PTT 156 Principles of Chemical Processes ENERGY BALANCE

PTT 156 Principles of Chemical Processes ENERGY BALANCE ON REACTIVE PROCESS Sriyana Abdullah Department of Chemical Engineering Technology University Malaysia Perlis

HEATS OF REACTION

HEATS OF REACTION Example of~Example~ Heat of Reaction

Types Heat OF of Reaction TYPE OFof HEATS REACTION

Theory of Heat of Reaction (Example) THEORY HEATS OF REACTION q. One O-O bond and two H-H bonds broken. System absorbs energy. Thus, Usystem and Hsystem increase from reactants to transition state. q 4 O-H bonds are formed. System releases energy. Thus, Usystem and Hsystem decrease from transition state to products.

Theory. HEATS of Heat. OF of Reaction THEORY REACTION q The change in enthalpy from reactants to products, is the heat of reaction. q For stoichiometric quantities of H 2 and O 2 reacting completely at T=25ºC and P =1 atm, q Negative value of heat of reaction denote; q More energy released by product bond formation than absorbed when reactant bonds break. The reaction is therefore exothermic.

Theory. HEATS of Heat. OF of Reaction THEORY REACTION • Thus the reaction can be written as: • The value of heat of reaction means: • If 5 mol/s H 2 is consumed in which the reactants and products are at 25°C, then the energy balance is;

Theory of Heat OF of Reaction THEORY HEATS REACTION

Properties of Heat of. PROPERTIES Reaction HEATS OF REACTION q q q Negative ΔĤr denoted exothermic reaction Positive ΔĤr denoted endothermic reaction ΔĤr (T, P) nearly independent of pressure at low and moderate pressures; Thus written as ΔĤr (T) The value of heats of reaction depends on how the stoichiometric equation is written (e. g. standard heat of the reaction for 2 A→ 2 B is twice of A→B) The value of heat of reaction depends on the states of aggregation (gas, liquid, solid) of the reactants and products.

INTERNAL ENERGY OF REACTION • If a reaction take place in a closed reactor at constant volume, the heat released/absorbed is determined by the change in internal energy between reactants and products, not enthalpy. • The internal energy of reaction, ΔÛr(T) is the difference Uproducts–Ureactants if stoichiometric quantities of reactants react completely at temperature T. • Suppose a reaction occurs by assuming ideal gas behaviour and neglecting specific volume where υi is the stoichiometric coefficient of species i, thus the internal energy of reaction is related to the heat of reaction (closed system) is given by; * If there are no gaseous reactant /products =

HEATS OF REACTION USING HESS LAW General statement of Hess’s Law: If the stoichiometric equation for reaction 1 can be obtained by algebraic operations (multiplication by constants, addition, and subtraction) on stoichiometric equations for reaction 2, 3, …, then the heat of reaction ΔĤr 1 can be obtained by performing the same operations on the heat of reactions ΔĤr 2, ΔĤr 3, … In the other words: Hess’s law states that if you can obtain a stoichiometric equation as a linear combination of the stoichiometric equations for other reactions, you can determine its heat of reaction by performing the same operations on the heats of the other reactions.

Example: Hess Law

FORMATION REACTION AND HEATS OF FORMATION Formation reaction: A reaction in which a compound is formed from its elemental constituents as they occur in nature [e. g. , O 2(g), and not O]. Standard heat of formation: Ø The enthalpy change associated with the formation of 1 mole of the compound at a reference pressure and temperature (25˚C, 1 atm). Ø Standard heats of formation of many species are given in Table B. 1. For example, liquid benzene: ØThe standard heat of formation of an elemental species [C(s), H 2(g), O 2(g), . . . ] is zero.

HEAT OF FORMATION

HEAT OF REACTION DETERMINATION FROM HEAT OF FORMATION

HEATS OF COMBUSTION

Determination of Heat of Reaction from Heat of Combustion

Determination of Reaction from HEATS of OFHeat COMBUSTION Heat of Combustion-Example

HEATS OF COMBUSTION Exercise 5 ~Example 9. 4 -1~ Calculate the standard heat of reaction for the dehydrogenation of ethane:

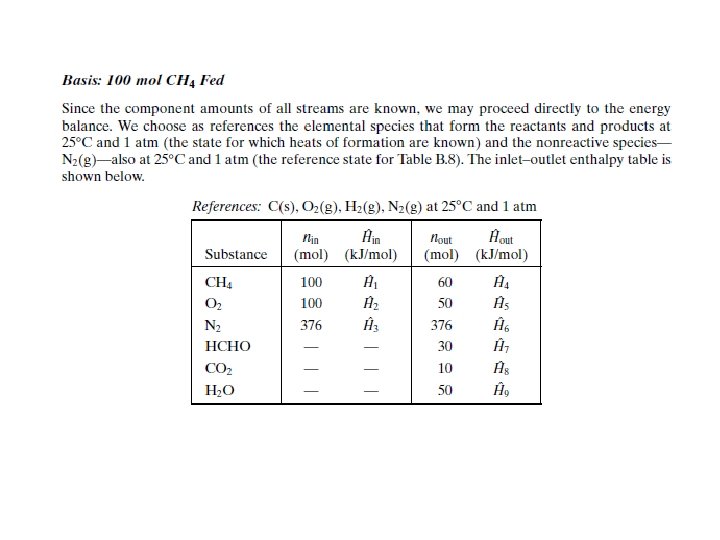
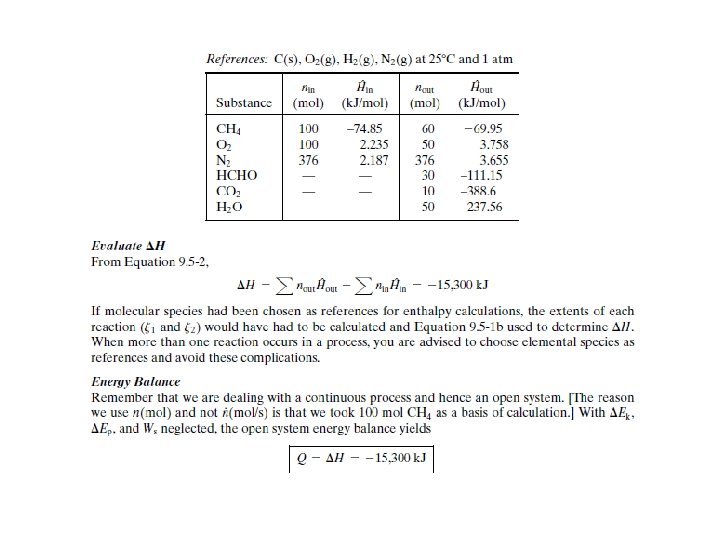
General Energy Balance Procedures on Reactive Processes To perform energy balance calculation on a reactive system, you must include the following aspects: 1. 2. 3. 4. 5. 6. Draw and label flowchart Use material balances and phase equilibrium relationship to determine the amount of stream component and flow rates Choose reference states for specific enthalpy/internal energy Prepare and fill the inlet-outlet enthalpy table Calculate

ENERGY BALANCE ON REACTIVE PROCESS Two methods are commonly used to choose reference states for enthalpy calculations: 1. Heat of Reaction Method - Generally preferable when there is a single reaction for which is known. 2. Heat of Formation Method - Generally preferable for multiple reactions and single reactions for which is not readily available.


Example: Heat of Reaction Method


Example : Heat of Formation Method


HEATS OF REACTION ~Exercise 1~ (a) The standard heat of reaction Calculate of n-butane vapor is; (k. J/s) if 2400 mol/s CO 2 is produced at 25ºC (b) What is the standard heat of reaction , Based on (a) Calculate for : if 2400 mol/s CO 2 is produced at 25ºC (c) The heats of vaporization n-butane and water at 25ºC are 19. 2 k. J/mol and 44. 0 k. J/mol, respectively. What is the standard heat of the reaction for; Based on (a) Calculate if 2400 mol/s CO 2 is produced at 25ºC

INTERNAL ENERGY OF 2 REACTION Exercise ~Exercise 2~ Based on the reaction below, calculate the internal energy of reaction at 25 °C (R= 8. 314 J/mol. K)

Exercise. USING 3 HESS LAW HEATS OF REACTION ~Exercise 3~ • Consider this reaction: By using Hess Law, determine the following reaction: for the

HEAT OF FORMATION ~Exercise 4~ Determine the standard heat of reaction for the combustion of liquid n-pentane, assuming H 2 O(l) is a combustion product at standard state.

THANK YOU FOR YOUR ATTENTION… ~DO YOUR BEST IN FINAL EXAMINATION SOON~
- Slides: 33