PROVE IT TIMI 22 Study Design Doubleblind randomized

PROVE IT (TIMI 22): Study Design Double-blind, randomized trial in 4, 162 patients with Acute Coronary Syndrome <10 days and Total Cholesterol < 240 mg/d. L ASA + Standard Medical Therapy Pravastatin 40 mg qhs Gatifloxacin * Placebo Atorvastatin 80 mg qhs Gatifloxacin * Placebo Duration: Mean 2 year follow-up (925 events) Primary Endpoint: Death, MI, Stroke, UA requiring hosp. , or revascularization (> 30 days after randomization) Cannon et al. Am J Card 2002; 89: 860– 861. * Gatifloxacin 400 mg qd X 10 days/month
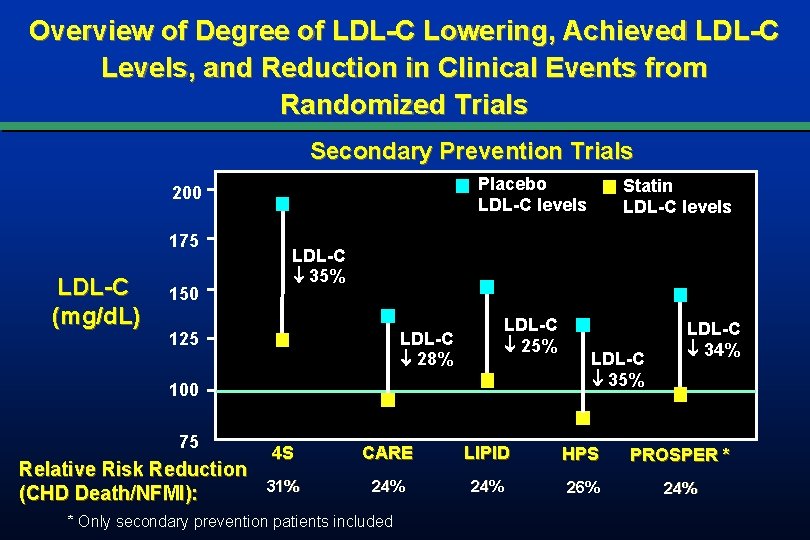
Overview of Degree of LDL-C Lowering, Achieved LDL-C Levels, and Reduction in Clinical Events from Randomized Trials Secondary Prevention Trials Placebo LDL-C levels 200 175 LDL-C (mg/d. L) 150 LDL-C 35% LDL-C 28% 125 100 75 Relative Risk Reduction (CHD Death/NFMI): Statin LDL-C levels LDL-C 35% LDL-C 34% 4 S CARE LIPID HPS PROSPER * 31% 24% 26% 24% * Only secondary prevention patients included
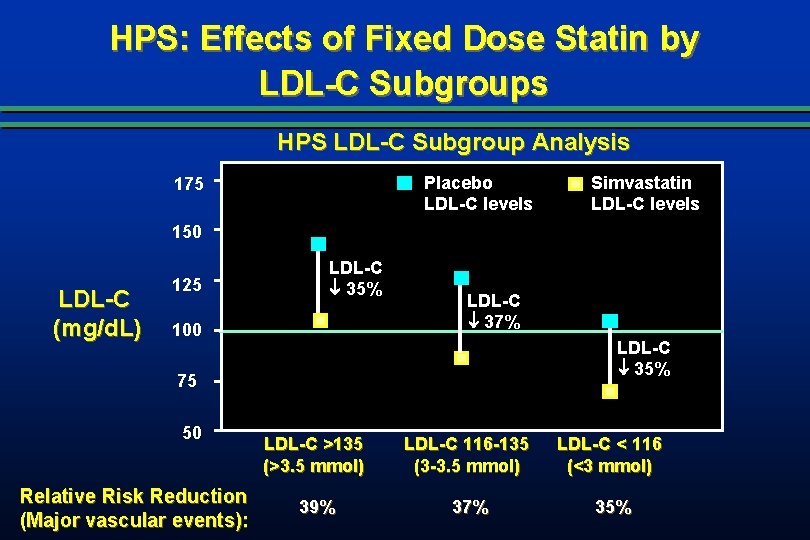
HPS: Effects of Fixed Dose Statin by LDL-C Subgroups HPS LDL-C Subgroup Analysis Placebo LDL-C levels 175 Simvastatin LDL-C levels 150 LDL-C (mg/d. L) 125 LDL-C 35% 100 LDL-C 37% LDL-C 35% 75 50 Relative Risk Reduction (Major vascular events): LDL-C >135 (>3. 5 mmol) LDL-C 116 -135 (3 -3. 5 mmol) LDL-C < 116 (<3 mmol) 39% 37% 35%
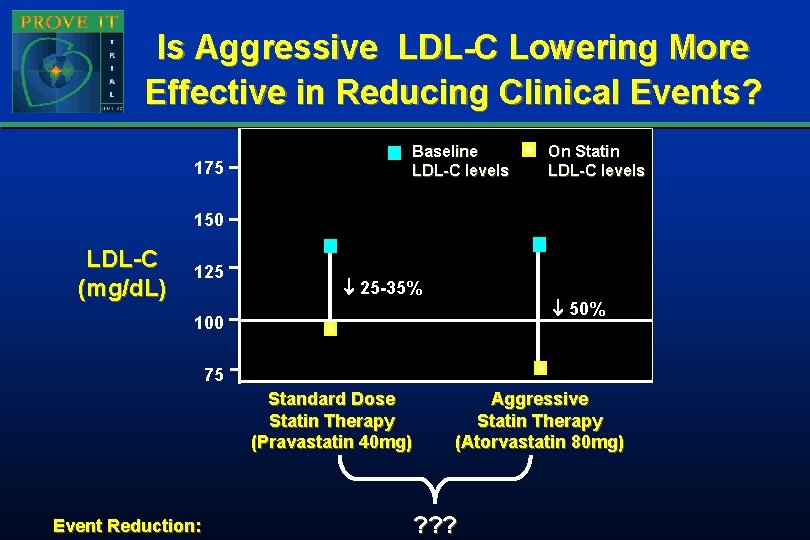
Is Aggressive LDL-C Lowering More Effective in Reducing Clinical Events? 175 Baseline LDL-C levels On Statin LDL-C levels 150 LDL-C (mg/d. L) 125 25 -35% 50% 100 75 Standard Dose Statin Therapy (Pravastatin 40 mg) Event Reduction: Aggressive Statin Therapy (Atorvastatin 80 mg) ? ? ?

