PTRT 1309 CORROSION BASICS Introduction to Corrosion Terminology

PTRT 1309 CORROSION BASICS Introduction to Corrosion Terminology and Basic Principles Oil and Gas Technology Program

Introductions/ Qualifications • Paul Barker • • • NACE certified Corrosion Technologist Senior Technologist Pending AAS in Corrosion Technology 1992 8 years with XTO as pipeline integrity foreman 20 years experience in corrosion control ranging from production, pipeline to process Oil and Gas Technology Program

Corrosion Basics • Definition – – – Deterioration of material by reaction to its environment. The corrosion occurs because of the natural tendency for most metals to return to their natural state; e. g. , iron in the presence of moist air will revert to its natural state, iron oxide. Metals can be corroded by the direct reaction of the metal to a chemical; e. g. , zinc will react with dilute sulfuric acid, and magnesium will react with alcohols Ref: NASA - CORROSION CONTROL AND TREATMENT MANUAL - TM-584 C REVISION C Oil and Gas Technology Program

Corrosion Basics • – Steel is an alloyed refined material This process introduces other materials are added to the Iron to make it a usable material • – – – • Carbon, Manganese, Nickel, Chromium etc. The process uses heat (energy) to blend the material The corrosion process is the material reverting to its natural state and releasing the energy added to the material THE SOURCE OF VOLTAGE IN THE CORROSION PROCESS COMES FROM THE different amount of ENERGY STORED IN THE METAL DURING REFINING Corrosion is an ELECTROCHEMICAL reaction. It is both an electrical and a chemical reaction occurring simultaneously. Oil and Gas Technology Program
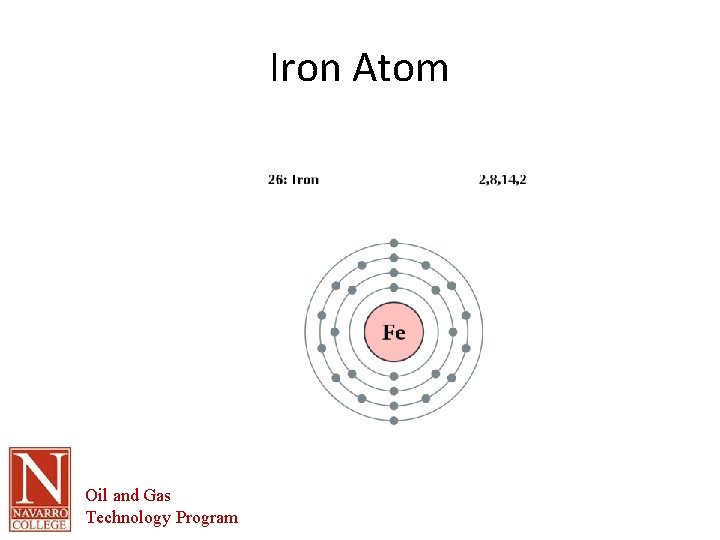
Iron Atom Oil and Gas Technology Program

Basic Electrical Theory Electrical current flow is defined as the movement of a charged particle (Electrons) in a specified direction. These charged particles can move through varying materials – • • Metallic Conductors- electrons flow through a conductor (metals). There is no detrimental effect on the conductor as the electrons are not lost Electrons go from one atom of the metal to the other. Electrolytic- Electron move through the liquid via Ions (charged particles). In our corrosion cell metal is lost where current is discharged into an electrolyte because the loss of an electron breaks the bonds that hold the metal together. Oil and Gas Technology Program

Basic Electrical Theory Units of Measurement Voltage- A unit of measurement which measures the difference in potential. Electrical pressure or differential pressure (PSI). – • Voltage can be measured without breaking the circuit (parallel). Oil and Gas Technology Program

Basic Electrical Theory Units of Measurement Ampere (amps) - A unit of measurement of electrical flow rate…. . How many electrons go through the circuit over a known time period? Just like speed is measured in miles per hours. Correlation- MMCFD, gallons/ minute, etc. – • • • The Circuit must be broken in order to measure amperage (series measurement) Amperage is usually measured indirectly through application of ohms law. Similar to natural gas flow measurement. Oil and Gas Technology Program

