Psychrometrics Lab not psychometrics Dr V Menta Assistant

Psychrometrics Lab (not psychometrics) Dr. V Menta Assistant Professor Department of Mechanical and Industrial Engineering University of Minnesota Duluth

Topics • • Analysis of Gas Mixtures Dalton’s Rule of Partial Pressures Properties of Ideal Gas Mixtures of Ideal Gases and Condensable Vapor – Psychrometrics • Common Processes – Air/Water Vapor Mixtures

Learning Outcomes By the end of this class, students will be able to: • Explain psychrometric parameters • Identify the relationships among parameters • Analyze the dry air/water vapor composition • Calculate psychrometric parameters using mathematical equations • Calculate psychrometric parameters using psychrometric charts

What is Psychrometrics? • • • The science involving thermodynamics properties of moist air Dry air vs. moist air Frequently used in weather forecasts Applications – HVAC systems, Air-Conditioning devices, Cooling Towers, Evaporative coolers, Industrial processes requiring close control of the vapor content in air, animal housing, plant sytems (Greenhouses, growth chambers etc. )

Psychrometrics Assumptions: a. Binary System – dry air + water vapor – Composition of dry air is nearly constant, therefore we treat it as a single gas b. IDEAL GAS mixture – i. e. we have a low enough pressure so that non-ideal gas effects can be neglected Note: – The thermodynamic state of the vapor in an air-water vapor mixture is fixed by its partial pressure and the temperature.

Hilton Air Conditioning Lab Unit

10 Psychrometric Parameters 1. Total Pressure 2. Dry Bulb Temperature 3. Wet Bulb Temperature 4. Specific Humidity 5. Relative Humidity 6. Specific Volume 7. Specific Enthalpy 8. Water Vapor Pressure (Partial) 9. Dry Air Pressure (Partial) 10. Dew Point Temp.


Analysis of Gas Mixtures • Consider a mixture of N species (gases). We can choose to analyze this mixture on a mass or molar basis. 1. Gravimetric Analysis – Analysis of a gas mixture on the basis of mass (or weight). Let mi be the mass of the ith species, The total mass, m is given by

Analysis of Gas Mixtures Mass fraction, yi is defined as: 2. Molar Analysis – Analysis of a gas mixture on a molar basis. Let ni be the number of moles of the ith species, The total number of moles, n is given by

Analysis of Gas Mixtures Mole fraction, xi is defined as: 3. Relations Between Gravimetric and Molal Quantities Let Mi be the molecular weight of the ith species,

Analysis of Gas Mixtures The average molecular weight of the gas mixture, M is given by: The mass fraction is related to the mole fraction via the following:

Analysis of Gas Mixtures 4. Example: Approximating air as a mixture having a mole fraction of 0. 79 Nitrogen and 0. 21 Oxygen, determine; a) the average molecular weight of air and b) the gravimetric analysis of air. a) M is given by: NOTE: considering all other species Mair = 28. 97 lbm/lbmol b) Gravimetric analysis:

Analysis of Gas Mixtures This same approximation for air may be expressed as:

Dalton’s Rule of Partial Pressures The total pressure of a mixture of IDEAL GASES is equal to the sum of the partial pressures where the partial pressures are determined at the volume and temperature of the mixture. (1) (2) (3)

Dalton’s Rule of Partial Pressures (4) Dividing equation (2) by equation (3) and rearranging yields the following: (5) Introducing the specific gas constant of the ith species Ri, which is given by: (6) We can show that the specific gas constant of the mixture R, is:

Dalton’s Rule of Partial Pressures • Dalton’s Rule of Partial Pressures is valid at relatively low pressures. It has been verified experimentally and is obtainable from statistical thermodynamics on the basis that each component is not influenced by the pressure of the other components.

Properties of Ideal Gas Mixtures • IDEAL GAS mixtures 1. Internal Energy a. Molar Basis

Properties of Ideal Gas Mixtures b. Mass Basis

Properties of Ideal Gas Mixtures 2. Similar equations for other properties: Molal Basis • Mass Basis These relations are exact for ideal gas mixtures and approximate for real gas mixtures.
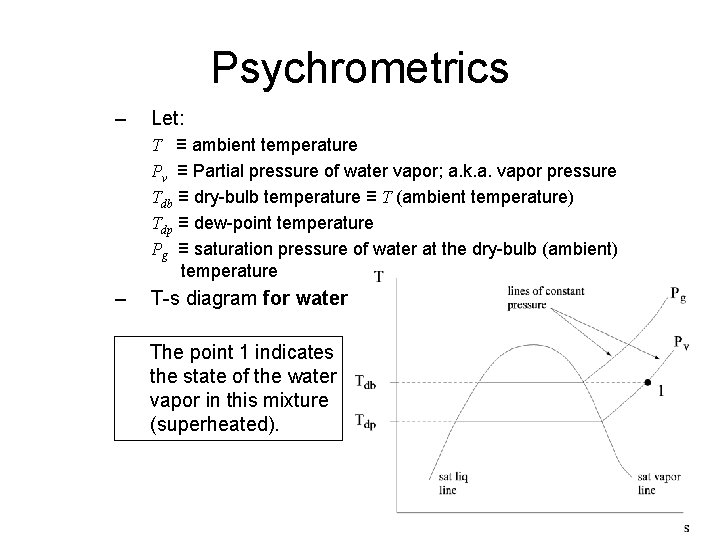
Psychrometrics – Let: T ≡ ambient temperature Pv ≡ Partial pressure of water vapor; a. k. a. vapor pressure Tdb ≡ dry-bulb temperature ≡ T (ambient temperature) Tdp ≡ dew-point temperature Pg ≡ saturation pressure of water at the dry-bulb (ambient) temperature – T-s diagram for water The point 1 indicates the state of the water vapor in this mixture (superheated).