PROVE IT: PRavastatin Or ator. Vastatin Evaluation and Infection Therapy (TIMI 22) Primary Objectives: l To determine if there is clinical equivalence between pravastatin and atorvastatin in reducing major cardiovascular events in patients with ACS AND l To determine if the antimicrobial agent gatifloxacin is superior to placebo in reducing major cardiovascular events in patients with ACS Cannon et al. Am J Card 2002; 89: 860– 1.

Primary Statin Non-inferiority Comparison: PROVE-IT is designed to determine whether the 2 year CV event rate of pravastatin is ‘clinically equivalent’ to atorvastatin – ‘Clinical equivalence’ declared if the upper 95% CI of relative risk of event rates at 2 years is < 1. 17 – Using Cox proportional Hazard model, hazard ratio (over the entire period of follow-up) of 1. 197 leads to a relative risk of 1. 17 for events at 2 years. – Assuming 22% event rate in atorvastatin arm, the greatest absolute difference between pravastatin and atorvastatin that would meet the pre-spcified definition would be 1. 2% (eg. atorva 22% vs. prava 23. 2%). Cannon et al. Am J Card 2002; 89: 860 -861; Crit Path Cardio 2003; 2: 150 -7.

Possible PROVE-IT Outcomes Secondary Analysis No clinical equivalence (& inferior) Uncertain Clinical equivalence (& superior) Pravastatin Better Atorvastatin Better 1. 0 1. 17 Relative Risk Upper 95% confidence limit of the relative risk between pravastatin and atorvastatin can be no worse than 1. 17 at 2 years (1. 198 hazard ratio) to demonstrate equivalence
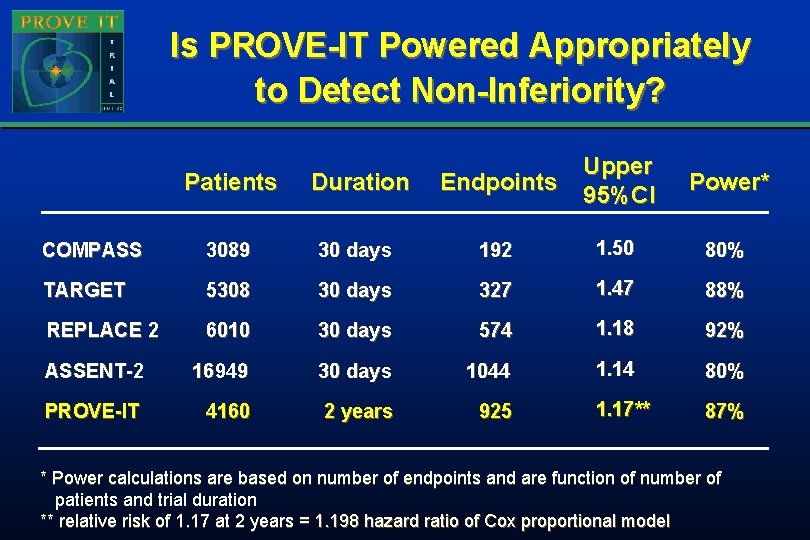
Is PROVE-IT Powered Appropriately to Detect Non-Inferiority? Patients Duration Endpoints Upper 95%CI COMPASS 3089 30 days 192 1. 50 80% TARGET 5308 30 days 327 1. 47 88% REPLACE 2 6010 30 days 574 1. 18 92% ASSENT-2 16949 30 days 1044 1. 14 80% PROVE-IT 4160 2 years 925 1. 17** 87% Power* * Power calculations are based on number of endpoints and are function of number of patients and trial duration ** relative risk of 1. 17 at 2 years = 1. 198 hazard ratio of Cox proportional model

PROVE-IT (TIMI 22): What Will it Tell Us? PROVE-IT designed to evaluate ACS patients and determine the 2 year impact of: l l l l Different degrees of LDL-C lowering Different statins (pravastatin vs atorvastatin) on safety Continual pulsed antibiotic therapy in patients following an acute coronary syndrome (ACS) will directly compare efficacy and safety of two statins strictly defines clinical equivalence follows 4162 patients for 2 years is endpoint driven is adequately powered (>87%)

Equivalence Trials More common in current era of multiple effective therapies l Used to test hypothesis of equivalence between 2 drugs or devices l Must have pre-defined criteria for “non-inferiority”, based on confidence intervals, to ensure adequate power l Definition of equivalence limit is 1) preserves benefit over placebo, or 2) is clinically based difference l

CME Evaluation form Please go to either: www. clincaltrialresults. org Or directly to: http: //www. medmeetings. org/webcasts/eval_091903_CB. php To complete the CME evaluation form and to receive your CME credit.
- Slides: 11