PROTONPUMP INHIBITORS Protonpump inhibitors PPIs are a group

PROTON-PUMP INHIBITORS • Proton-pump inhibitors (PPIs) are a group of drugs whose main action is a pronounced and long-lasting reduction of gastric acid production. Proton pump inhibitors are used to: • Relieve symptoms of acid reflux, or gastroesophageal reflux disease (GERD). This is a condition in which food or liquid moves up from the stomach to the esophagus (the tube from the mouth to the stomach). • Treat a peptic or stomach ulcer. • Treat damage to the lower esophagus caused by acid reflux.

• Gastroesophageal reflux disease (GERD) is a condition in which the stomach contents leak backwards from the stomach into the esophagus • A peptic ulcer is an open sore or raw area in the lining of the stomach or intestine. • A gastric ulcer occurs in the stomach. • A duodenal ulcer occurs in the first part of the small intestine. • Available PPIs are omeprazole, lansoprazole, rabeprazole, pantoprazole, and esomeprazole are inactive prodrugs that are activated in the acid environment of the gastric glands.
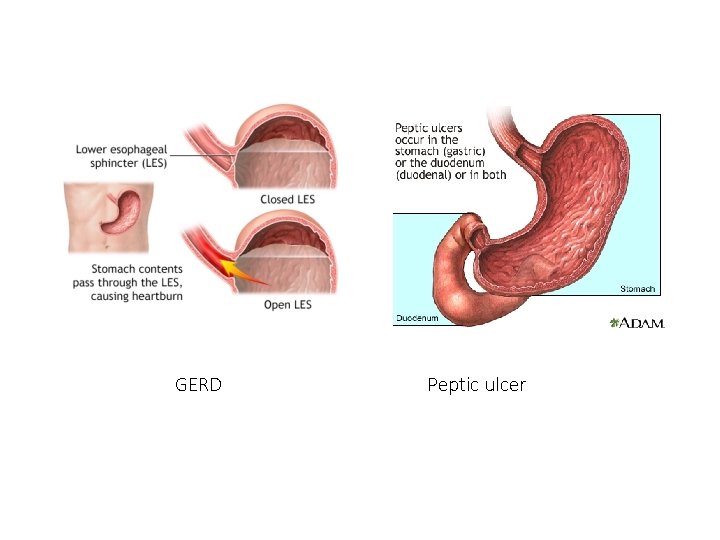
GERD Peptic ulcer
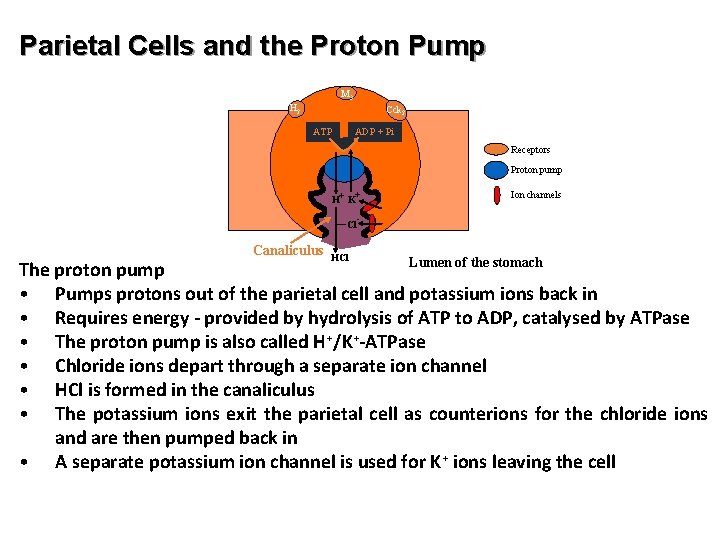
Parietal Cells and the Proton Pump M 3 H 2 Cck 2 ATP ADP + Pi Receptors Proton pump H+ K+ Cl Canaliculus HCl Ion channels - Lumen of the stomach The proton pump • Pumps protons out of the parietal cell and potassium ions back in • Requires energy - provided by hydrolysis of ATP to ADP, catalysed by ATPase • The proton pump is also called H+/K+-ATPase • Chloride ions depart through a separate ion channel • HCl is formed in the canaliculus • The potassium ions exit the parietal cell as counterions for the chloride ions and are then pumped back in • A separate potassium ion channel is used for K+ ions leaving the cell

• Mechanism of PPI: • PPIs inhibit gastric acid secretion by inhibiting the enzyme H+/K+ -ATPase, which is located on the luminal surface of gastric parietal cells. • When administered at neutral p. H, these weak bases are chemically stable, lipid-soluble, and have no effect on gastric acid secretion. • The inactive PPI diffuses from the bloodstream into the parietal cells and subsequently into the acid environment of the secretory canaliculi, where it rearranges to form a sulfenic acid in equilibrium with a sulfenamide. • This chemical entity is then able to interact covalently with thiol groups at cysteine residues located on the luminal surface of the a-subunit of the H+/K+-ATPase. • This covalent binding results in specific and essentially irreversible inactivation of the enzyme, leading to long-lasting inhibition of gastric acid secretion. Such a profile is thus ideally suited to once-daily administration in the management of acidpeptic disorders.
