Protons for Breakfast Electricity Week 1 November 2014

Protons for Breakfast Electricity Week 1 November 2014
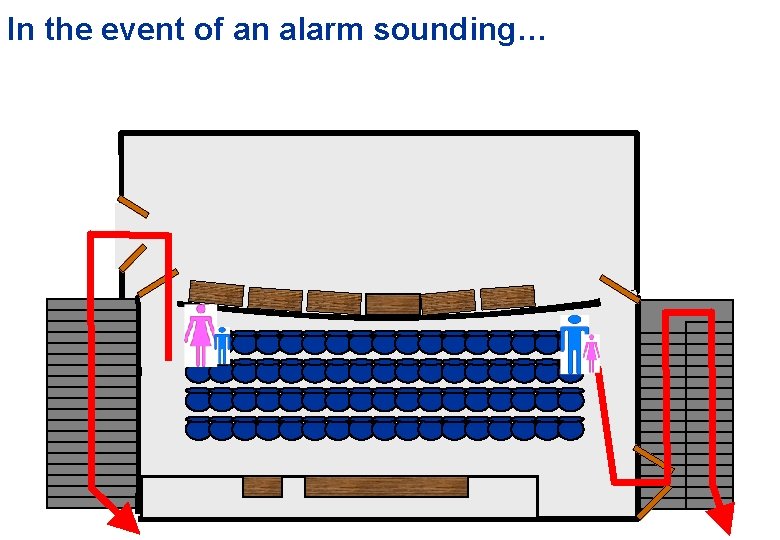
In the event of an alarm sounding…
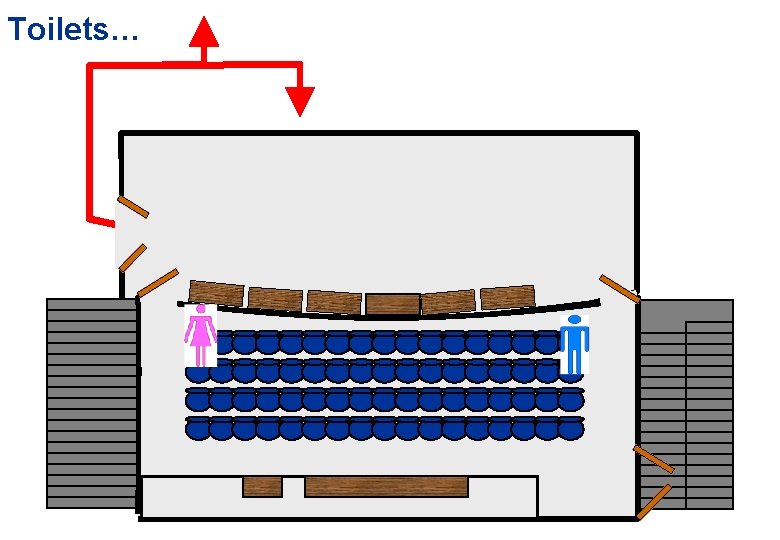
Toilets…

Parents and children…
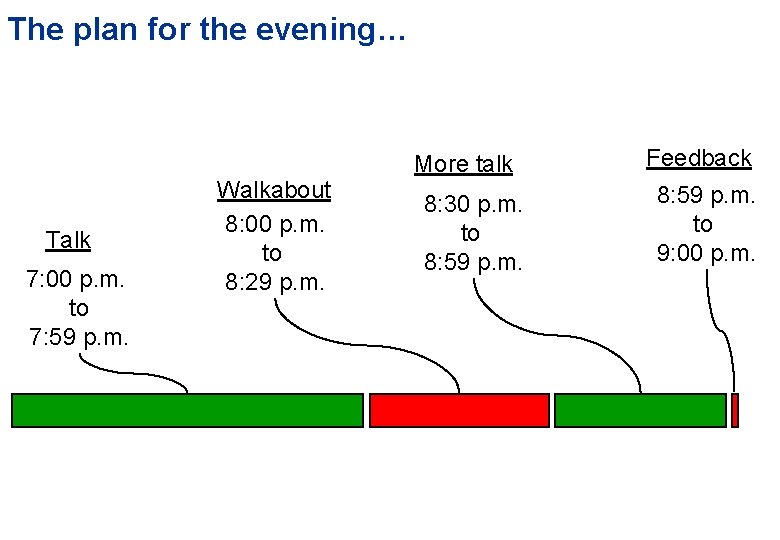
The plan for the evening… Talk 7: 00 p. m. to 7: 59 p. m. Walkabout 8: 00 p. m. to 8: 29 p. m. More talk 8: 30 p. m. to 8: 59 p. m. Feedback 8: 59 p. m. to 9: 00 p. m.
Amelia Marks Who is helping? Amir Kayani Andrew Hanson Andy Knott Averil Horton Bo Li Claire Greenwell Clark Stacey Dale Partridge David Clay Deborah Lea Edward Brightman Gianluca Memoli James Claverley James Miall Jane Burston Jenny Hully Jessica Cross Brown Joanne Nightingale John Gallop John Makepeace John Mountford Jon Helmore Jonathan Pearce Jordan Tompkins Joseph Thom Kate Wilkinson Kieran Edmonds Laura Duke Lauren Petrie Leigh Stanger Lindsay Chapman Lloyd England Louise Brown Maria Lodeiro Marieke Beckmann Marta Doval Minarro Matt Cashmore Michael Mc. Carthy Nadia Smith Paul Carroll Paul Green Peter Nisbet-Jones Peter Quested Peter Woolliams Rainer Winkler Ralf Mouthaan Robert Goddard Ruth Pearce Sharmila Hanson Sam Alvey-Taylor Sam Flynn Sean Woodward Stephanie Bell Subrena Harris Sue Gibbons Sue Oakley Tracey Skinner Vicky Hall

Michael de Podesta Age 20, 036 days: Weight 85 kg • Lecturer in Physics at University of London for 13 years • Understanding the Properties of Matter • At NPL for 14 years. • Most accurate thermometer ever made! • International Surface Temperature Initiative • Married with two sons (aged 15 & 17) • Keen on Water Rockets

Why am I here? I am here because I believe … Science is humanity’s greatest achievement

Why are you here? • • • Accompanying Grandmother To support my daughter who is also attending Always had an interest in science My son loves science I enjoy science and like learning about how the world works. Made to come by parent. Because I'm very interested and it will help me with my science, because I'm not that good So I can teach it to secondary school-aged kids To improve my knowledge in physics and to make it accessible to students.

Why are you here? • • • I am in the top set for science and want to stay there! It sounds really interesting I wish to learn more about science and I think it will be a valuable experience for my 11 year old and 16 year old sons I like science - it's my favourite subject I just want to learn more about life, the universe, and every thing. To help enthuse my daughter before GCSE and to refresh my knowledge and perhaps use some ideas at my work. Enjoyed the NPL Open Day and wanted to learn more. I have developed an interest in science through my son It has a good reputation. I liked science at school but am now rusty. Also I know little about newer technology for instance mobile. I work at NPL in finance, I want to get more knowledge on our key product, science!