Psychrometrics a. Unsaturated Air – a mixture of dry air and superheated water vapor b. Saturated Air – a mixture of dry air and saturated water vapor c. Dew-Point Temperature (Tdp) – Mixture temperature at which water vapor starts to condense out of the mixture: i. e. Tdp = Tsat @ Pv d. Dry-Bulb Temperature (Tdb) – Equilibrium temperature of the mixture indicated by an ordinary thermometer e. Wet-Bulb Temperature (Twb) – For unsaturated air, it is always less than the dry-bulb and greater than the dewpoint temperature. For saturated air, Twb = Tdp. Note: Twb is a good approximation to the adiabatic saturation temperature at atmospheric conditions. Psychrometer measures the dry-bulb and wet-bulb temperature

Psychrometrics f. Humidity Ratio (ω) – a. k. a. Specific Humidity assuming both dry air and water vapor are IDEAL GASES

Psychrometrics

Psychrometrics g. Relative Humidity ( ) – indicates the degree of saturation of the mixture

Psychrometrics 3. Example: Moist air at 100 F, 1. 0 atm and a relative humidity of 60% is cooled at a constant mixture pressure. Its final temperature is 70 F. Determine: a. The final relative humidity b. The change in the specific humidity a. From Steam Tables

Psychrometrics From Steam Tables Note that this pressure is below the Pv 1 value. Therefore, water vapor must have condensed out and we are at saturated conditions. b. Change in specific humidity

Psychrometrics 4. Enthalpy of Air/Water Vapor Mixture a. Enthalpy of Air

Psychrometrics b. Enthalpy of Water Vapor hv = f(Pv, Tdb) in general hv ≈ hg for the range of water vapor partial pressures in atmospheric air Approximation: c. Enthalpy of Mixture English Units: SI Units:

Psychrometrics 5. Carrier’s Equation – Approximation for vapor pressure of water vapor in air English Units: SI Units:

Psychrometrics 6. Measurement/Calculation Steps – – – Measure P (barometer) and TDB and TWB Use Carrier’s Equation to get Pv Calculate , , and h using (for SI units)

Reading Psychrometric Chart
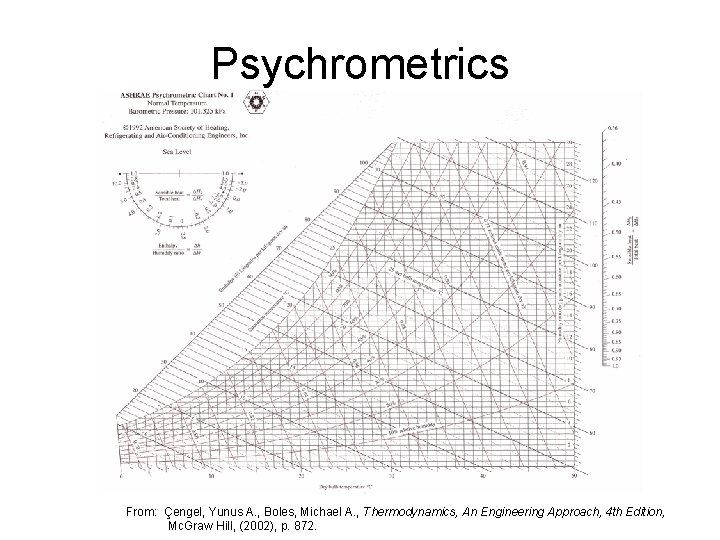
Psychrometrics From: Çengel, Yunus A. , Boles, Michael A. , Thermodynamics, An Engineering Approach, 4 th Edition, Mc. Graw Hill, (2002), p. 872.
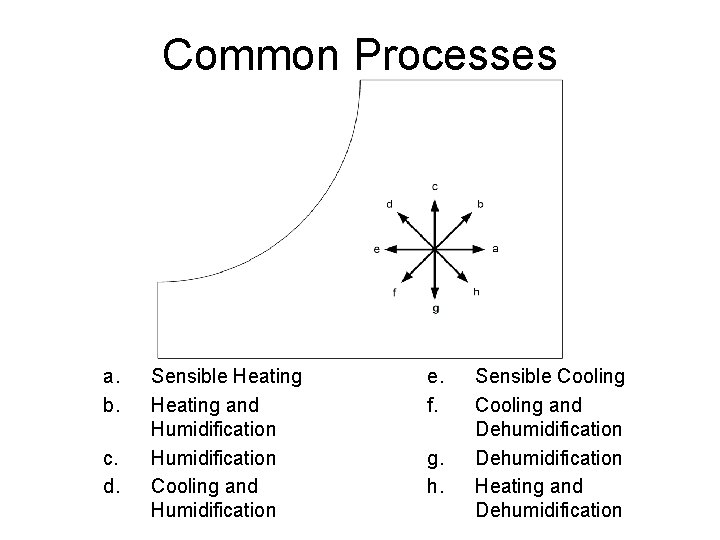
Common Processes a. b. c. d. Sensible Heating and Humidification Cooling and Humidification e. f. g. h. Sensible Cooling and Dehumidification Heating and Dehumidification


What is Moist Air? • A binary mixture of dry air and water vapor • Each mixture component behave as ideal gases at the states under present consideration
- Slides: 37