Protozoa Flagellates Giardia lamblia Dientamoeba fragilis Chilomastix mesnili

Protozoa
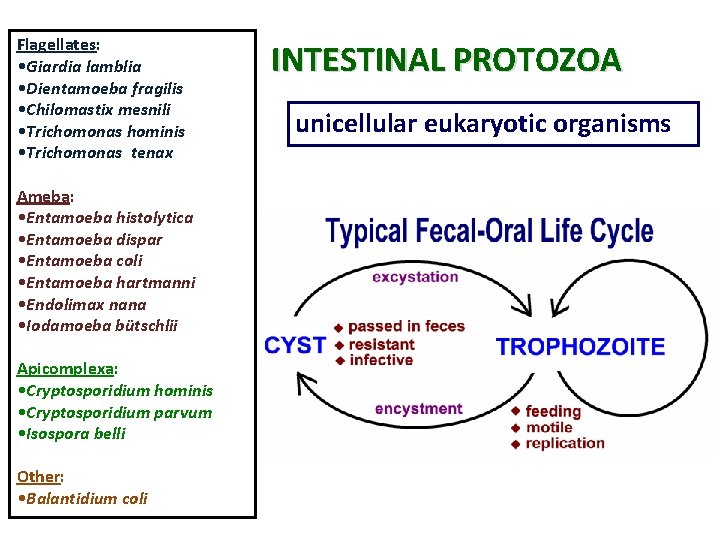
Flagellates: • Giardia lamblia • Dientamoeba fragilis • Chilomastix mesnili • Trichomonas hominis • Trichomonas tenax Ameba: • Entamoeba histolytica • Entamoeba dispar • Entamoeba coli • Entamoeba hartmanni • Endolimax nana • Iodamoeba bütschlii Apicomplexa: • Cryptosporidium hominis • Cryptosporidium parvum • Isospora belli Other: • Balantidium coli INTESTINAL PROTOZOA unicellular eukaryotic organisms

Entamoeba Histolytica (disease: amoebiasis or amoebic dysentery)
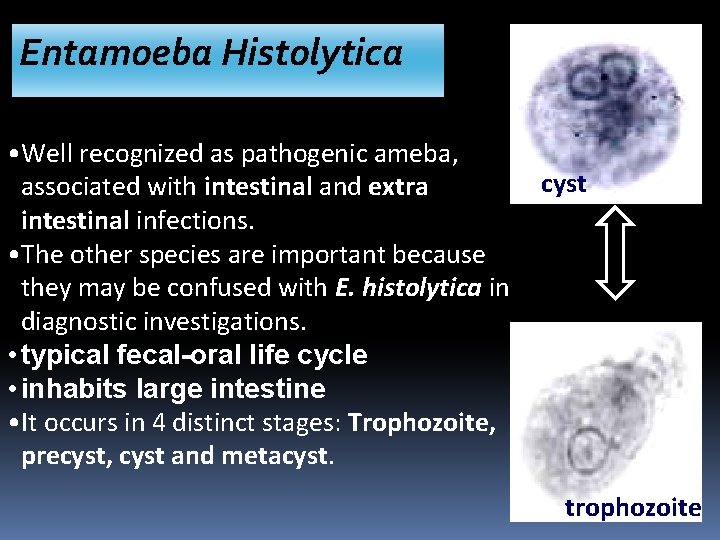
Entamoeba Histolytica • Well recognized as pathogenic ameba, associated with intestinal and extra intestinal infections. • The other species are important because they may be confused with E. histolytica in diagnostic investigations. • typical fecal-oral life cycle • inhabits large intestine • It occurs in 4 distinct stages: Trophozoite, precyst, cyst and metacyst. cyst trophozoite
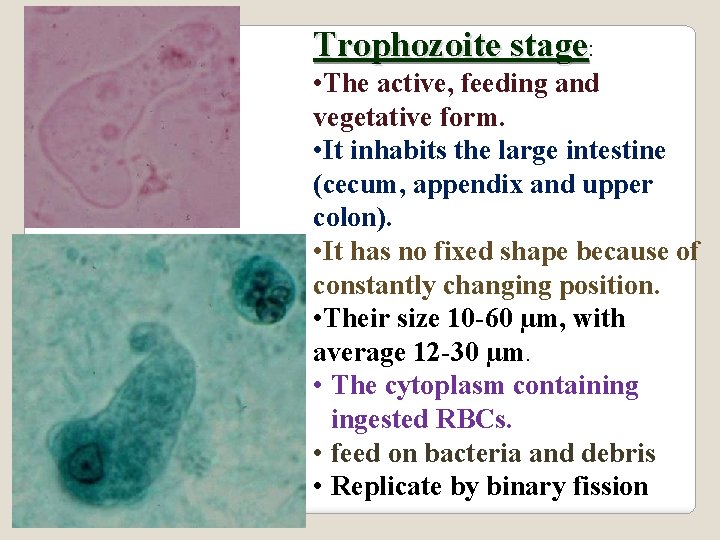
Trophozoite stage: • The active, feeding and vegetative form. • It inhabits the large intestine (cecum, appendix and upper colon). • It has no fixed shape because of constantly changing position. • Their size 10 -60 µm, with average 12 -30 µm. • The cytoplasm containing ingested RBCs. • feed on bacteria and debris • Replicate by binary fission
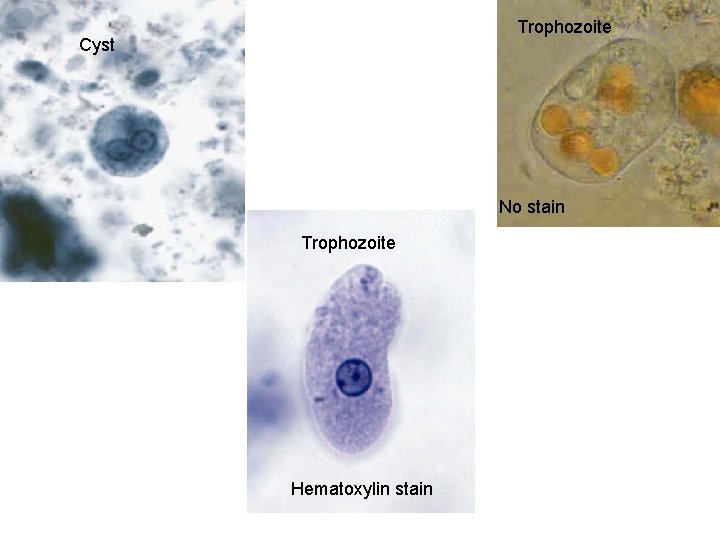
Trophozoite Cyst No stain Trophozoite Hematoxylin stain