…there is a problem about how we, as citizens, relate to science…

The image of science: 1 Mad Muppets top cult science poll Dr Honeydew is known the world over for his disastrous research at Muppet Labs, "where the future is being made today". His experiments invariably go awry, with poor old Beaker usually being blown to bits or electrocuted. BBC 6/9/2004
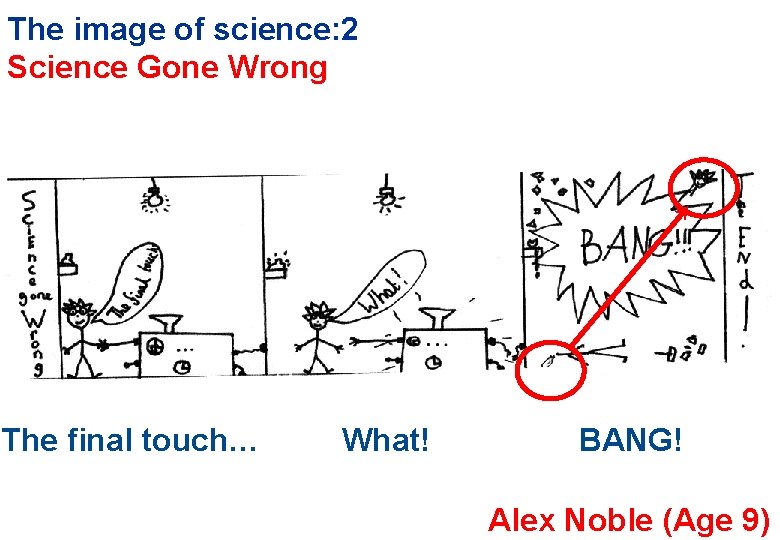
The image of science: 2 Science Gone Wrong The final touch… What! BANG! Alex Noble (Age 9)

The image of science: 3 An un-scientific experiment Scientist ……… Scientist

In contrast… • A room full of people who want to learn about science • Helped by volunteers • In a world where ignorance makes us powerless

Tonight’s talk • The scale and size of the Universe • Electricity • How Electricity works • Atoms • Light

Tonight’s talk The scale and size of the Universe or ‘How not to be boggled!’

You area i z a m ! g n
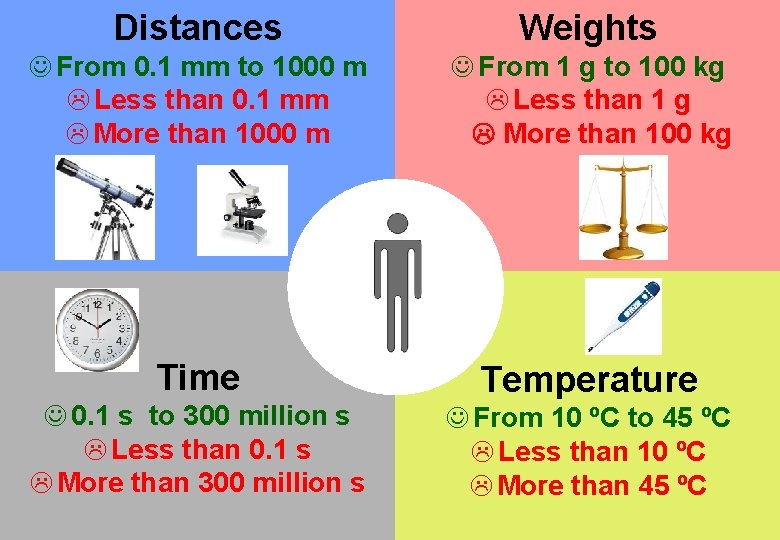
Distances Weights J From 0. 1 mm to 1000 m L Less than 0. 1 mm L More than 1000 m J From 1 g to 100 kg L Less than 1 g More than 100 kg Time Temperature J 0. 1 s to 300 million s L Less than 0. 1 s L More than 300 million s J From 10 ºC to 45 ºC L Less than 10 ºC L More than 45 ºC
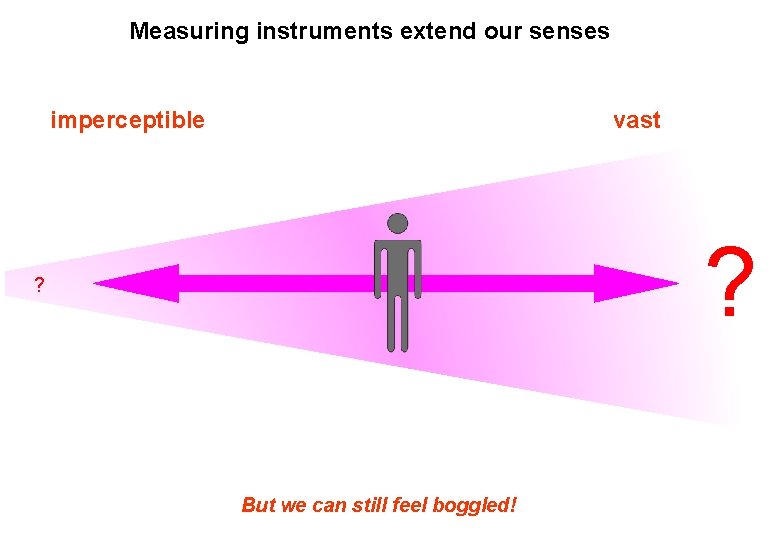
Measuring instruments extend our senses imperceptible vast ? ? Science helps us extend our senses But we can still feel boggled!

The Planet Earth Diameter: 12, 800 km Deepest hole: 10 km Atmosphere: 10 km Photo Credit: NASA