Basic Electrical Theory Units of Measurement Ohms- a unit used to quantify electrical resistance to flow – • • In electricity it’s the size of conductor or size of load Oil and gas correlation – – pipe size chokes Oil and Gas Technology Program

Basic Electrical Theory Ohm’s Law • Ohms law- a mathematical relationship between volts ohms and amperes where 2 are known the unknown can be calculated. This is extremely useful for current flow measurement since the circuit must be broken in order to measure current flow. – – V-IR I=V/R R=V/I Amperage is measured using a measured voltage drop across a known resistance. Example 1 volt of drop across a 2 Ohm resistor =. 5 amps flow. Oil and Gas Technology Program

Basic Electrical Theory – – Polarity- defines a charge as positive or negative. Like charges repel opposing charges attract Direction of flow- The direction the current flow in an electrical circuit Oil and Gas Technology Program

Basic Electrical Theory Alternating Current – – – Alternating Current (AC) AC is the type of electrical power that is commercially available When discussing AC circuits flow direction is not an issue as the electrons go back an fourth along the conductor The primary reason AC is used so widely is the ease of electrical transmission The electrons do not have to “move” long distances therefore reducing loss. Oil and Gas Technology Program

Basic Electrical Theory Direct Current (DC) – – – Direct Current (DC) - Current flows in one direction the electrons have to move through the circuit from start to finish This causes power loss over long distances rendering it inefficient for long distance transmission When we discuss the basic corrosion cell it is DC. Oil and Gas Technology Program

Corrosion Cell - Ideal Oil and Gas Technology Program

The Corrosion Cell • Corrosion cell- the corrosion cell has 4 parts. – – • • • Anode Cathode Electrolyte Metallic Path All 4 parts must be present in order for the material to corrode. Removal of one part of the cell renders it inactive All corrosion mitigation methods are based on this premise. Oil and Gas Technology Program

The Corrosion Cell • Anode – The area of the corrosion cell where metal loss occurs – Where oxidation occurs. – Oxidation is the loss of electrons – The loss of electrons causes the material to break down or corrode. Oil and Gas Technology Program

The Corrosion Cell • Cathode – The area of the corrosion cell where NO metal loss occurs – Where Reduction occurs. – Reduction is the gain of electrons – This gain in electrons creates protective mineral deposits due to the chemical reaction occurring at the cathodic surface Oil and Gas Technology Program

The Corrosion Cell Electrolyte- An electrically conductive solution • • • – – – – • For a solution to e conductive it must contain ions. The more Ions the more conductive or corrosive the solution. Ions- charged particles that can transport electrical current through the solution Examples in Oil and Gas production Produced water Sea water Fresh water Soil Electrolyte- in most scenarios this is the part of the corrosion cell that is utilized for control of corrosion. This is done through barrier coatings of chemically altering the electrolyte. Water is the primary electrolyte. All electrolytes are electrically conductive. Pure water is not conductive, but the addition of minerals makes it conductive. Oils are not conductive by their nature. Oil and Gas Technology Program

The Corrosion Cell • – Some factors that effect the corosivity of an electrolyte • • • – • • • Water- pure water is a POOR conductor. Minerals must be present to support current flow Soils Salt Content- produced fluids Ionic concentration p. H Dissolved Oxygen Temperature Salt content Moisture content Soil make up relating to electrical resistivity – • • • – • • • Sand has a high electrical resistance while clays are low. p. H Oxygen concentration Temperature Atmospheric Corrosion (most to least severe) Industrial and marine or seacoast Industrial and high humidity Marine or seacoast High humidity only Inland, industrial low humidity Rural, non-industrial, low humidity Oil and Gas Technology Program

The Corrosion Cell Metallic path- a metallic connection between the anode and cathode In the oil and gas industry this part of the cell is inherent in the structure to be protected. • • – – Anodic and cathodic areas are part of the pipe Therefore the anodes and cathodes cannot be electrically separated. Oil and Gas Technology Program

The Corrosion Cell Other factors that determine the rate of the corrosion reaction • Anode to cathode proportion – • • – – Large anode small cathode- slows reaction Small anode large cathode- speeds the reaction Dissimilar metals connected to one another Difference in concentration of corrosive agents in the electrolyte Oil and Gas Technology Program