Precyst stage: • Rounded or ovoid. • Sluggish movement • Blunt pseudopodium. • No RBC. • 10 -12 µm.
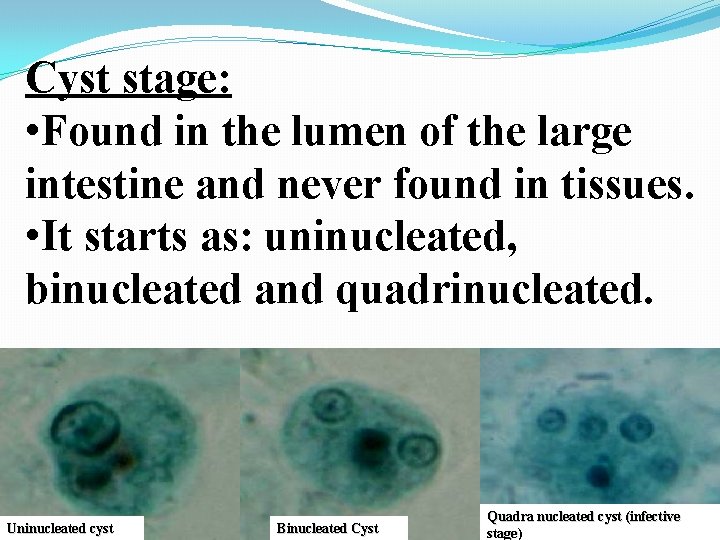
Cyst stage: • Found in the lumen of the large intestine and never found in tissues. • It starts as: uninucleated, binucleated and quadrinucleated. Uninucleated cyst Binucleated Cyst Quadra nucleated cyst (infective stage)
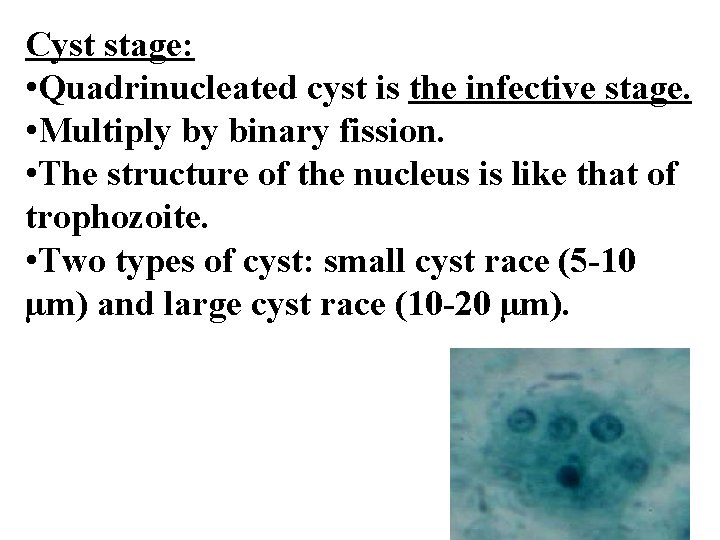
Cyst stage: • Quadrinucleated cyst is the infective stage. • Multiply by binary fission. • The structure of the nucleus is like that of trophozoite. • Two types of cyst: small cyst race (5 -10 µm) and large cyst race (10 -20 µm).
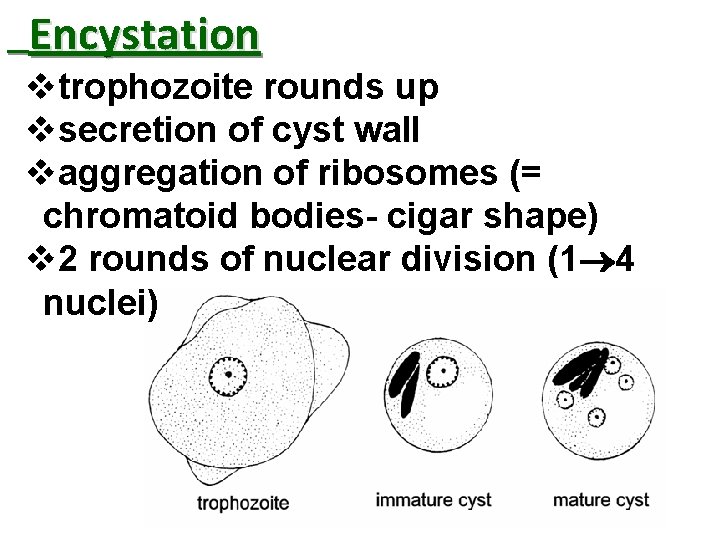
Encystation vtrophozoite rounds up vsecretion of cyst wall vaggregation of ribosomes (= chromatoid bodies- cigar shape) v 2 rounds of nuclear division (1 4 nuclei)