The Moon Diameter Earth: 12, 800 km Moon: 3476 km Photo Credit: NASA

The Sun Diameter: 1, 390, 800 km Photo Credit: NASA

Powers of Ten I hope that you are now a little unsettled and ready to go on a 9 minute journey to see how the world looks at different levels of ‘fantasy magnification’ Photo Credit: Powers of 10
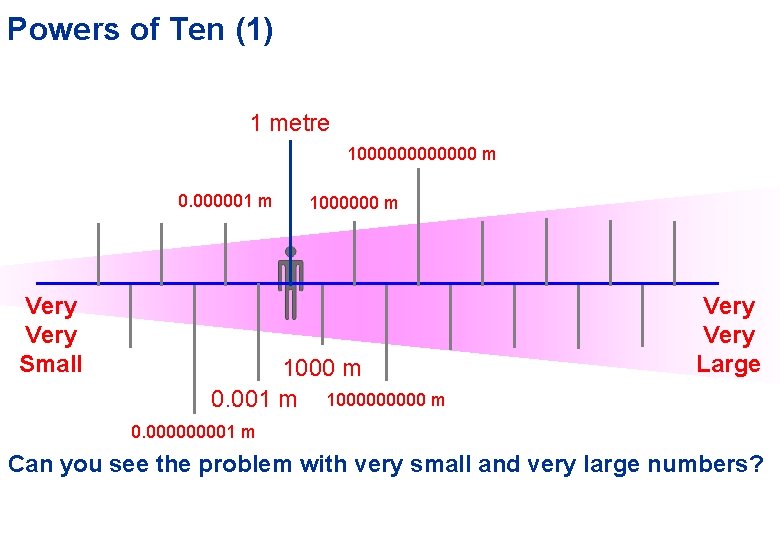
Powers of Ten (1) 1 metre 1000000 m 0. 000001 m Very Small 1000000 m 1000 m 0. 001 m 100000 m Very Large 0. 00001 m Can you see the problem with very small and very large numbers?
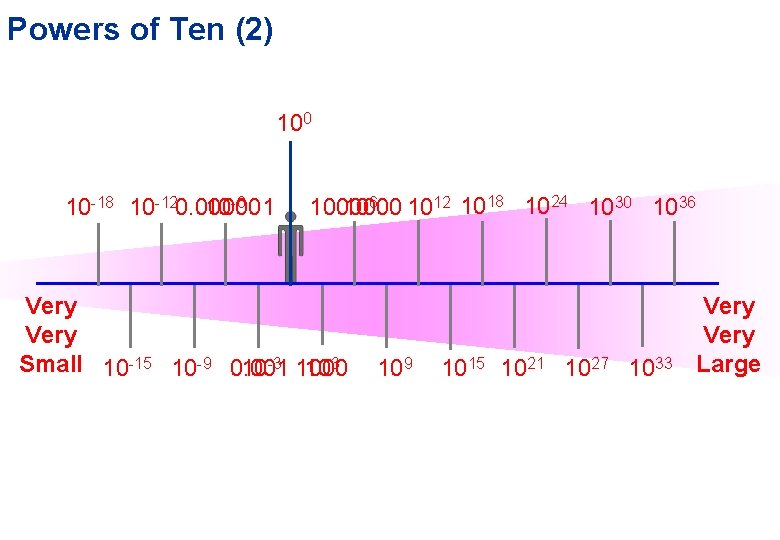
Powers of Ten (2) 10 10 10 -18 10 -120. 000001 10 -6 1000000 106 1012 1018 1024 1030 1036 Very Small 10 -15 10 -9 0. 001 10 -3 1000 103 109 1015 1021 1027 1033 Very Large
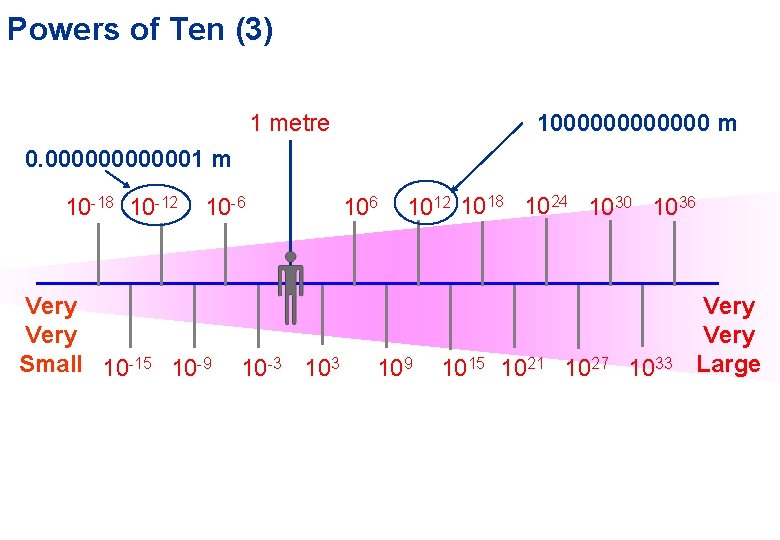
Powers of Ten (3) 1 metre 1000000 m 0. 0000001 m 10 -18 10 -12 10 -6 Very Small 10 -15 10 -9 10 -3 106 1012 1018 1024 1030 1036 109 1015 1021 1027 1033 Very Large
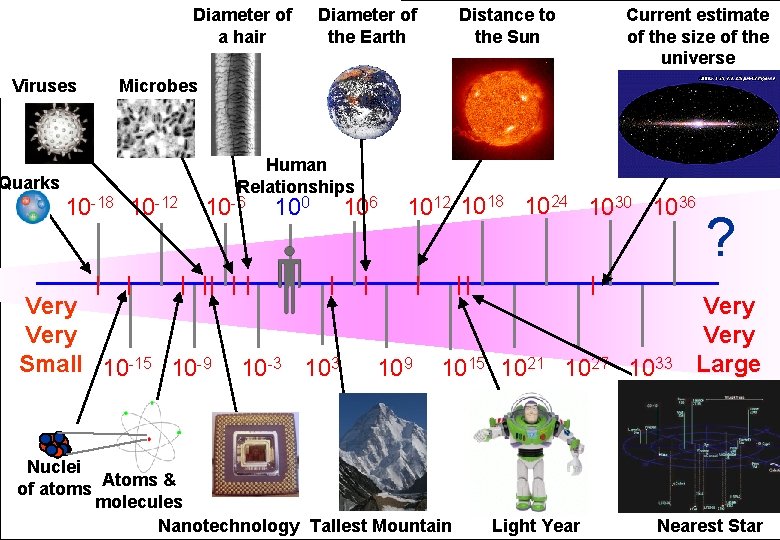
Diameter of a hair Distance to the Sun Diameter of the Earth Powers of Ten Length Scale in metres Microbes Viruses Quarks 10 -18 10 -12 Human Relationships 10 -6 Very Small 10 -15 10 -9 100 10 -3 106 Current estimate of the size of the universe 1012 1018 1024 1030 1036 109 1015 1021 1027 1033 Nuclei of atoms Atoms & molecules Nanotechnology Tallest Mountain Light Year ? Very Large Nearest Star
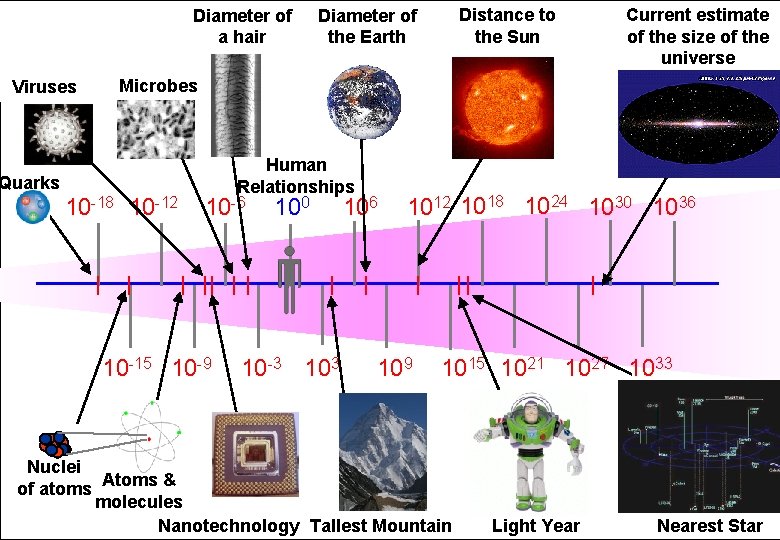
Diameter of a hair Distance to the Sun Diameter of the Earth Powers of Ten Length Scale in metres Microbes Viruses Quarks 10 -18 10 -12 Human Relationships 10 -6 10 -15 10 -9 100 10 -3 106 Current estimate of the size of the universe 1012 1018 1024 1030 1036 109 1015 1021 1027 1033 Nuclei of atoms Atoms & molecules Nanotechnology Tallest Mountain Light Year Nearest Star
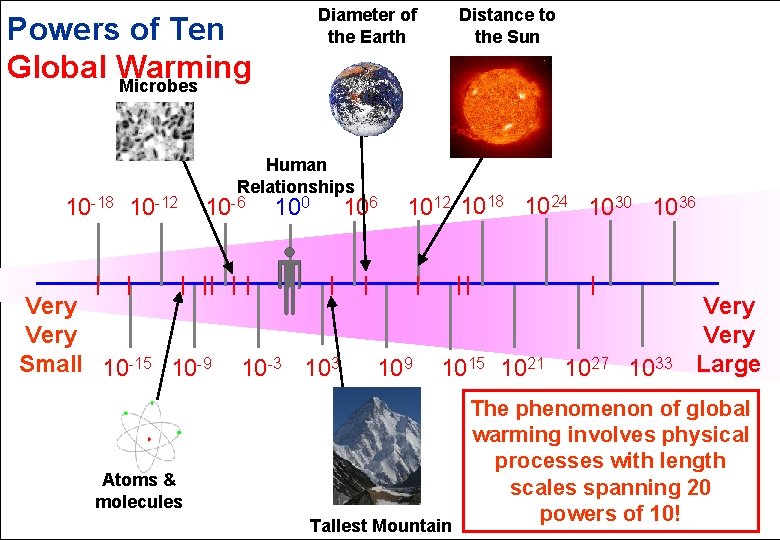
Diameter of the Earth Powers of Ten Global Warming Microbes 10 -18 10 -12 Human Relationships 10 -6 Very Small 10 -15 10 -9 100 106 10 -3 103 Distance to the Sun 1012 1018 1024 1030 1036 109 1015 1021 1027 1033 Atoms & molecules Tallest Mountain Very Large The phenomenon of global warming involves physical processes with length scales spanning 20 powers of 10!
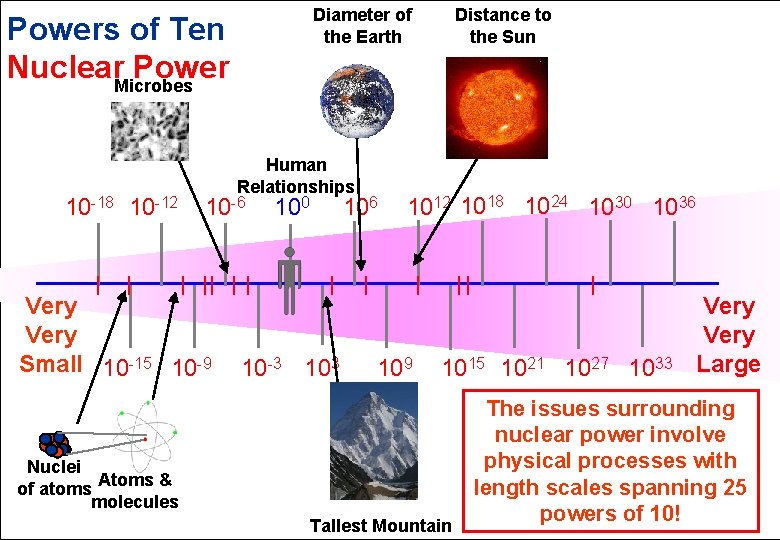
Diameter of the Earth Powers of Ten Nuclear. Microbes Power 10 -18 10 -12 Human Relationships 10 -6 Very Small 10 -15 10 -9 100 106 10 -3 103 Distance to the Sun 1012 1018 1024 1030 1036 109 1015 1021 1027 1033 Nuclei of atoms Atoms & molecules Tallest Mountain Very Large The issues surrounding nuclear power involve physical processes with length scales spanning 25 powers of 10!
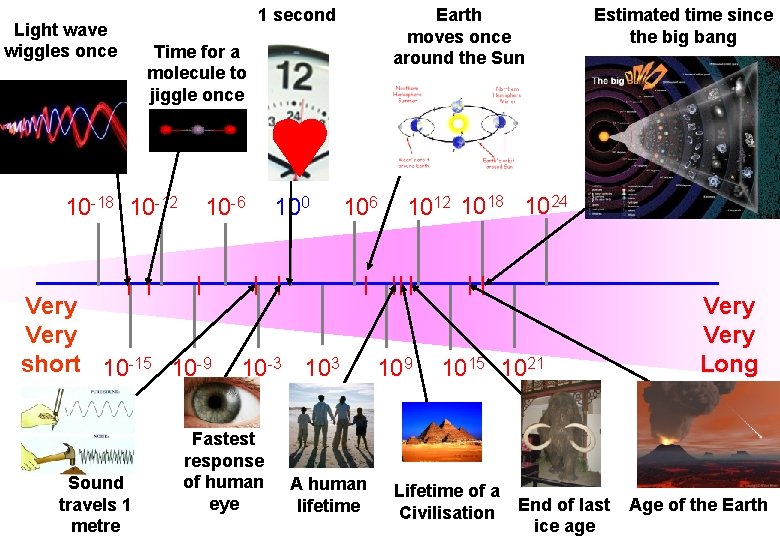
Earth moves once around the Sun 1 second Powers of Ten (time) Time for a Time scale in seconds molecule to Light wave wiggles once Estimated time since the big bang jiggle once 10 -18 10 -12 10 -6 Very short 10 -15 10 -9 Sound travels 1 metre 100 106 10 -3 103 Fastest response of human eye A human lifetime 1012 1018 1024 109 1015 1021 Lifetime of a Civilisation End of last ice age Very Long Age of the Earth
The Universe: It is very big, but full of very small things. imperceptible ? vast ?