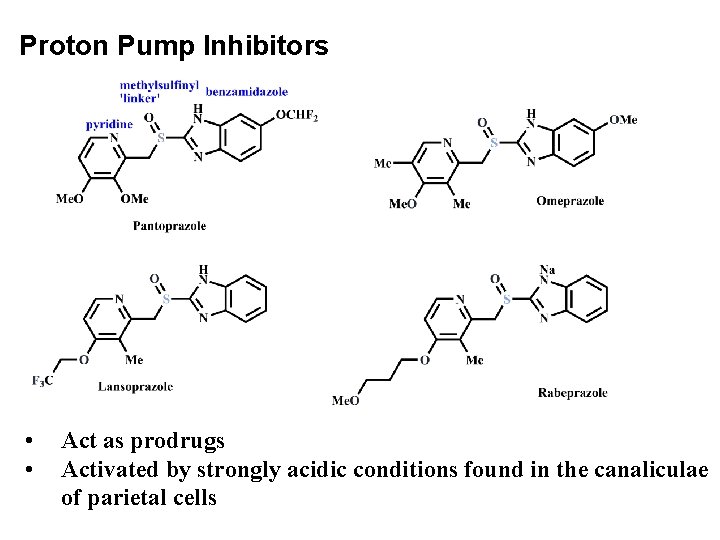
Proton Pump Inhibitors • • Act as prodrugs Activated by strongly acidic conditions found in the canaliculae of parietal cells
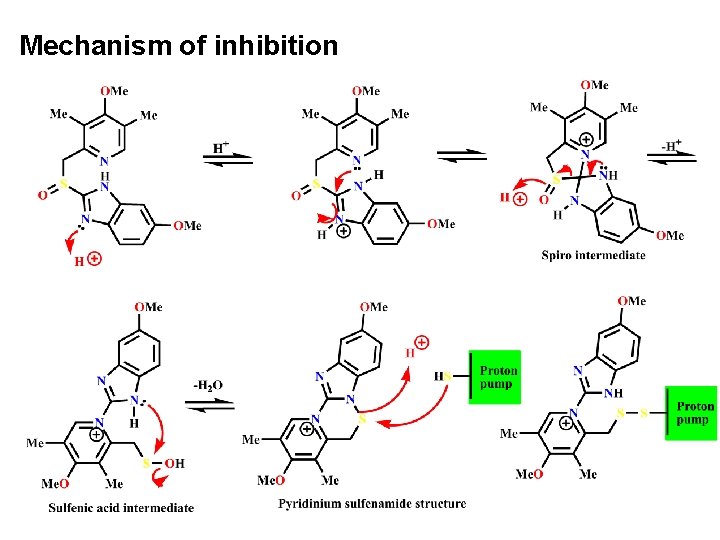
Mechanism of inhibition

Structure and SAR of the Proton-Pump Inhibitors • In 1973 workers in Sweden identified timoprazole (118) as one of the first well-defined inhibitors of the newly discovered gastric proton pump. This compound stemmed from efforts to separate the toxicity and acid-inhibitory properties of 2 -pyridylthioacteamide (CMN 131). CMN 131 • Removal of the thioamide group was considered to be the most likely solution to the toxicity of CMN 131, which prompted the preparation of sulfur containing heterocycles, as well as imidazoline and benzimidazole-linked sulfides.
