Protocol Nichol Mc Bee MPH CCRP BIOS Coordinating

Protocol Nichol Mc. Bee, MPH, CCRP BIOS Coordinating Center Johns Hopkins University

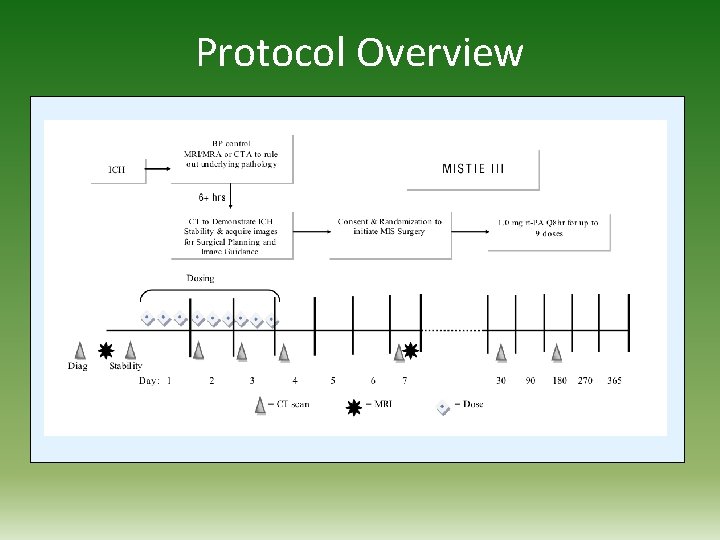
Protocol Overview
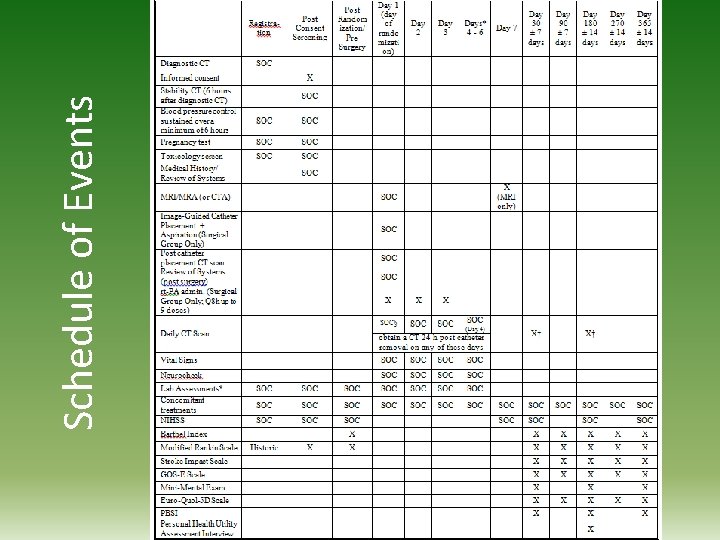
Schedule of Events

Registration/Screening • • • Diagnostic CT Blood pressure control Pregnancy test Toxicology screen Lab assessments Concomitant treatments NIHSS Barthel Index (historic) Modified Rankin scale (historic)

Early Notification is Important I t n i CH ! D E he

Diagnostic CT • • • Within 24 hours of symptom onset Time 0 CT or CTA ICH > 30 cc Infratentorial hemorrhage – Brainstem involvement or third nerve palsy • No underlying pathology
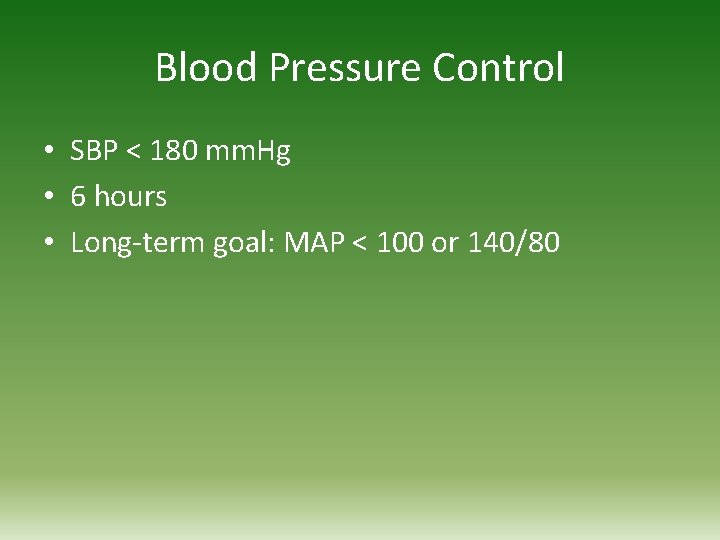
Blood Pressure Control • SBP < 180 mm. Hg • 6 hours • Long-term goal: MAP < 100 or 140/80

Pregnancy Test • Urine or serum • Prior to consent: Review if done standard of care • Post consent: Order if not done per standard of care • Pregnancy during follow-up

Toxicology Screen • • • Cocaine, barbiturates, etc Contributor(s) to the bleed As close to presentation to ED as possible Standard of care? Data collection only

Lab Assessments • Screening: – Plt count – INR – Pregnancy test (if applicable) • Once prior to randomization: – Plasma plasminogen activity – Fibrinogen • Daily through day 6: – WBC – Hct – Plt count – a. PTT – INR

Concomitant Treatments • Concomitant medications • Concomitant procedures

NIHSS • ED presentation • Certified examiner • Day 7, 30, 180, and 365

Barthel Index • Screening: historical • Level of functioning prior to symptom onset • Comparison of scores at days 30, 90, 180, 270, and 365

Modified Rankin Scale • • Screening: historical 0 -1 Level of functioning prior to symptom onset Comparison of scores at days 30, 90, 180, 270, and 365