Viability: v. Trophozoite may live for 1 hr at 5 C 0, v Cyst stage viable in stool for 10 days at 22 C 0. v. The cyst does not destroyed by chlorinate of water. v. The trophozoite absorbs food from surrounding tissue including RBCs, fecal materials and bacteria dissolve it by cytolytic enzymes. v. E. histolytica live in poor O 2 atmosphere, under complete or partial anaerobic conditions. It lacks the mitochondria.

Strains: v. Non pathogenic“commensal” E. hartmani. v. Avirulent minute form (small cyst race), E. dispar. v. Virulent magna form (large cyst race), E. histolytica.

E. dispar: q. Entamoeba histolytica isolates from pathogenic (invasive) and non-pathogenic (non-invasive) patients exhibit many phenotypic and genotypic differences. q. Substantial biochemical and molecular indicates that the non-pathogenic isolates of E. histolytica are genetically distinct from the pathogenic isolates.

E. dispar: q. The pathogenic isolates (invasive) known as E. histolytica and non-pathogenic (noninvasive) known as E. dispar. q. E. histolytica is observed with ingested red blood cells (erythrophagocystosis) while E. dispar non.

Epidemiology: • The infection is due to transmission of mature cysts with contaminated foods (fruit, vegetables), drinking water or fecally contaminated hands of infected persons or carriers. • Flies and cockroaches can function as mechanical transmitters by carrying cysts from the feces to foods.

• In contrast to the vegetative forms, the cysts are quite resistant in a moist environment (i. e. , they survive at 28– 34 C 0 for about eight days, at 10 C 0 for about one month); under conditions of desiccation and temperatures exceeding 55 C 0 they are quickly killed. • The amounts of chlorine normally added to drinking water are insufficient to kill the cysts.
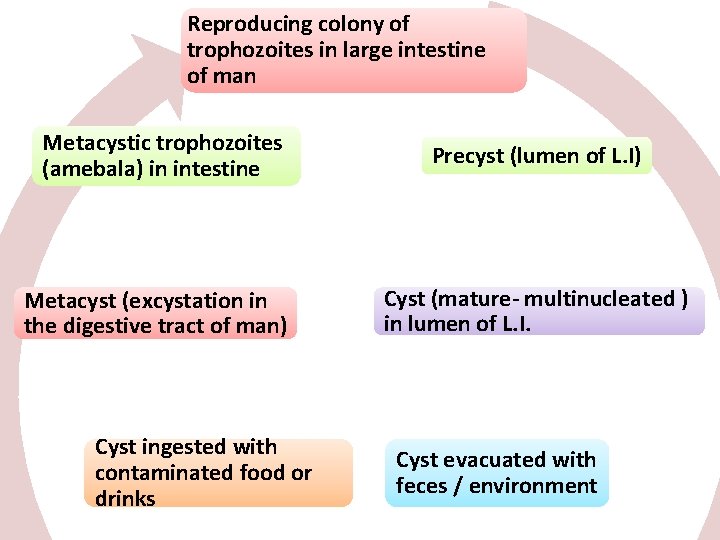
Reproducing colony of trophozoites in large intestine of man Metacystic trophozoites (amebala) in intestine Precyst (lumen of L. I) Metacyst (excystation in the digestive tract of man) Cyst (mature- multinucleated ) in lumen of L. I. Cyst ingested with contaminated food or drinks Cyst evacuated with feces / environment
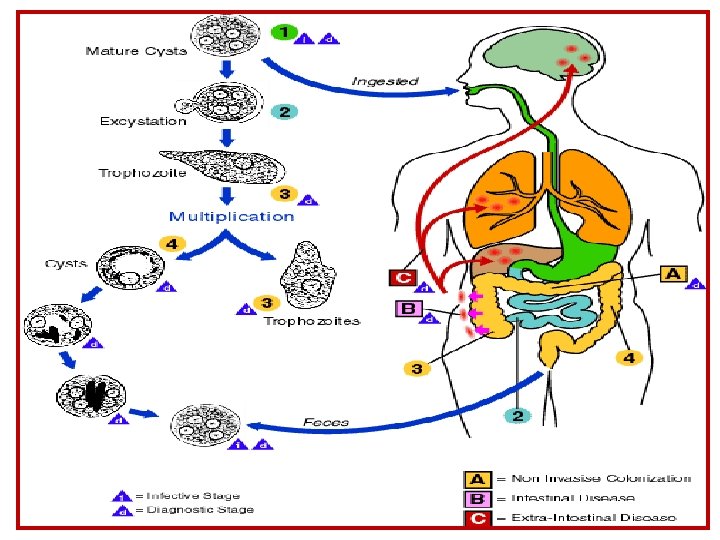
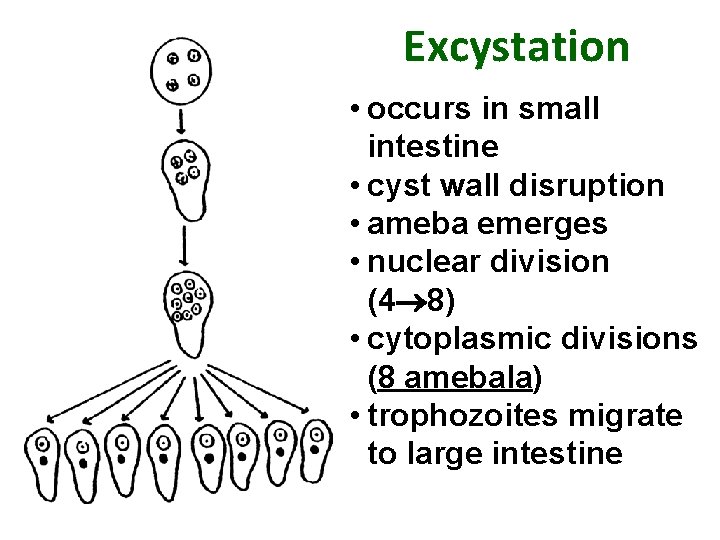
Excystation • occurs in small intestine • cyst wall disruption • ameba emerges • nuclear division (4 8) • cytoplasmic divisions (8 amebala) • trophozoites migrate to large intestine