Corrosion Cell - Ideal Oil and Gas Technology Program
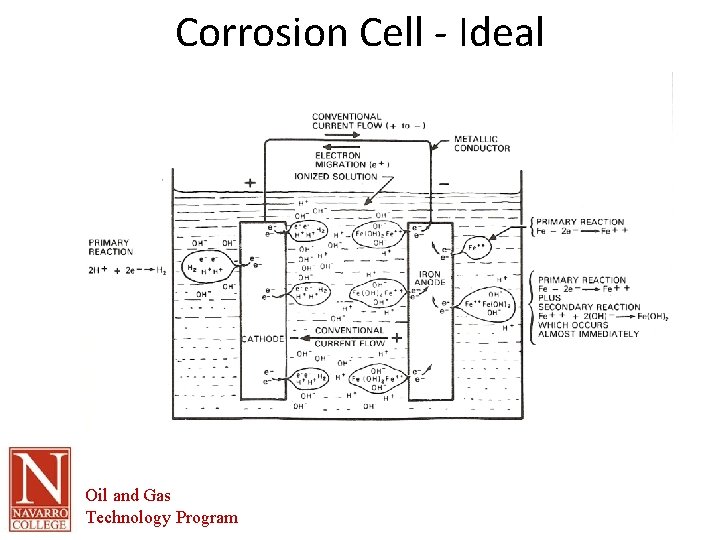
Corrosion Cell - Ideal Oil and Gas Technology Program
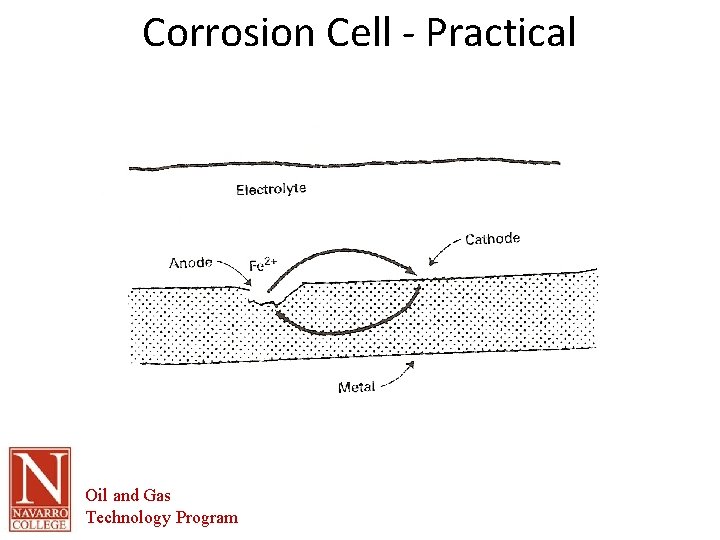
Corrosion Cell - Practical Oil and Gas Technology Program

Justification for Corrosion Mitigation Asset life expectancy- design life – • • • will the asset last long enough to justify the cost of remediation Cost-Results of the study show that the total annual estimated direct cost of corrosion in the U. S. is a staggering $276 billion— approximately 3. 1% of the nation’s Gross Domestic Product (GDP). Safety and environmental concerns – – – Affected public safety Employee safety Environmental contamination Oil and Gas Technology Program

Justification for Corrosion Mitigation – • Regulatory consideration- This is an industry that is heavily regulated by both state and federal agencies. PHMSA – • EPA – • Regulates corrosion control methods and standards for pipelines based their proximity to population and/ or the materials transported Regulates environmental standards. Leaks caused by corrosion get their attention State agencies (Texas Railroad Commission, Oklahoma Corporation Commission etc. ) » » Oil and Gas Technology Program Regulate oil and gas production Regulate intrastate pipelines under the direction of PHMSA

Oil and Gas Equipment at Risk – • • – – • • • Well bore tubing casing production equipment Pipelines- Each category within the pipeline industry has unique inherent risks to integrity Gathering- Internal corrosion Transmission- External Corrosion Distribution- Third party damage/ External corrosion Process Equipment Vessels Processing plant piping Storage tanks Oil and Gas Technology Program

