Public Health Electronic Case Reporting e CR John

Public Health Electronic Case Reporting (e. CR) John W. Loonsk MD FACMI Chief Medical Informatics Officer, Consultant Association of Public Health Laboratories

Surveillance is a (the? ) core public health function 1. Monitor diseases and other conditions at the local, state, and national levels 2. Support routine PH investigation, outbreak investigation, and emergency response 3. Identify environmental health events and impacts 4. A broad spectrum of activities from: • suppressing disease spread in SARs and Ebola outbreaks • managing exposures to wildfire smoke or white powder reports (Anthrax) • determining the cause of foodborne outbreaks • Investigating new conditions or virulence

Why is surveillance so hard? • Differing state laws define what data are needed where and when • Many jurisdictions x many conditions x many clinical care sites = exponential interoperability problem • Disease program funding leads to historically siloed activities • Specific public health data authorities – only identifiable data for reportable conditions in state laws • Complex, changing case definitions • Dependency on clinical data that are complex, poorly structured, and until recently, not in electronic form at all • Little or no feedback (read as perceived value) to clinical care participants and provider burden

You may know these as surveillance • Electronic Laboratory Reporting • Successful for certain disease monitoring • Dependent on pathognomonic lab test for condition, limited lab result data, lab testing-based timing, limited ability to reach back to provider • Lab Information Systems (LIMS) were electronic = an opportunistic solution • Syndromic Surveillance • Focused on the patient’s, not the clinical staff’s, problem presentation • No interventions or follow-up possibilities on de-identified data • Hospital Admissions Discharge and Transfer (ADT) systems were electronic = an opportunistic solution
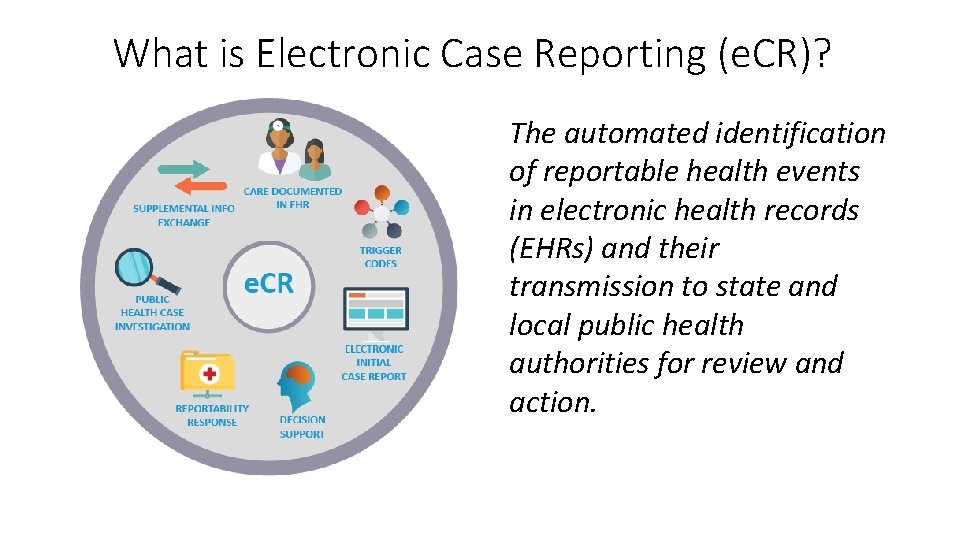
What is Electronic Case Reporting (e. CR)? The automated identification of reportable health events in electronic health records (EHRs) and their transmission to state and local public health authorities for review and action.
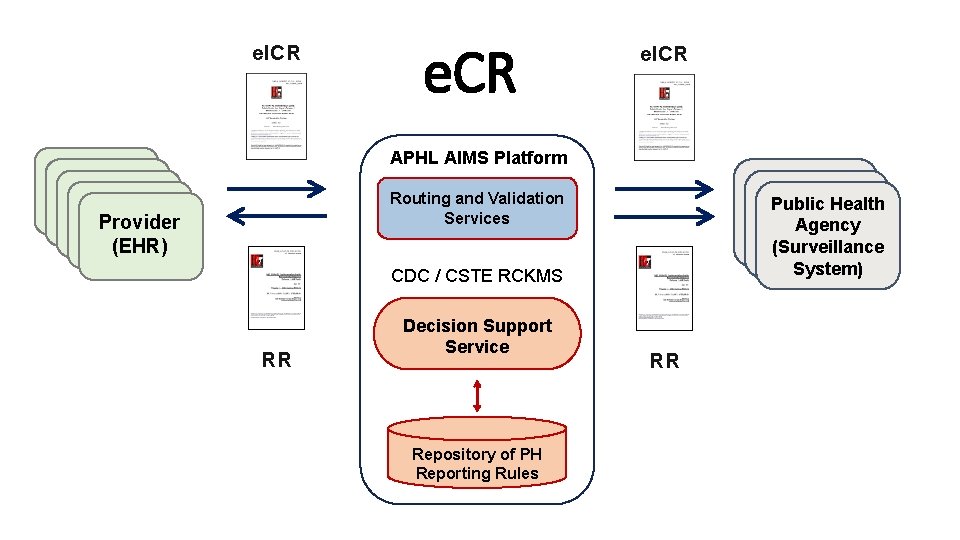
e. ICR APHL AIMS Platform Provider (EHR) Provider (EHR) Routing and Validation Services Public Health H Agency (Surveillance System) CDC / CSTE RCKMS RR Decision Support Service Repository of PH Reporting Rules RR

e. CR solution and outcomes • Supporting acronyms: • CDC, CSTE, APHL, ASTHO, NACCHO, RWJ, PHII, DB • Supporting EHRs: • EPIC, Cerner, Allscripts, and growing • Automates reporting – reduces provider burden • Hides jurisdictional variation from clinical care / EHR vendors • Creates single technical interface for EHRs (technical scaling) • Partnership with the e. Health Exchange to avoid additional Business Associate or data use agreements (policy scaling)

e. CR solution and outcomes • Bidirectional communications / feedback in EHR less than two clicks away • e. CR - a positive provider EHR outcome? !
- Slides: 8