Post Consent Screening • • • Medical history/review Informed consent of systems Stability CT Blood pressure control • MRI/MRA (or CTA) Pregnancy test Toxicology screen Lab assessments Concomitant treatments NIHSS Modified Rankin Scale

Inclusion Criteria • • • ICH ≥ 30 m. L on d. CT GCS ≤ 14 or a NIHSS ≥ 6 ICH stability Symptom onset < 24 h prior to d. CT MIS 12 -72 h post d. CT 1 st dose up to 76 h post d. CT SBP < 180 mm. Hg Historical m. RS 0 or 1 Age ≥ 18 and ≤ 80

Exclusion Criteria • • • Infratentorial hemorrhage Intraventricular hemorrhage requiring EVD Thalamic bleeds with midbrain extension Irreversible impaired brain stem function Underlying pathology Unstable mass or evolving intracranial compartment syndrome

Exclusion Criteria (cont. ) • • • Coagulopathy Long-term anti-coagulation required Dabigatran use Systemic bleeding Pregnancy Allergy/sensitivity to rt-PA Prior enrollment Other interventional trial Survival to d 365 is not expected

Exclusion Criteria (cont. ) Concurrent illness Mechanical heart valve Known risk for embolization Investigator judgment Active drug or alcohol use or dependence that would interfere with follow-up • Unstable patient who needs specific intervention • No consent • • •

Randomization • Only after I/E criteria satisfied and informed consent signed • Adaptive design • Timelines

Post-Randomization/Pre-Surgery • • • Lab assessments Concomitant treatments NIHSS Barthel Index Modified Rankin Scale MRI/MRA

Day 1 (Day of Randomization) • • • Medical management Image-guided catheter placement + aspiration Post catheter placement CT Review of systems (post surgery) rt-PA administration Daily CT Vital signs Neurocheck Lab assessments Concomitant treatments

Medical Management • • ICP management Neurological status Cardiovascular management Respiratory care Nutritional support DVT and PE prophylaxis Withdrawal of care

MIS Procedure • • Credentialed neurosurgeon Trajectory determination Surgical Center review Antibiotic therapy Catheter placement Post catheter placement CT Stabilization period Catheter adjustment/replacement

Review of Systems Post Surgery • Neurological status • 3 hours post MIS • Clinical worsening or improvement

DOSING • Who – Certified investigator or designee • What – 1. 0 mg/1. 0 m. L of Cathflo followed by 3. 0 m. L of flush • Where – At the bedside – Into the clot system

DOSING • When – 1 st dose no sooner than 12 hours and no later than 76 hours post Diagnostic CT scan and after stability is confirmed. – Every 8 hours for up to 9 doses • How – Sterile field – Monitor vital signs

DOSING ELIGIBILITY • Daily CT scans • Catheter tract stability • Dosing endpoints

Days 2 and 3 (Post Randomization) • • • rt-PA administration Daily CT Vital signs Neurocheck Lab assessments Concomitant treatments

DAILY SAFETY LABS • Serum – White blood cell count – Hematocrit – Platelet count – INR – PT – a. PTT – Plasminogen – Fibrinogen – D-dimer

Days 4 -6 (Post Randomization) • • • Daily CT (day 4) CT 24 h post catheter removal Vital signs Neurocheck Lab assessments Concomitant treatments

Day 7 (Post Randomization) • MRI • NIHSS
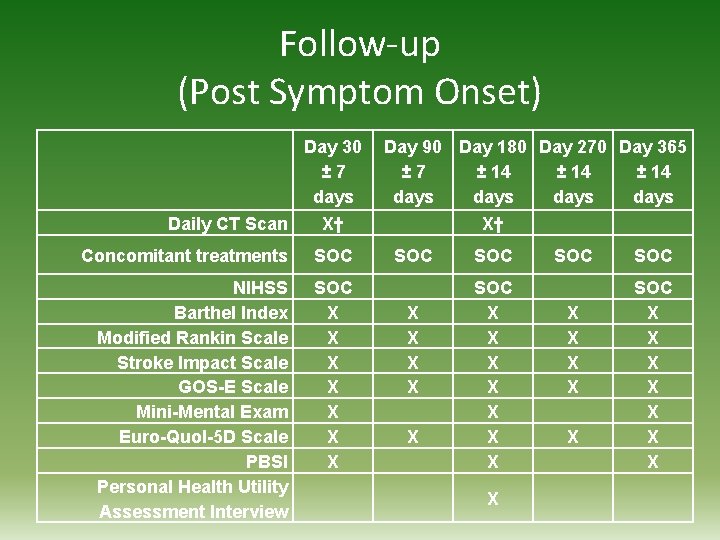
Follow-up (Post Symptom Onset) Day 30 ± 7 days Daily CT Scan Day 90 Day 180 Day 270 Day 365 ± 7 ± 14 days X† X† Concomitant treatments SOC SOC SOC NIHSS Barthel Index Modified Rankin Scale Stroke Impact Scale GOS-E Scale Mini-Mental Exam Euro-Quol-5 D Scale PBSI Personal Health Utility Assessment Interview SOC X X X X X X X X X SOC X X X X X

RESOURCE UTILIZATION • ICU stay • Hospital stay • Time at home

CONCOMITANT MEDICATIONS • • • 7 days prior to symptom onset Acute treatment phase Follow-up phase Required interventions Prohibited interventions Precautionary interventions

AE & SAE REPORTING • All AEs and SAEs through day 7 • All SAEs and neurological AEs through day 365 • SAEs must be reported using the EDC system – No paper worksheet!

FINAL VISIT • • Day 365 follow-up visit CT scan Outcome Scales Record new AE/SAE or procedures – Neurosurgical procedures • Confirm ongoing or document resolution of previously recorded events

- Slides: 39