAPDAP Antimalarial Product Development MDP: Microbicides Development Program MMV: Medicines for Malaria Venture MVI: Malaria Vaccine Initiative at PATH MVP: Meningitis Vaccine Project at PATH (Program on Technologies for Health) PDVI: Pediatric Dengue Vaccine Initiative Pneumo. ADIP: Pneumococcal Vaccines Accelerated Development and Introduction Plan Rota. ADIP: Rotavirus Vaccine Program SAAVI: South African AIDS Vaccine Initiative TB Alliance: Global Alliance for Tuberculosis Drug Development

1. 4 Introduction: Basic PDP model n n n PDP is a non-profit entity that manages a globallydispersed portfolio for a disease. Generalize with great caution PDPs vary on: n n n n n One disease or multiple Breadth or depth in portfolio management External or in-house research & production capacity Relative emphasis on public vs private sector Level and diversity of funding sources Definition of core and secondary missions Approach to intellectual property management Governance structures and styles Scientific & political challenges faced

1. 5 Introduction: Basic PDP model n Frequently-conducted functions include: n n n n n Finances research ($ to and from private & public sectors) Negotiates access to private sector compounds, experts, labs Reduces risk of projects Offers reputational benefits, CSR, employee morale Provides access to research with profitable spillovers (Amyris) Liaises with developing countries re: clinical trials & delivery Helps open up new emerging markets Focuses on adaptedness and affordability Advocates for more R&D Sometimes: n Conducts in-house research (Aeras, IAVI)

1. 6 Introduction: ND R&D Funding to PDPs Of $2. 5 billion invested in ND R&D in 2007, 23% ($575 mln) routed through PDPs. Source: From: Neglected Disease Research and Development: How Much Are We Really Spending? Moran M, Guzman J, Ropars AL, Mc. Donald A, Jameson N, et al. PLo. S Medicine Vol. 6, No. 2, e 30 doi: 10. 1371/journal. pmed. 1000030
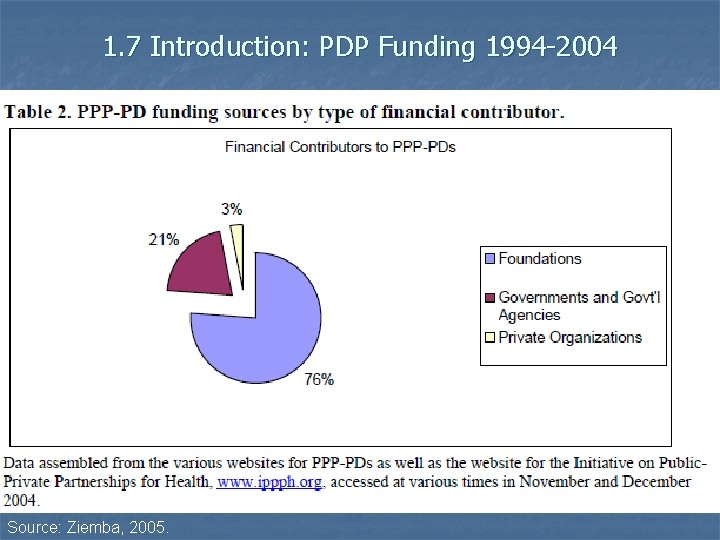
1. 7 Introduction: PDP Funding 1994 -2004 Source: Ziemba, 2005.

1. 8 Introduction: PDP Funding n n n Neglected disease research: $2. 5 billion in 2007 Overall global health research: $125 billion/yr Largely financed by bilateral donors and foundations, Gates in particular Gates is majority funder of many PDPs (Aeras, FIND, Io. WH) Funding short term & uncertain: n n n Long-term strategic planning difficult Missed opportunities when rapid reaction needed Weakened negotiating leverage due to risk Dramatically increased future needs as products move through clinical trials Answers? : n n Profit-making to underwrite research? Auctioning assets e. g. using PRV? UNITAID-type global tax? CGIAR-type donor support?