Pathogenesis ØE. histolytica is a facultative pathogen. Normally E. histolytica lives in the human large intestine and feeds on the bacterial fauna. ØApproximately 85 -90% of people infected with E. histolytica are asymptomatic carriers. ØAmong the symptomatic patients only 10% will develop severe dysentery or invasive disease.

Pathogenesis Infection with E. histolytica implies colonization. The speed of which depend on many factors: üGeneral condition of the host. üBacterial flora. üDiet. üCytotoxic power of trophozoites upon the leukocytes. üThe protolytic enzyme found in trophozoites. üPathogenic activity of the strain (invasive or non invasive ameba).
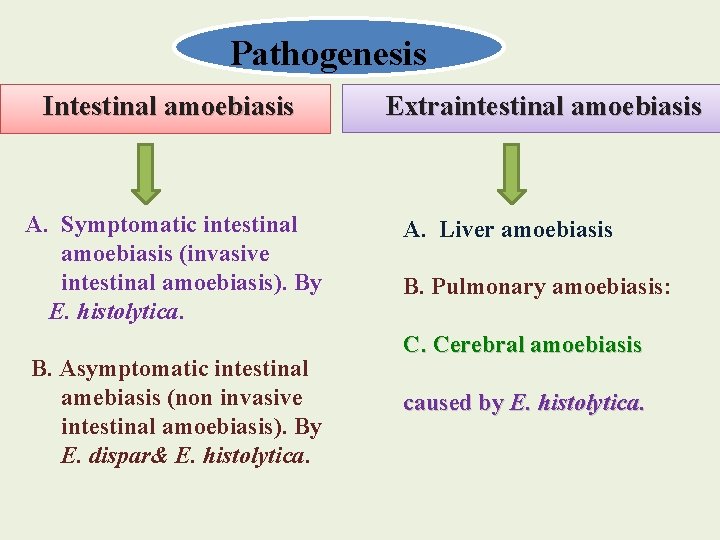
Pathogenesis Intestinal amoebiasis A. Symptomatic intestinal amoebiasis (invasive intestinal amoebiasis). By E. histolytica. B. Asymptomatic intestinal amebiasis (non invasive intestinal amoebiasis). By E. dispar& E. histolytica. Extraintestinal amoebiasis A. Liver amoebiasis B. Pulmonary amoebiasis: C. Cerebral amoebiasis caused by E. histolytica.

Intestinal amoebiasis NON-INVASIVE (asymptomatic intestinal amoebiasis): (E. dispar & E. histolytica) • ameba colony on intestinal mucosa • asymptomatic cyst passer • non-dysenteric diarrhea, abdominal cramps, other GI symptoms. INVASIVE (symptomatic intestinal amoebiasis): (E. histolytica) • necrosis of mucosa ulcers, dysentery • ulcer enlargement severe dysentery, colitis, peritonitis • metastasis extraintestinal amoebiasis

Symptomatic intestinal amoebiasis (invasive intestinal) Cause by virulent strain of E. histolytica. I. Acute intestinal amoebiasis (acute amebic dysentery) : The trophozoites colonize the large intestine mucosa or lumen (colon, cecum, rectum, sometimes terminal ileum). Initially at the coecal level. The characteristic lesion is a superfacial minute cavity resulting from necrosis of mucosal epithelium.