Tonight’s talk • The scale and size of the Universe It’s very big, but full of very small things • Electricity • How it works • Atoms • Light
How it all fits together… Electricity Atoms Electromagnetic waves Heat

Eeeee. Electricity - lec- tric-ity

Electricity Some experiments…
Lets take a look at some odd phenomena… • A balloon and a piece of paper

Lets take a look at some odd phenomena… • If I balance my glasses carefully…
Even a sausage… • Sausages…

…its everything… The balloon affects anything and everything nearby To understand this, we need to understand • what matter is made of, and • how this ‘influence’ is communicated across ‘space’

A simple scientific instrument: The gold leaf electroscope • • Scientists can develop instruments to measure the relative strengths of the ‘electric influence’ Based on the same effect we saw with bits of paper

The Van de Graaff Generator • Scientists can develop machines to automate and amplify the ‘rubbing’ process with the balloon Photo Credits: Katherine Robinson and MIT
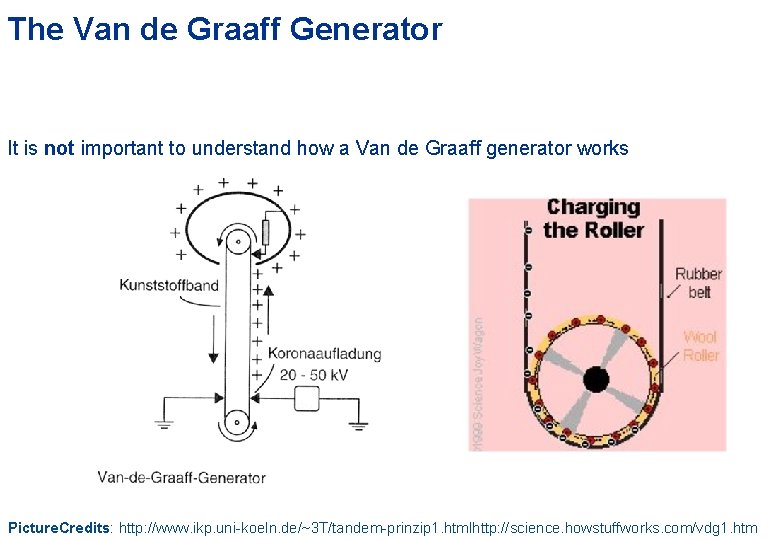
The Van de Graaff Generator It is not important to understand how a Van de Graaff generator works Picture. Credits: http: //www. ikp. uni-koeln. de/~3 T/tandem-prinzip 1. htmlhttp: //science. howstuffworks. com/vdg 1. htm
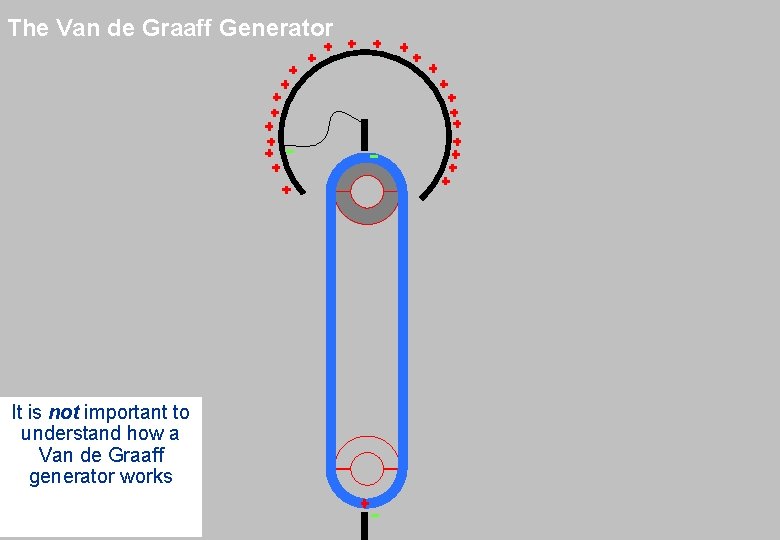
The Van de Graaff Generator It is not important to understand how a Van de Graaff generator works
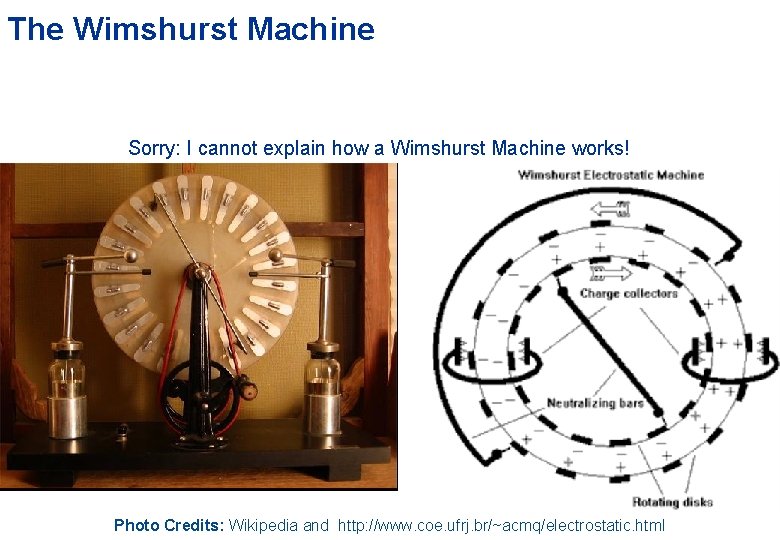
The Wimshurst Machine Sorry: I cannot explain how a Wimshurst Machine works! Photo Credits: Wikipedia and http: //www. coe. ufrj. br/~acmq/electrostatic. html