Oil and Gas Equipment at Risk Corrosion prevention is of special importance to the oil and gas industry – • • • Flammable materials Pressurized equipment- the unintended release of pressure form this equipment is usually violent and dangerous Environmentally sensitive materials High cost of materials and equipment Value of product lost. Safety of employees and effected public Oil and Gas Technology Program

External Corrosion that occurs from the soil or atmospheric side of the equipment Mitigation Methods – – • Cathodic protection – – – • requires the surface to be immersed in a continuous electrolyte Does not stop corrosion dictates the anodic area Protects the pipe at coating holidays Protective coatings – – – THERE IS NO PERFECT COATING Form a protective barrier between the surface and the electrolyte Some coatings (IE galvanizing) utilize anodic material in the coating to create a protective current. Oil and Gas Technology Program

FBE Coating Failure Oil and Gas Technology Program
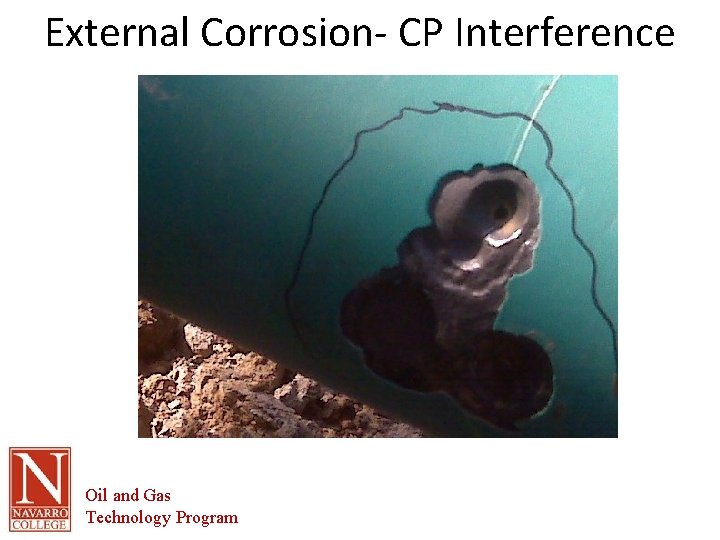
External Corrosion- CP Interference Oil and Gas Technology Program

Internal Corrosion – • Internal Corrosion- corrosion that occurs inside of the equipment as a result of the corrrosivity of the material held in the equipment. Localized (pitting) - corrosion that is concentrated in a small area. These mechanisms typically go through the wall of the vessel, pipe, or tank quickly. – – • Acid gasses » H 2 S » CO 2 MIC- Microbial induced corrosion » SRB » APB General- metal is lost over the entire surface of the material. This can be combated based on the life of the asset and thickness of the material. Oil and Gas Technology Program

Oil and Gas Technology Program

CO 2 Corrosion Oil and Gas Technology Program

H 2 S Corrosion Oil and Gas Technology Program

SRB- Tank Bottom Oil and Gas Technology Program

Internal Corrosion Mitigation • – Mitigation Methods • • • – Corrosion Inhibiting chemicals Water soluble- dissolve in the corrosive media reducing the corrrosivity of the water (electrolyte Oil soluble- coat the inside of the pipe forming a barrier Biocide- kills corrosive bacteria Design- This can cover a broad range of topics Material selection- Use a material that is not attacked by its environment Eliminate stagnant area Use thicker materials- only effect when dealing with general forms of corrosion Design for easy replacement- Adding bypasses, isolation valves etc. Pigging Used to displace free liquids from the pipeline Chemical application Internal coatings- form a barrier between surface and electrolyte Typically ineffective Oil and Gas Technology Program

10” Brush Pig Oil and Gas Technology Program

10” Poly Pig Oil and Gas Technology Program

Review • • • Corrosion costs the US $276, 000 each year Most of that corrosion is preventable Corrosion in the Oil and Gas Industry is both a financial issue as a safety issue Corrosion cell has 4 intrinsic parts Localized corrosion does not take long to render the asset “useless” Corrosion prevention in the oil and gas industry is required by law No water no corrosion Corrosion is an electrochemical reaction Shale plays and other resource development will bring corrosion issue to the forefront due to their prevalence and location All facets of this industry are effected by corrosion. Oil and Gas Technology Program
- Slides: 40