The lesion enlarged somewhat as the ameba reach the muscular mucosa, then gradually it erode a passage through the muscularis mucosa to submucosa and serosa where they spread to the surrounding tissues.
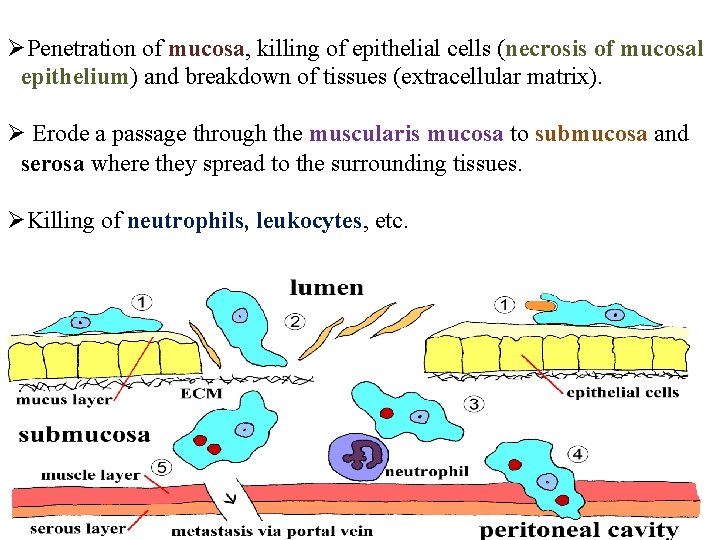
ØPenetration of mucosa, killing of epithelial cells (necrosis of mucosal epithelium) and breakdown of tissues (extracellular matrix). Ø Erode a passage through the muscularis mucosa to submucosa and serosa where they spread to the surrounding tissues. ØKilling of neutrophils, leukocytes, etc.
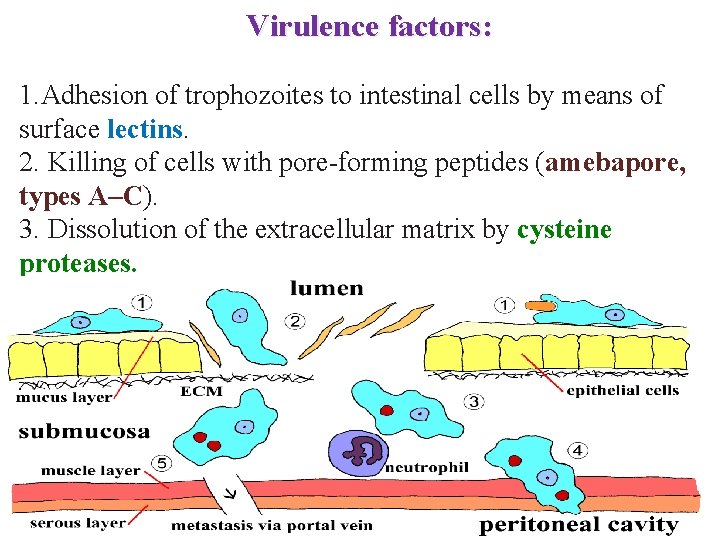
Virulence factors: 1. Adhesion of trophozoites to intestinal cells by means of surface lectins. 2. Killing of cells with pore-forming peptides (amebapore, types A–C). 3. Dissolution of the extracellular matrix by cysteine proteases.
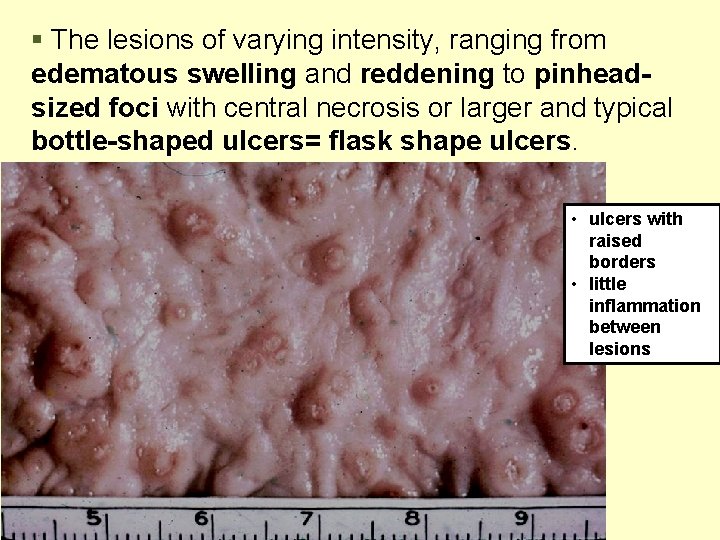