Electrostatic Generators • People have been doing this for a long time… Photo Credits: http: //www. ikp. uni-koeln. de/~3 T/tandem-prinzip 1. html

Conclusion… Electricity is present inside ALL matter Its ‘influence’ can be communicated across ‘empty’ space

Tonight’s talk • The scale and size of the Universe Its very big, but full of very small things • Electricity It’s everywhere! • How it works • Atoms • Light

How do we describe the world? Stuff • matter Not Stuff • the gaps in between matter • fields

How do we describe the world?

How do we describe the world? Two different kinds of physical entity Stuff (Particles) • • Atoms • Electrons • Neutrons • Protons Very small Not Stuff (Fields) • Fields Gravitational Electroweak Strong • Extend throughout space We need to know about both particles and fields
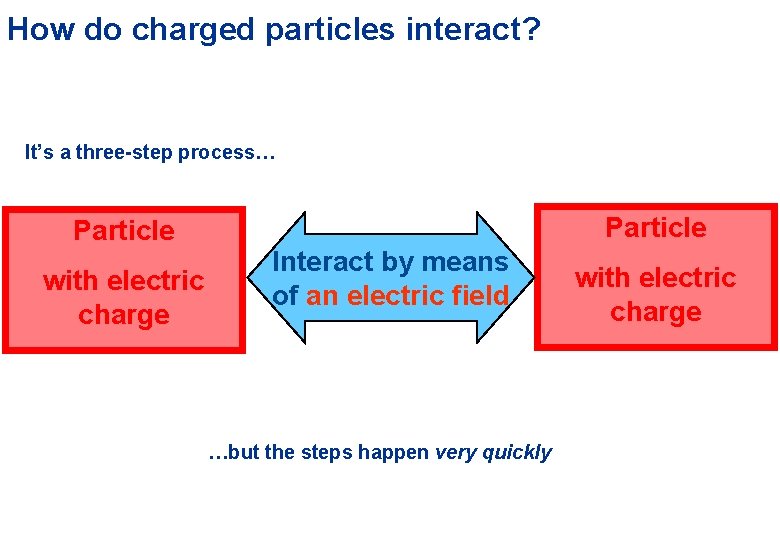
How do charged particles interact? It’s a three-step process… Particle with electric charge Particle Interact by means of an electric field …but the steps happen very quickly with electric charge

How do we describe the world?

The electrical nature of matter • Electric charge is a fundamental property of electrons and protons. • Two types of charge (+ and -) If particles have the same sign of electric charge they repel If particles have different signs of electric charge they attract The forces (attractive or repulsive) get weaker as the particles get further apart.

Tonight’s talk • The scale and size of the Universe Its very big, but full of very small things • Electricity It’s everywhere! • How it works There are ‘particles’ and ‘fields’ • Atoms • Light
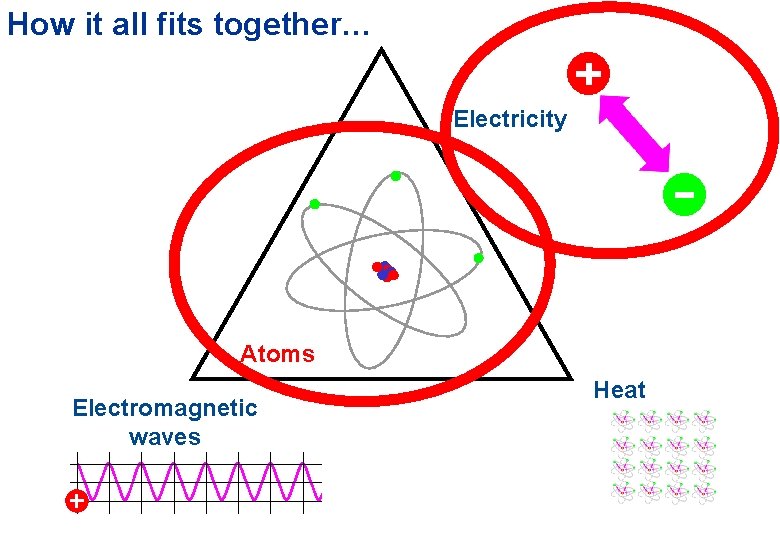
How it all fits together… Electricity Atoms Electromagnetic waves Heat
Atoms Protons, neutrons and electrons normally exist inside atoms
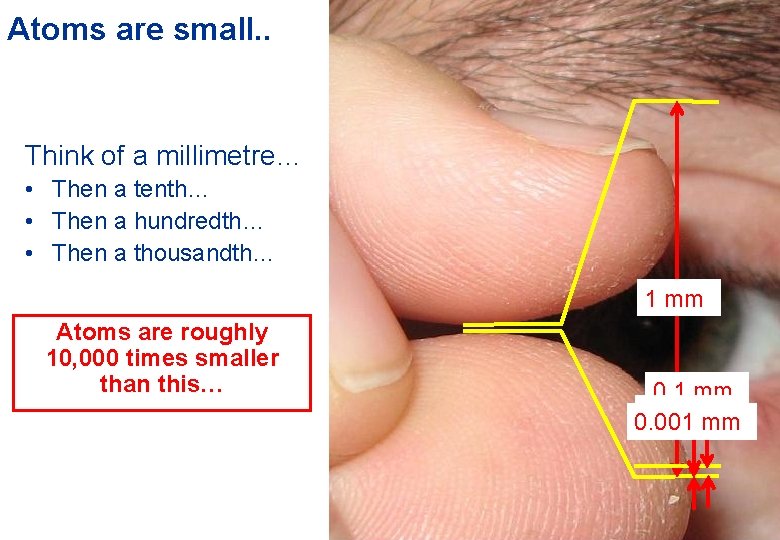
Atoms are small. . Think of a millimetre… • Then a tenth… • Then a hundredth… • Then a thousandth… 1 mm Atoms are roughly 10, 000 times smaller than this… 0. 1 mm 0. 001 mm