Overview 1. Introduction: History, Organizational Form, and Funding 2. Strengths and Weaknesses 3. Targeted Diseases 4. Access Provisions: Policies & IP Management 5. PDPs in the broader R&D Ecosystem 6. Governance Issues 7. Conclusions

2. 1: Strengths & Weaknesses: Challenges for Evaluation Evaluating individual PDPs difficult because: • Varying disease profiles • Scientific • Economic • Political • Long lead times • Complex expectations • Varied approaches For a full discussion of the challenges of evaluating PDPs, see “Toward a New Approach to Product Development Partnership Performance Measurement, ” June 2007, by FSG Social Impact Advisors.

2. 2: Strengths & Weaknesses: What does the literature say? Strengths: Weaknesses: n n Renewed ND R&D n n n From 2000 -2005: 0 to 63 drug projects = 8 or 9 new drugs expected Affordability & adaptedness as central criteria for new products Harnessing private sector capacity for public ends (CSR) Light networked structure Global coordination of scarce investments n n n Untested institutional model: will it work? Financial sustainability unclear Governance unclear (accountability, decision-making, transparency) Developing country participation is limited Risk that private interests undermine public goals Limited to ND (diseases without a market) Sources: Kettler & Towse (2001), Ziemba (2005), Moran et al. (2005), Widdus (various), Buse (various), Turner & Makgoba (2008), Moon (forthcoming)

2. 3: Strengths & Weaknesses: Selected Achievements* New products: 1. Paramomycin (V. leishmaniasis in India) 2. ASAQ (malaria) 3. ASMQ (malaria) 4. Pediatric AR-LU (Coartem for malaria) Phase III clinical trials/equiv: 1. 2. 3. 4. 5. 6. 7. *Based on study of only 8 PDPs – list is non-exhaustive. Moxifloxacin (TB) Pyronaridine+AR (malaria) Dihydroartemisinin (malaria) Paramomycin (V. leishmaniasis in Africa) Fast. Plaque diagnostic (TB) LAM-based diagnostic (TB) Synthetic artemisinin (malaria)

2. 4: Strengths & Weaknesses: Open Questions • How many PDPs? • One disease vs. Many? • Compete vs Collaborate? • Push vs Pull mechanisms? • Access/affordability vs. Sustainability? • Efficiency vs. Capacity building? • Product development vs Delivery?

Overview 1. Introduction: History, Organizational Form, and Funding 2. Strengths and Weaknesses 3. Target Diseases 4. Access Provisions: Policies & IP Management 5. PDPs in the broader R&D Ecosystem 6. Governance Issues 7. Conclusions

3. 1: Target Diseases: Overview Dengue: PDVI Diarrheal Disease: IOWH, Rotavirus: Rota. ADIP Family Planning: PATH, CONRAD, CICCR HIV: IAVI, SAAVI Hookworm: HHVI Malaria: MMV, EMVI, Io. WH, DNDi, Lap. Dap, FIND Meningitis: MVP Microbicides (HIV & STIs): IPM, MDP, GMP Pneumonia: Pneumo. ADIP Tropical Neglected Diseases (Multiple): IOWH, DNDi, BVGH, FIND, Gates/UNC, IDRI TB: Aeras, FIND, GATB

3. 2 Target Diseases: Priority-Setting n n Among diseases: no norms for how to measure needs and translate into investments Within diseases: PDP Scientific Advisory bodies

Overview 1. Introduction: History, Organizational Form, and Funding 2. Strengths and Weaknesses 3. Target Diseases 4. Access Provisions 5. PDPs in the broader R&D Ecosystem 6. Governance Issues 7. Conclusions

4. 1: Access provisions: Affordability a central part of the mission: n But, most access provisions confidential (“trust me” approach): why? n n Fear of loss of competitive advantage among firms Loss of negotiating leverage for PDP Compromised patent application for firms Lack of detailed information on compound in early stages

4. 2: Access provisions: Strategies n n Limited Experience Access strategies include: n Global market segmentation (tiered pricing) n n n Country categories often undefined Challenge: Middle income countries Public sector vs private markets in LMICs n n n e. g. TB drugs in India Exclusive licensing Cost audits Target prices Open access (no patent or multiple licenses) n DNDi