§ The lesions of varying intensity, ranging from edematous swelling and reddening to pinheadsized foci with central necrosis or larger and typical bottle-shaped ulcers= flask shape ulcers. • ulcers with raised borders • little inflammation between lesions
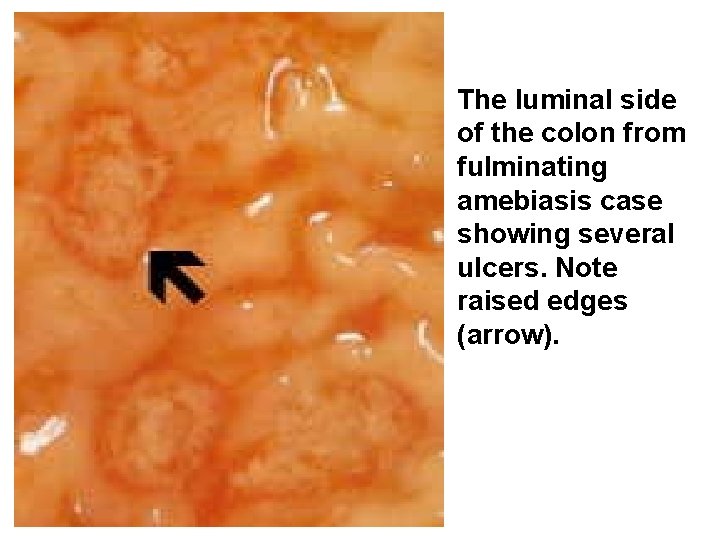
The luminal side of the colon from fulminating amebiasis case showing several ulcers. Note raised edges (arrow).
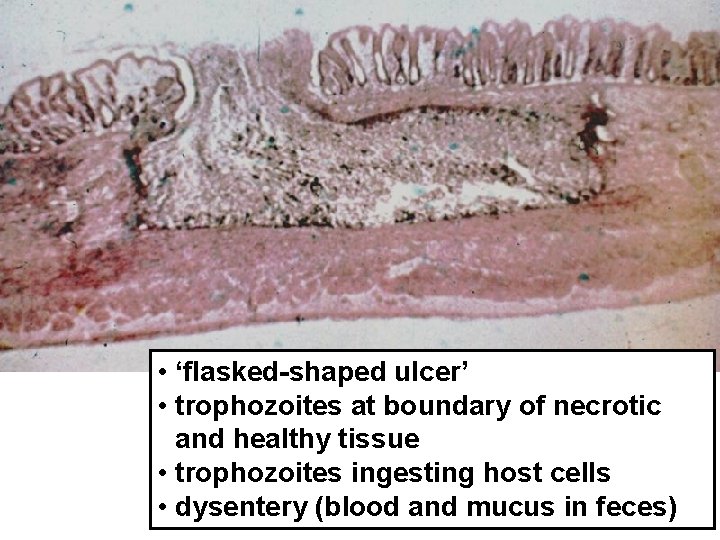
• ‘flasked-shaped ulcer’ • trophozoites at boundary of necrotic and healthy tissue • trophozoites ingesting host cells • dysentery (blood and mucus in feces)
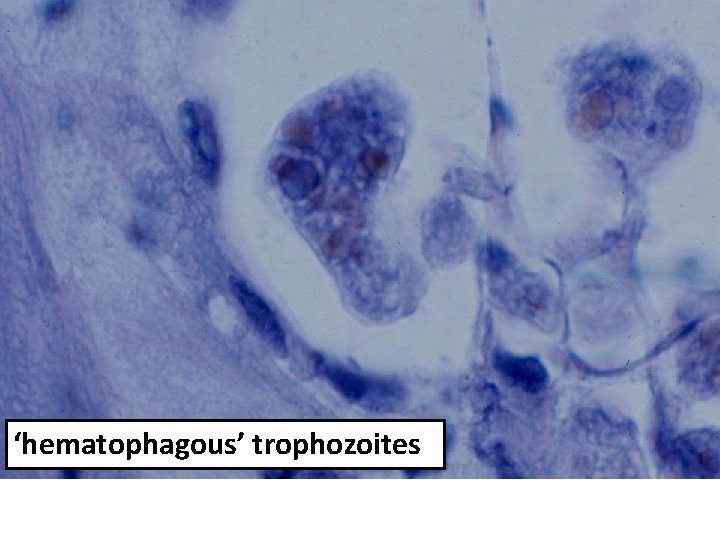
‘hematophagous’ trophozoites
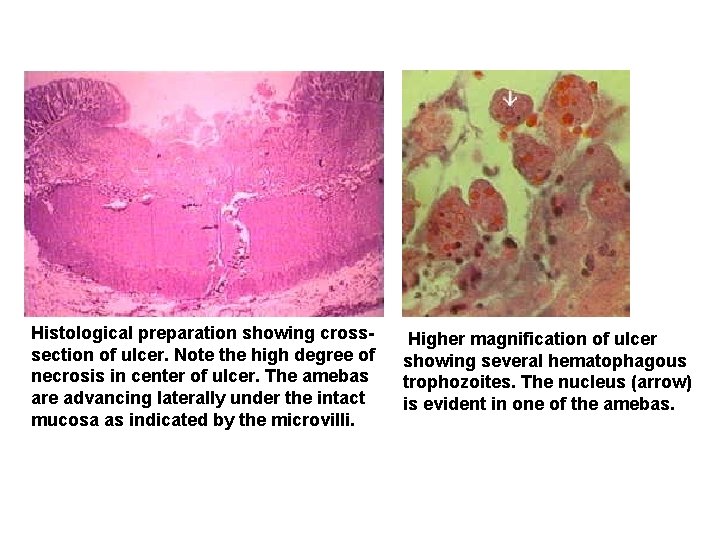
Histological preparation showing crosssection of ulcer. Note the high degree of necrosis in center of ulcer. The amebas are advancing laterally under the intact mucosa as indicated by the microvilli. Higher magnification of ulcer showing several hematophagous trophozoites. The nucleus (arrow) is evident in one of the amebas.