Atoms • There are VAST numbers of atoms in everything. In just a handful of anything there about the same number of atoms as there are grains of sand on all the beaches and deserts on Earth combined Photo Credit: http: //www. morguefile. com ID = 104101

The electrical nature of matter Atoms Internal Structure

Tonight’s Drama: The importance of being electrical

How are atoms made? Electrical Repulsion proton Interact by the short range ‘strong’ force – not electrical
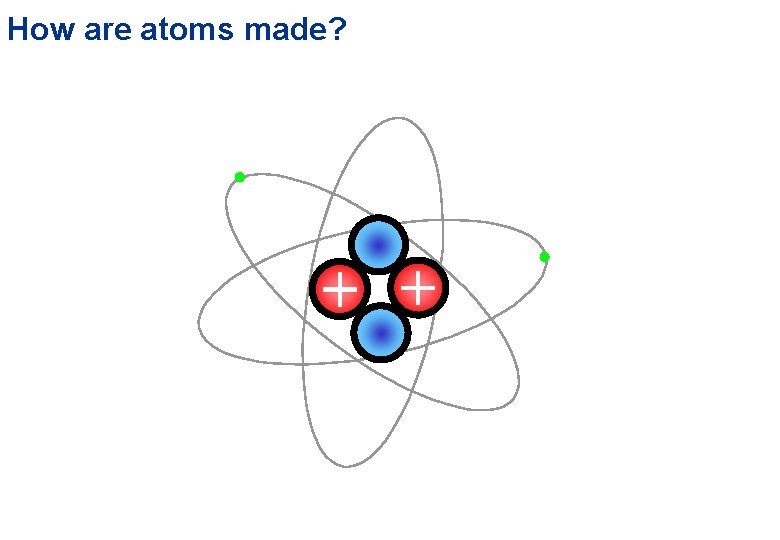
How are atoms made?
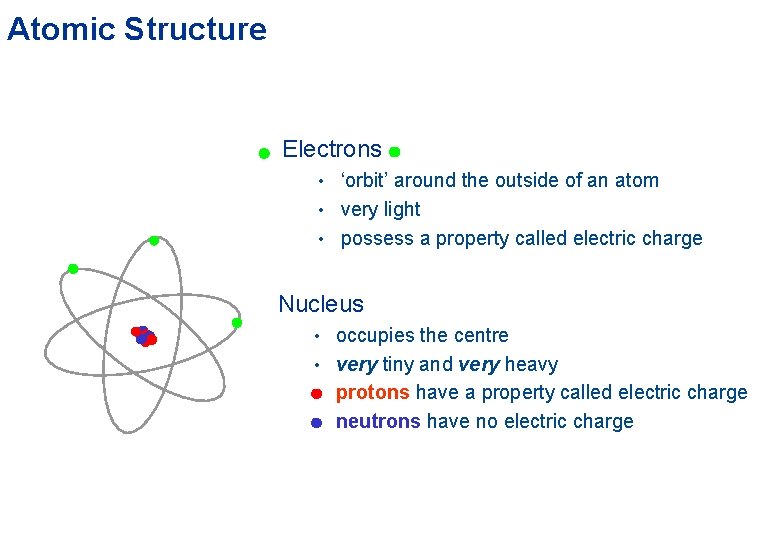
Atomic Structure Electrons • ‘orbit’ around the outside of an atom • very light • possess a property called electric charge Nucleus • occupies the centre • very tiny and very heavy • protons have a property called electric charge • neutrons have no electric charge
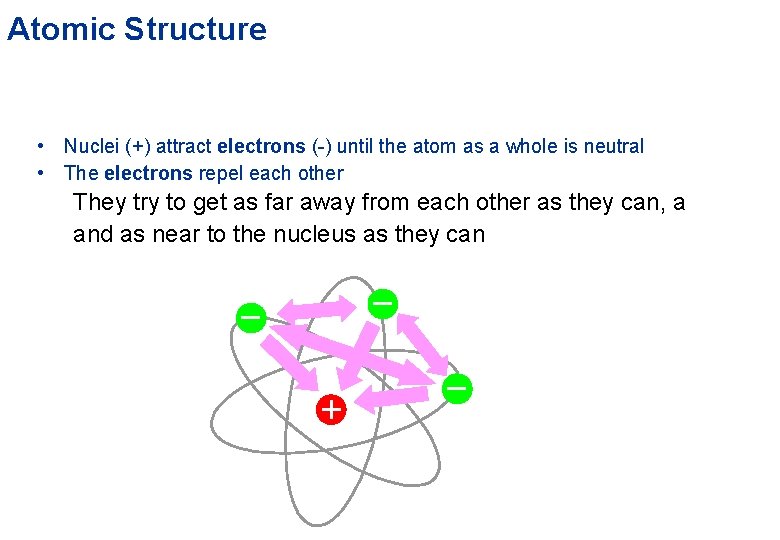
Atomic Structure • Nuclei (+) attract electrons (-) until the atom as a whole is neutral • The electrons repel each other They try to get as far away from each other as they can, a and as near to the nucleus as they can

The electrical nature of matter Chemistry Atoms, Elements & Molecules
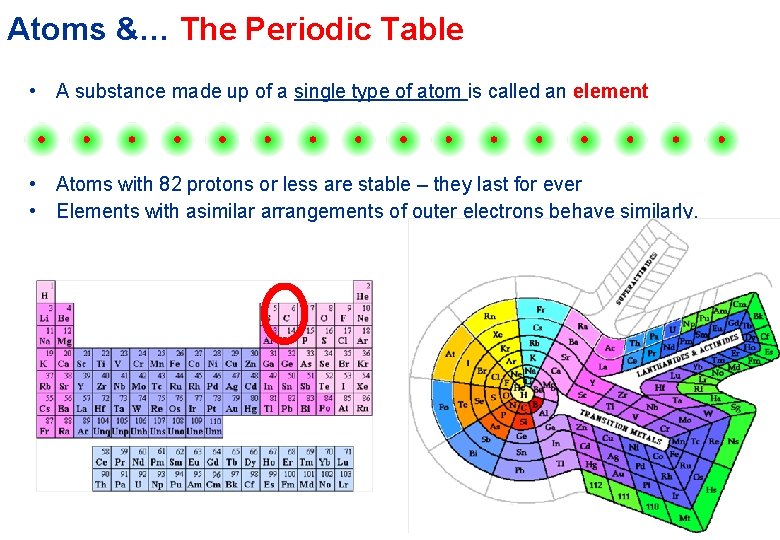
Atoms &… The Periodic Table • A substance made up of a single type of atom is called an element • Atoms with 82 protons or less are stable – they last for ever • Elements with asimilar arrangements of outer electrons behave similarly.
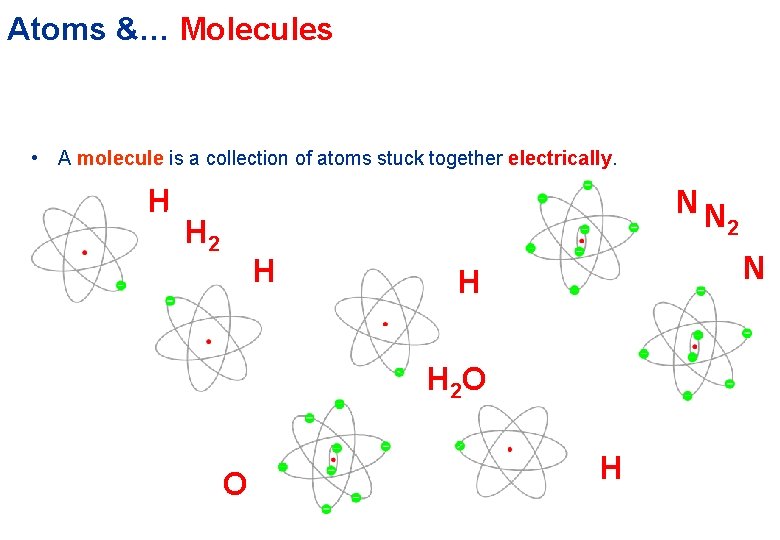
Atoms &… Molecules • A molecule is a collection of atoms stuck together electrically. H NN H 2 H N H H 2 O O 2 H