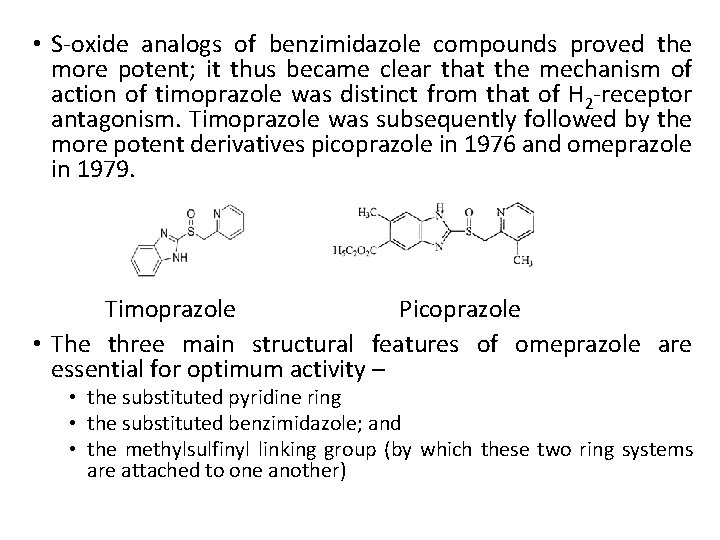
• S-oxide analogs of benzimidazole compounds proved the more potent; it thus became clear that the mechanism of action of timoprazole was distinct from that of H 2 -receptor antagonism. Timoprazole was subsequently followed by the more potent derivatives picoprazole in 1976 and omeprazole in 1979. Timoprazole Picoprazole • The three main structural features of omeprazole are essential for optimum activity – • the substituted pyridine ring • the substituted benzimidazole; and • the methylsulfinyl linking group (by which these two ring systems are attached to one another)
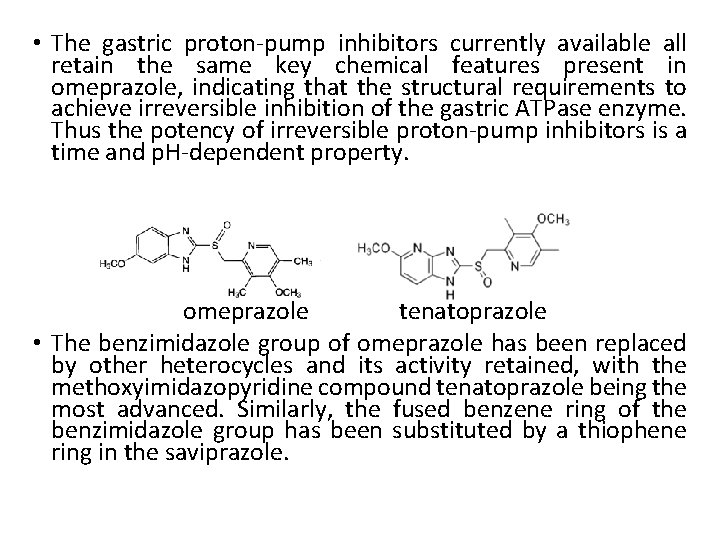
• The gastric proton-pump inhibitors currently available all retain the same key chemical features present in omeprazole, indicating that the structural requirements to achieve irreversible inhibition of the gastric ATPase enzyme. Thus the potency of irreversible proton-pump inhibitors is a time and p. H-dependent property. omeprazole tenatoprazole • The benzimidazole group of omeprazole has been replaced by other heterocycles and its activity retained, with the methoxyimidazopyridine compound tenatoprazole being the most advanced. Similarly, the fused benzene ring of the benzimidazole group has been substituted by a thiophene ring in the saviprazole.