Deep ulcer which is limited to submucosa or extend deeper to muscular layer and serosa causing peritonitis, pericoecal abscess and gangrene of intestine.

Chronic intestinal amoebiasis (chronic amebic dysentery: q. As the lesion become chronic, round cell infiltrating the tissue then become infiltrated with neutrophil and fibroblast which tend to form a wall around the margin of the ulcer. The following changes are observed: v. Small ulcer involving only the mucosa. v. Extensive superficial ulcer with hyperemia.
v. Localized thickening of the intestinal wall leading to narrow lumen. v. Formation of tumor like masses (amoebic granuloma) or amoeboma ameboma = inflammatory thickening of intestinal wall around the abscess (can be confused with tumor)

Lateral and Downward Expansion of Ameba into Lamina Propria • ulcers coalesce • perforation of intestinal wall


Asymptomatic intestinal amebiasis (non invasive) Caused by E. dispar, less frequently by E. histolytica. E. dispar adheres to host cells in very much the same way as E. histolytica, but it produces only very small amounts of amebapore A and B and none of the particularly potent type C at all. E. dispar is lacking several genes that code for certain cysteine proteases. The activity of certain proteases in E. dispar is greatly reduced compared to E. histolytica.
Clinical manifestations develop as early as two to four weeks after infection with E. histolytica or after asymptomatic periods of months or even years. Asymptomatic intestinal form • ameba colony on intestinal mucosa • asymptomatic cyst passer (1000 cyst/day) • non-dysenteric diarrhea, abdominal cramps, other GI symptoms. • Antibodies to E. histolytica antigens are usually not found in serum.

Invasive intestinal form: Caused by. E. histolytica. The acute disease • Usually begins with abdominal discomfort and episodes of diarrhea. • The diarrhea of varying duration, at first mushy then increasing mucoid, including blood-tinged, so-called “red currant jelly stools = dysentery = bloody diarhea” in which amebas can detected, including trophozoites containing erythrocytes. • Fever, dehydration and toxemia can also present. • In such cases, antibodies are usually present in serum.

In chronic amoebiasis v the ulcers sometimes perforate into the peritoneal cavity. The symptoms include: 1. recurrent attacks of dysentery, 2. constipation, 3. loss of weight, 4. ulcerative colitis and 5. one or more amoeboma 6. Obstruction of intestine due to healing processes with scar formation may reduce the intestinal lumen.

Extraintestinal Amebiasis
Extraintestinal Amebiasis metastasis via blood stream • primarily liver (portal vein) • other sites less frequent • (lung, brain, skin and spleen) ameba-free stools common • high antibody titers •
Liver abscess Liver amebiasis

q. Early liver abscess is small oval mass. contains a brownish or yellowish, puslike liquid, in most cases bacteriologically sterile, q later becoming a necrotic mass with grayish matrix of necrosed hepatic cells; qamebas are often only detectable in the transition zone between the lesion and intact hepatic tissue. q. Later the abscess increase in size, the center liquefies, the wall is thick, and the content becomes chocolate reddish mass of hepatic cells, RBCs and bile fat.
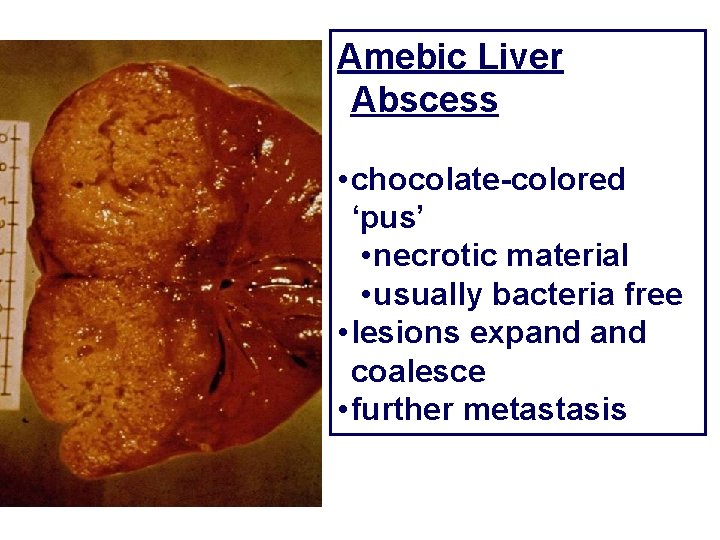
Amebic Liver Abscess • chocolate-colored ‘pus’ • necrotic material • usually bacteria free • lesions expand coalesce • further metastasis
Clinical manifestations: • The liver abscess causes remittent fever (sometimes high), upper abdominal pain, liver enlargement, elevation of the diaphragm, general weakness, and other symptoms. • Large liver abscesses that are not treated in time are often lethal. Antibodies are detectable in most cases (around 95%).

Pulmonary amoebiasis: • Primary: rare condition occurs even without hepatic amoebiasis. The trophozoites can reach the pulmonary capillaries (single or multiple), via the portal circulation. • Secondary: arise as a complication of liver abscess from the liver to the base of right lung, resulting in pneumonia.
Pulmonary Amebiasis rarely primary rupture of liver abscess through diaphragm 2 o bacterial infections common fever, cough. • •

• Cerebral amoebiasis: A rare complication of hepatic or pulmonary amoebiasis or of both. It is single and of small size located mostly in one of the cerebral hemisphere. • Splenic amoebiasis: found in association with hepatic abscess.