Second Performance: A Chemical Reaction carbon atom oxygen atom hydrogen atom water molecule carbon dioxide
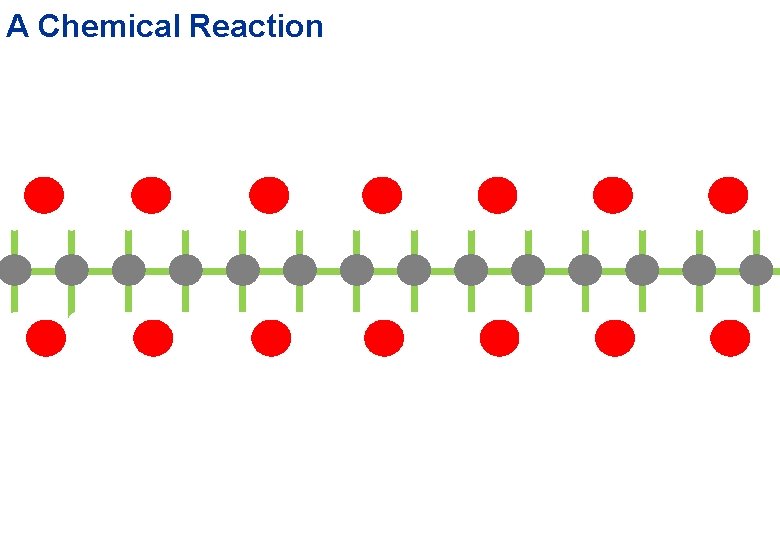
A Chemical Reaction
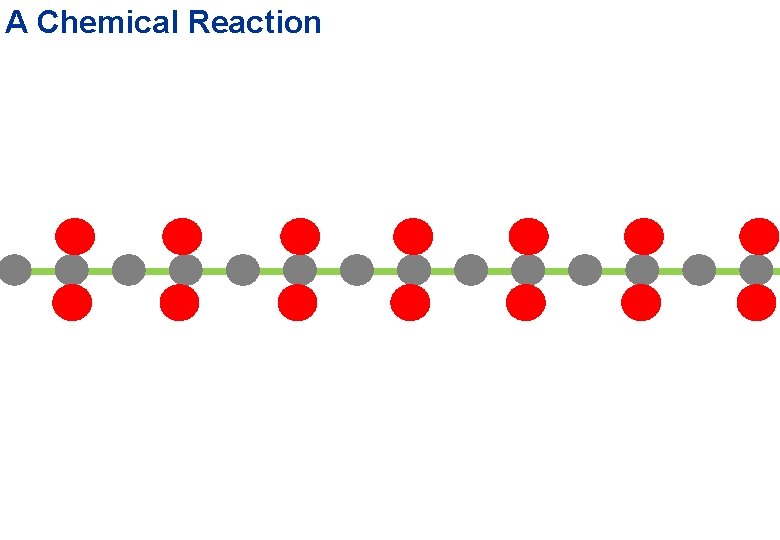
A Chemical Reaction
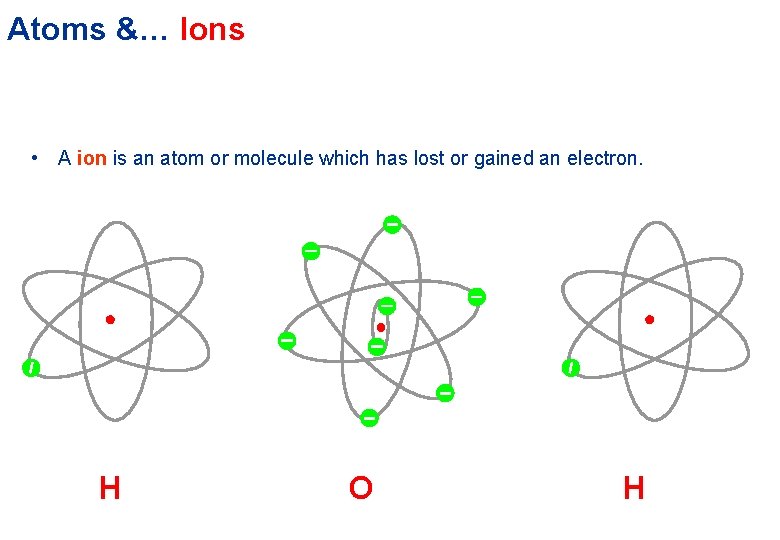
Atoms &… Ions • A ion is an atom or molecule which has lost or gained an electron. H O H

The electrical nature of matter Solids

Atoms in solids • Atoms can be imaged on a surface Photo Credit: Patrick Josephs Franks: NPL

The electrical nature of matter • In ‘normal’ matter, there are equal quantities of positive and negative charge so that there is no attraction or repulsion of objects. Object 1 Object 2

The electrical nature of matter Mechanical Properties
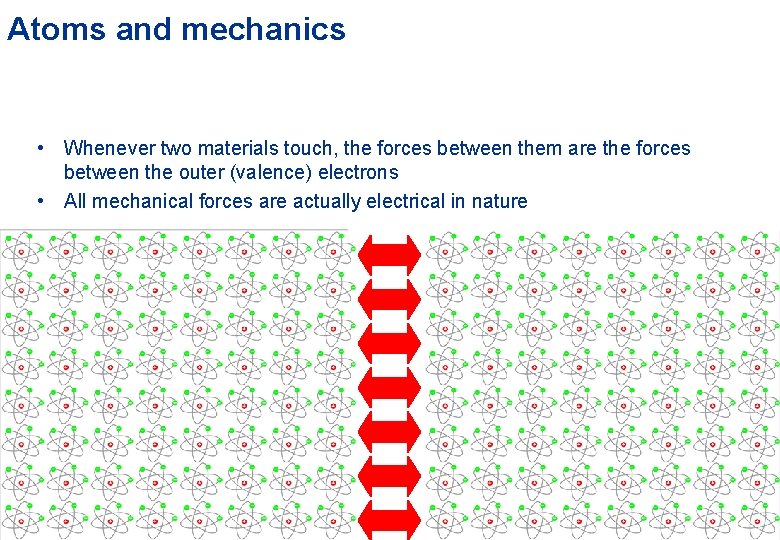
Atoms and mechanics • Whenever two materials touch, the forces between them are the forces between the outer (valence) electrons • All mechanical forces are actually electrical in nature Object 1 Object 2

The electrical nature of matter Conductors and Insulators

Atoms in solids Solids are made up out of lots atoms very close together. If the electrons can’t move easily from atom to atom: The material is called an insulator If the electrons can move easily from atom to atom: The material is called a conductor