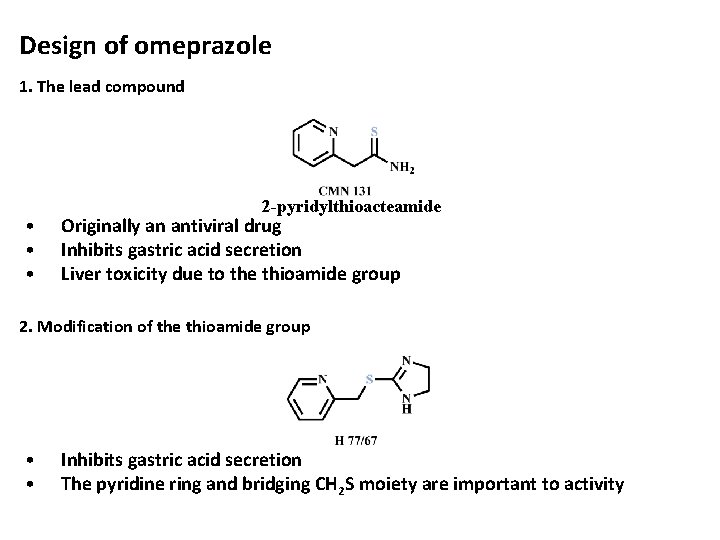
Design of omeprazole 1. The lead compound • • • 2 -pyridylthioacteamide Originally an antiviral drug Inhibits gastric acid secretion Liver toxicity due to the thioamide group 2. Modification of the thioamide group • • Inhibits gastric acid secretion The pyridine ring and bridging CH 2 S moiety are important to activity
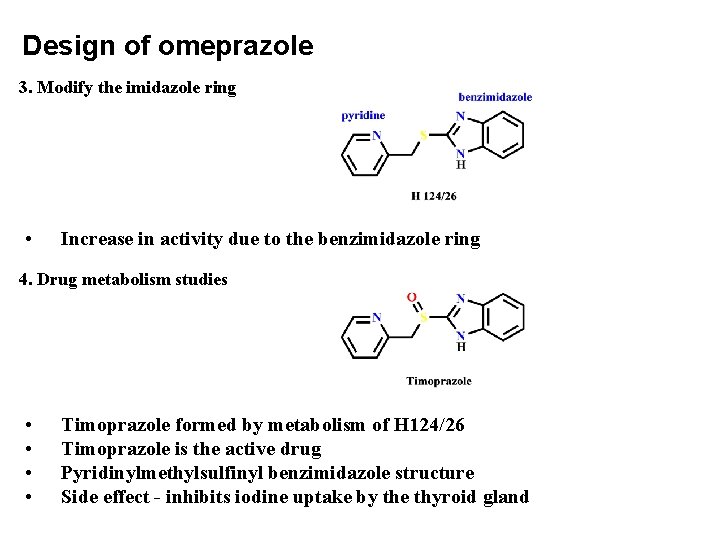
Design of omeprazole 3. Modify the imidazole ring • Increase in activity due to the benzimidazole ring 4. Drug metabolism studies • • Timoprazole formed by metabolism of H 124/26 Timoprazole is the active drug Pyridinylmethylsulfinyl benzimidazole structure Side effect - inhibits iodine uptake by the thyroid gland
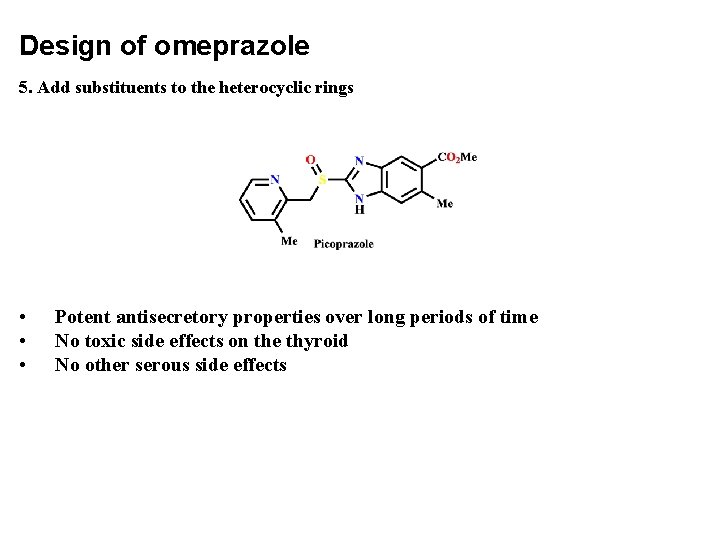
Design of omeprazole 5. Add substituents to the heterocyclic rings • • • Potent antisecretory properties over long periods of time No toxic side effects on the thyroid No other serous side effects
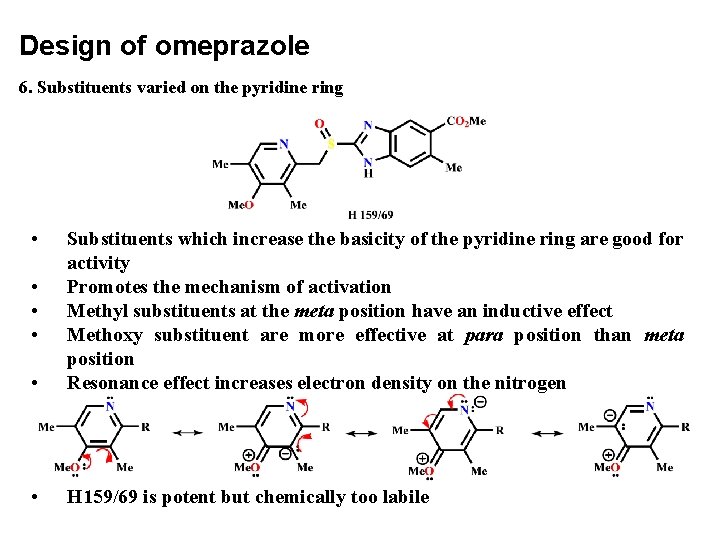
Design of omeprazole 6. Substituents varied on the pyridine ring • • Substituents which increase the basicity of the pyridine ring are good for activity Promotes the mechanism of activation Methyl substituents at the meta position have an