2. 3: Strengths & Weaknesses: Selected Achievements* New products: 1. Paramomycin (V. leishmaniasis in India) 2. ASAQ (malaria) 3. ASMQ (malaria) 4. Pediatric AR-LU (Coartem for malaria) Phase III clinical trials/equiv: 1. 2. 3. 4. 5. 6. 7. *Based on study of only 8 PDPs – list is non-exhaustive. Moxifloxacin (TB) Pyronaridine+AR (malaria) Dihydroartemisinin (malaria) Paramomycin (V. leishmaniasis in Africa) Fast. Plaque diagnostic (TB) LAM-based diagnostic (TB) Synthetic artemisinin (malaria)

4. 3: Access provisions: Intellectual Property Management Ideally PDPs get access to desired compounds & technologies and maximize control over IP. Practically, will depend on negotiating leverage: n Size of firm, n Stage of development, n Disease area, n PDP n Who paid? How to strengthen leverage: n Priority review voucher? n Mandatory donor access provisions? n Reliable, plentiful funding?
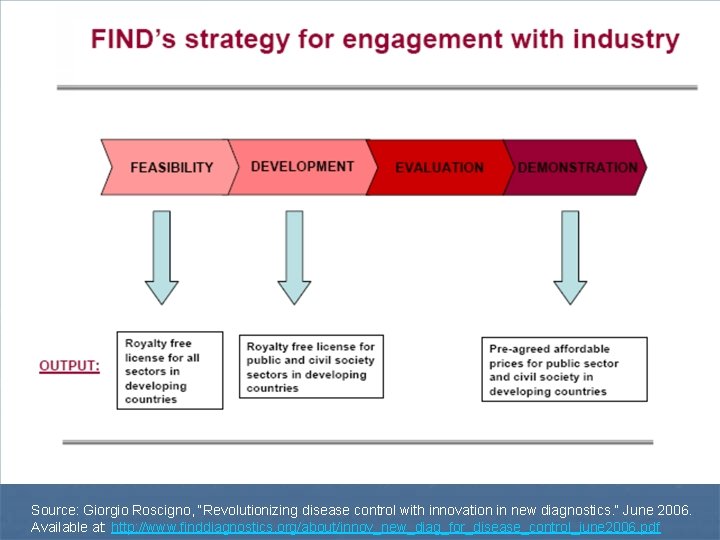
Source: Giorgio Roscigno, “Revolutionizing disease control with innovation in new diagnostics. ” June 2006. Available at: http: //www. finddiagnostics. org/about/innov_new_diag_for_disease_control_june 2006. pdf

4. 4: Access provisions: IP Management (cont’d) Generic Competition & Affordability: Factors to consider n Non-patent barriers to entry: eg technology transfer n Economies of scale & competitor’s cost audits n License to multiple producers when significant benefits expected Follow-on innovation: n No norm (yet) of open access n Information sharing across PDPs n “Giant sucking sound”: Unequal contributions by PDPs & private actors?

Overview 1. Introduction: History, Organizational Form, and Funding 2. Strengths and Weaknesses 3. Targeted Diseases 4. Access Provisions: Policies & IP Management 5. PDPs in the broader R&D Ecosystem 6. Governance Issues 7. Conclusions

5. 1 PDPs in the R&D Ecosystem n n Delicate balance: charitable vs profitable enterprise Effects of new market-based incentives? (AMC, PRV): n n n Firms may seek market-value remuneration if available Small firms may enter disease areas and reduce PDPs’ ability to build portfolios and synergies across firms/research groups; Priorities of PDPs may shift toward products that can generate revenue to keep an organization running; PDPs may have stronger leverage with PRV PDPs may be reluctant to share information with each other if competing for the same valuable reward (e. g. PRV).