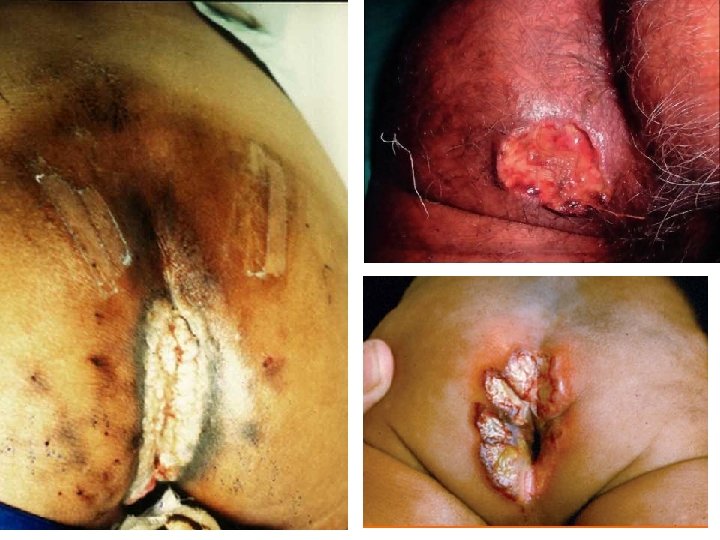
Cutaneous amoebiasis (Amebiasis cutis) Cutaneous amoebiasis most frequently occurs in the perianal area, associated with rectal changes or may develop when the skin is in prolonged contact with amoeba from any cause, such as areas of draying of liver abscess, or colostomy wound in the site of ruptured appendicular and peri-colic abscesses. Also the granulomatas ulceration in perianal region.
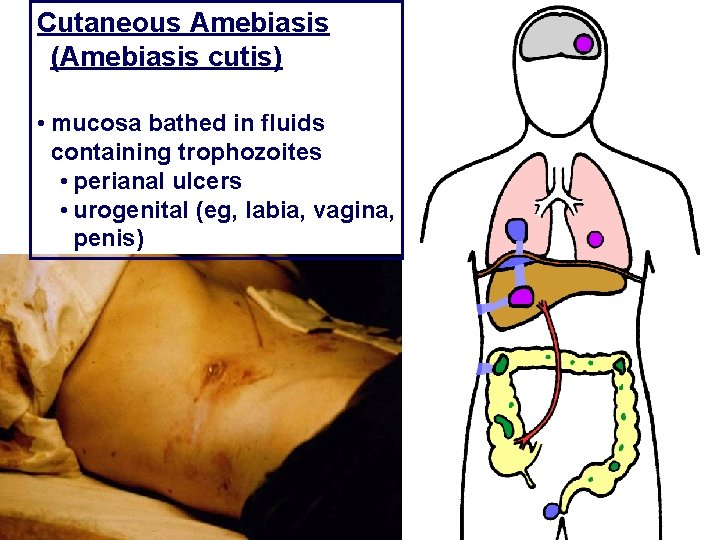
Cutaneous Amebiasis (Amebiasis cutis) • mucosa bathed in fluids containing trophozoites • perianal ulcers • urogenital (eg, labia, vagina, penis)

Immunity: Extracellular protozoa (include E. histolytica, are eliminated by: Øphagocytosis and complement activation. Ø Th 2 cytokines released for antibody production. ØAbs responses includes: opsonization, complement activation and ADCC.

ØReinfections are possible since sufficient immunity is not conferred in the course of an infection. ØAntibodies are usually detectable in serum in invasive intestinal and extraintestinal amoebiasis caused by E. histolytica.

Diagnosis: üIntestinal infections: A. Stool examination to identify: 1. Cyst in formed stool. 2. Cyst and troph in diarrheal stool. • Charcot Leyden crystals present. B. Sigmoidoscopy. C. Serological tests for chronic & extra intestinal cases. D. Culture of feces. E. Animal inoculation. F. Blood examination for leukocytosis.

Diagnosis: üExtraintestinal amoebiasis: Hepatic amoebiasis: based mainly on aspirate & liver biopsy to identify trophozoites. Pulmonary amoebiasis: based on identify trophozoites in sputum sample.
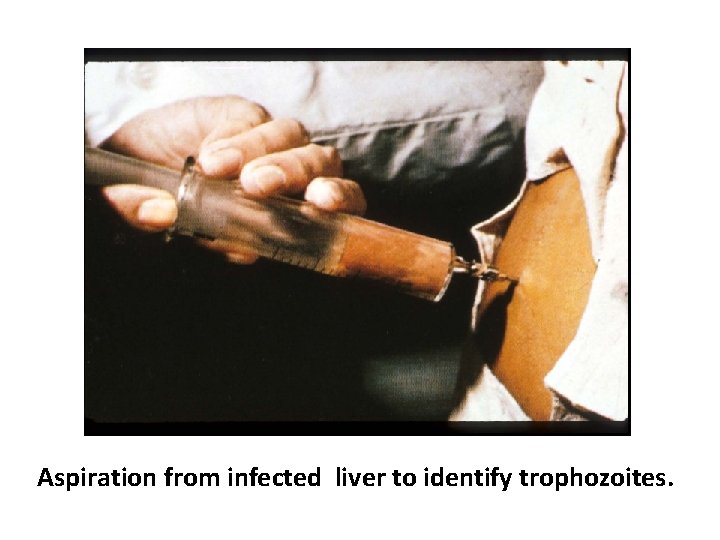
Aspiration from infected liver to identify trophozoites.

Treatment: For symptomatic intestinal disease, or extra intestinal, infections (e. g. , hepatic abscess), the drugs of choice are metronidazole. Control: • Personal hygiene. • Group hygiene. • Protection of water supply from being contaminated with feces. • Avoid using human feces as fertilizer.
- Slides: 59