inductive effect Methoxy substituent are more effective at para position than meta position Resonance effect increases electron density on the nitrogen • H 159/69 is potent but chemically too labile • • •
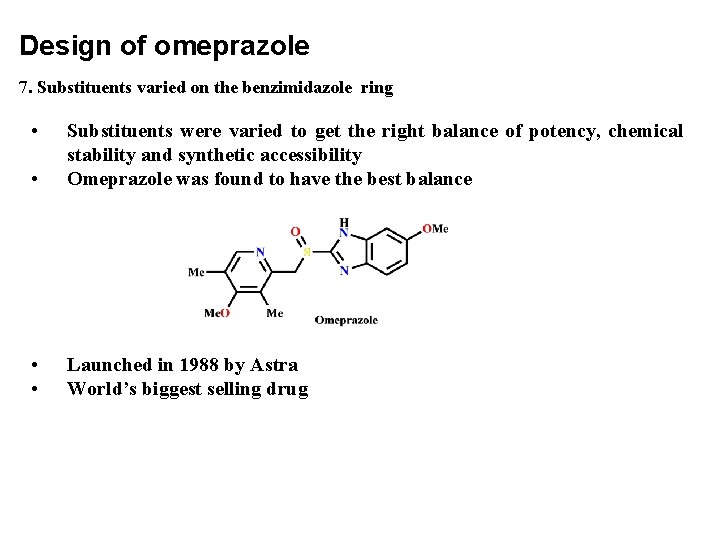
Design of omeprazole 7. Substituents varied on the benzimidazole ring • • Substituents were varied to get the right balance of potency, chemical stability and synthetic accessibility Omeprazole was found to have the best balance • • Launched in 1988 by Astra World’s biggest selling drug
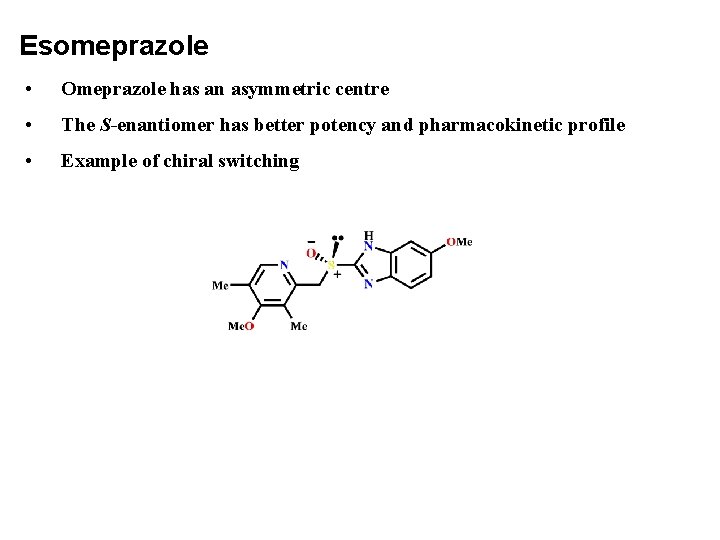
Esomeprazole • Omeprazole has an asymmetric centre • The S-enantiomer has better potency and pharmacokinetic profile • Example of chiral switching

Esomeprazole • Omeprazole showed an inter-individual variability and therefore a significant number of patients with acidrelated disorders required higher or multiple doses to achieve symptom relief and healing. • Astra started a new research program in 1987 to identify a new analogue to omeprazole with less interpatient variability. • Only one compound proved superior to omeprazole and that was the (S)-(−)-isomer, esomeprazole, which was developed. • Esomeprazole magnesium received its first approval in 2000 and provided more pronounced inhibition of acid secretion and less inter-patient variation compared to omeprazole.