5. 2 R&D Ecosystem: Unintended Consequences of Market-based Incentives “We note that commercialising low-value neglected disease markets, for example, through the use of advance purchase commitments or roaming patent extensions, is likely to increase industry activity (particularly by small companies), but at the cost of curtailing these positive behaviours and returning R&D to the more secretive and non-collaborative approaches that are characteristic of commercial R&D. ” (Moran et al. , 2005) n n Prizes, AMC, PRV could make a big difference, but… Careful attention to system-wide effects of new incentives

Overview 1. Introduction: History, Organizational Form, and Funding 2. Strengths and Weaknesses 3. Targeted Diseases 4. Access Provisions: Policies & IP Management 5. PDPs in the broader R&D Ecosystem 6. Governance Issues 7. Conclusions

6. 1 Governance: Transparency Transparent re: n Composition of Board, Scientific Advisory, Stakeholders, Staff, Limited transparency re: n Funding sources n Budgets & Spending Very little transparency re: n Decision-making procedures n Agreements between PDPs and industry n Cost of R&D How much transparency should we expect or demand from a hybrid public-private entity? From a non-profit organization?

6. 2 Governance: Endemic country involvement n n n Clinical trials Research Production Governance BRICS vs LDCs?
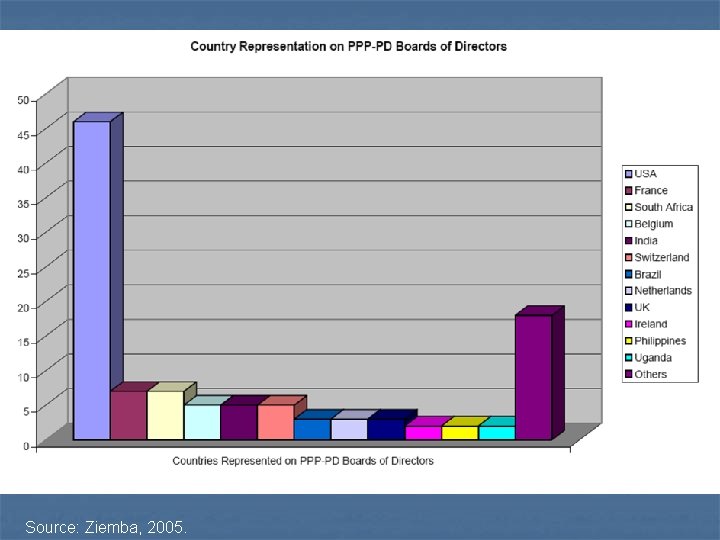
Source: Ziemba, 2005.

6. 3 Governance: Sustainability n n Scientific: need to invest in capacity of more countries to contribute Financial: need innovative financing model Political: need broader base of support How do PDPs fit into a rapidly changing institutional landscape?

6. 4 Governance: Accountability Accountable to: mechanism n Donors: future funding n Partner firms: future partners n Patients: reputation n Public (as taxpayers and potential beneficiaries): reputation Hybrid structure = unclear expectations of accountability Example: FDA Priority Review Vouchers n n n New neglected disease product gets tradable voucher for accelerated FDA review Worth up to ~300 million USD Who decides how to spend, how to invest, how to ensure public interest?

6. 5 Governance: Public goals and private partners Tension between public & private objectives: n Impacts governance (secrecy) n Impacts innovation (secrecy, patent applications, competitiveness, delays [moxifloxacin]) n Impacts affordability (price discrimination) n Impacts financial position: direct tradeoff between control over end-user access and PDP investment n Impacts public trust: public money to private sector n Private free-riding on PDP knowledge-generation n Unintended consequences of private incentives on public -private cooperation?

7. Conclusions Advances of PDPs over old pure public or private R&D: • Increased resources for R&D dedicated to diseases affecting world’s poorest • Improved emphasis on access & adaptedness • Increased transparency (compared to private) Key questions re: PDP model: • Financing: how to sustain? • Governance: Who sets the agenda? How is IP managed? Transparency? Public accountability? • Sustainable: how to achieve scientific, financial, political sustainability of global public goods provision? • How do we manage the tensions inherent in harnessing private actors for public ends? What expectations should we have?

Thank you Questions to: suerie_moon@hksphd. harvard. edu
- Slides: 37