The electrical nature of matter Magnetic Forces

Relativity Guten Abend There is no absolute space or time. The laws of physics depend only on the relative motion of objects
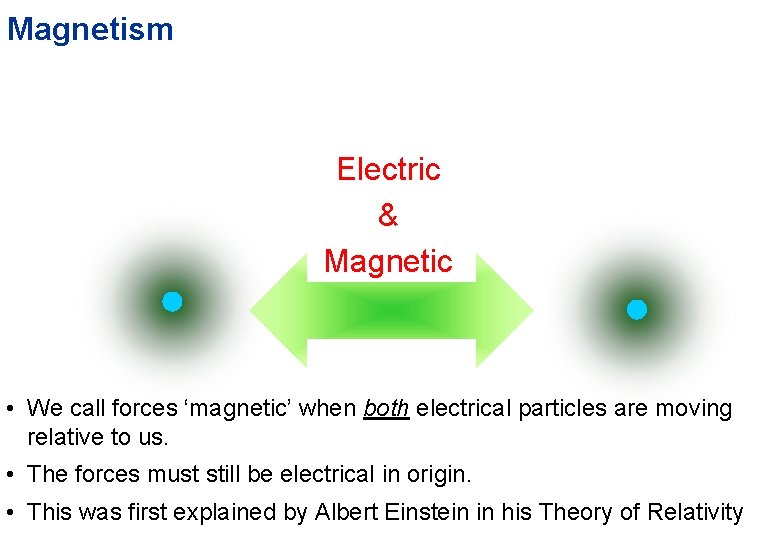
Magnetism Electric & Magnetic • We call forces ‘magnetic’ when both electrical particles are moving relative to us. • The forces must still be electrical in origin. • This was first explained by Albert Einstein in his Theory of Relativity

Winding up… • • • Mechanical Forces are electrical Chemistry is electrical Electronics is electrical Magnetism is electrical Sausages are electrical

The electrical nature of matter How the balloon affected the paper…
Odd phenomena… • A balloon and a piece of paper
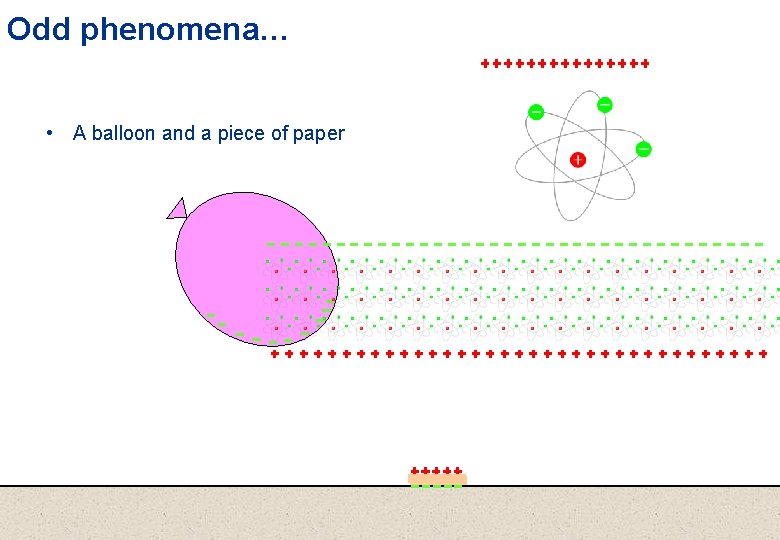
Odd phenomena… • A balloon and a piece of paper

Tonight’s talk • The scale and size of the Universe Its very big, but full of very small things • Electricity It’s everywhere! • How it works There are ‘particles’ and ‘fields’ • Atoms They’re everywhere! And they are all electrical! • Light

The electrical nature of matter How is the electrical force transmitted from one charged particle to another?
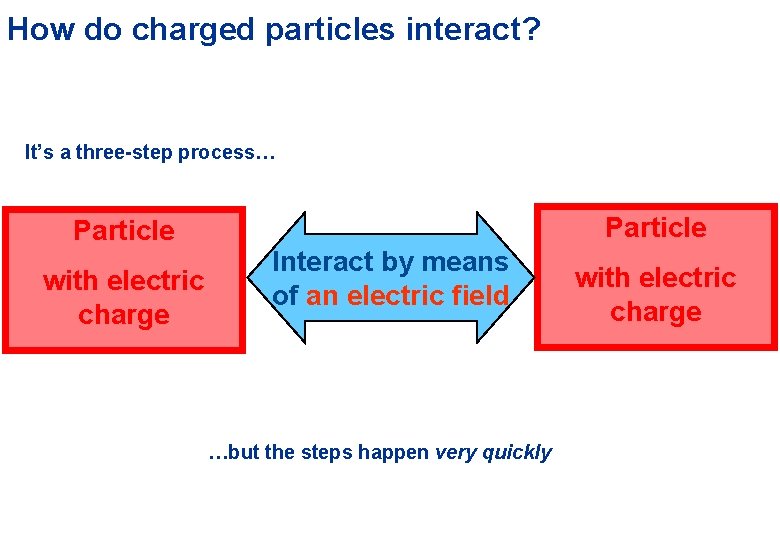
How do charged particles interact? It’s a three-step process… Particle with electric charge Particle Interact by means of an electric field …but the steps happen very quickly with electric charge

The nature of interactions (1) Analogy with water level and water waves

Tonight’s talk • The scale and size of the Universe Its very big, but full of very small things • Electricity It’s everywhere! • How it works There are ‘particles’ and ‘fields’ • Atoms They’re everywhere! And they are all electrical! • Light An electro-magnetic wave
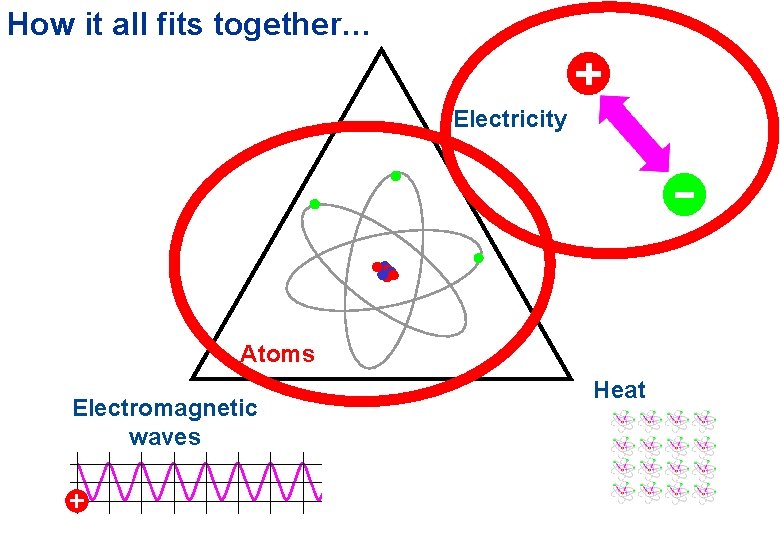
How it all fits together… Electricity Atoms Electromagnetic waves Heat

Summary • Physics concepts span vast ranges of mass, length and time. • The universe has two kinds of objects in it: Matter and Fields • All matter (on Earth) is made of atoms which interact electrically. • In matter as we normally experience it, there are equal amounts of the two types of electric charge and their effects cancel • If we add or remove some particles with electric charge from matter then we can see the electrical effects.

Homework • Activity: Remember when you have your breakfast that you are eating protons and neutrons coated with tasty electrons. • Research: What is the ‘frequency’ of your favourite radio station? Don’t just get the number (98. 9, 198 etc. ) get the units as well! They should be in Hertz Kilohertz Megahertz

One minute feedback • • • On the back of your handouts! Rip off the last sheet Please write down what is in on your mind RIGHT NOW! A question? OK A comment? OK A surprising thought in your mind? I’d love to hear it!

Goodnight • blog. protonsforbreakfast. org This Powerpoint File Links to other sites & resources Me going on about things See you next week! Don’t forget your pencils and badges!

Goodnight
- Slides: 98