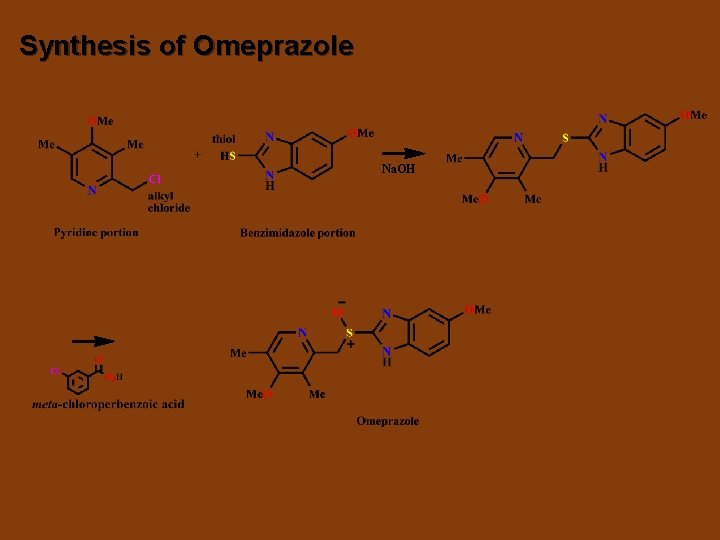
Synthesis of Omeprazole Na. OH

Pharmacodynamics § They are the most potent inhibitors of acid secretion available today. § Produce marked inhibition of basal & meal stimulatedacid secretion (90 -98%). § Reduce pepsin activity. § Promote mucosal healing & decrease pain. § Proton pump inhibitors heal faster the ulcers than H 2 blockers, and have H. pylori inhibitory properties.

Pharmacokinetics § Given orally § Are pro-drugs § rapidly absorbed from the intestine. § Activated in the acidic medium of parietal cell canaliculi. § Inactivated if at neutral p. H. § Should not combined with H 2 blockers or antacids. § Have long duration of action (> 12 h-24 h). § Once daily dose is sufficient § Given 1 h before meal. § Bioavailability is reduced by food. § metabolized in the liver by CYP-450. § Dose reduction is required in severe liver failure.

USES ØEradication of H. pylori (combined with antimicrobial drugs). Ø Peptic ulcer ( 4 -8 weeks) resistant to H 2 antagonists. Ø Reflux esophagitis. ØHypersecretory conditions as Zollinger Ellison syndrome (drug of choice).
- Slides: 21