Proteins Proteins Atoms carbon hydrogen oxygen nitrogen Monomer

Proteins
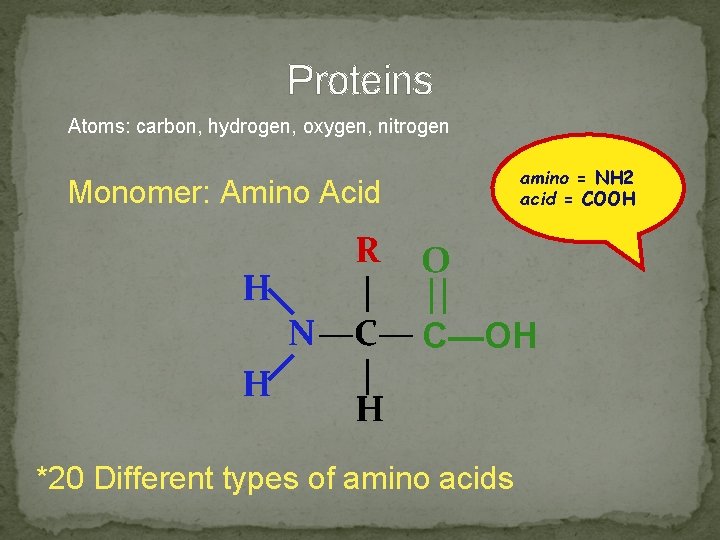
Proteins Atoms: carbon, hydrogen, oxygen, nitrogen Monomer: Amino Acid amino = NH 2 acid = COOH R O H | || N —C— C—OH | H H *20 Different types of amino acids
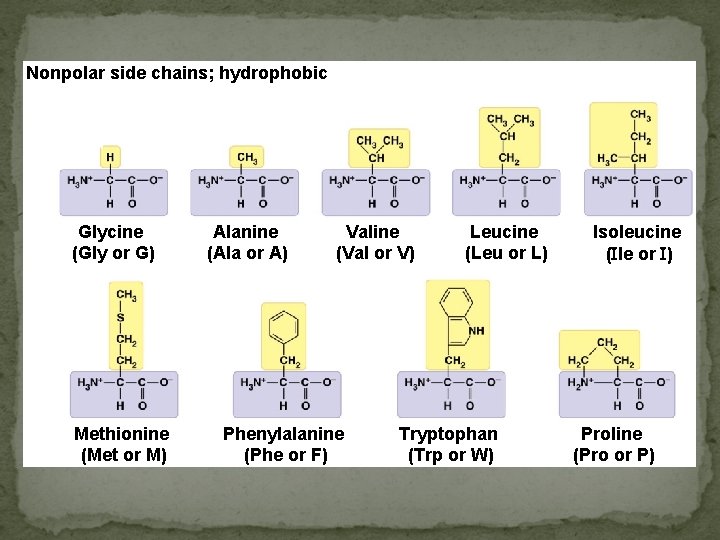
Nonpolar side chains; hydrophobic ) Glycine (Gly or G) Methionine (Met or M) Alanine (Ala or A) Valine (Val or V) Phenylalanine (Phe or F) Leucine (Leu or L) Tryptophan (Trp or W) Isoleucine ( le or ) Proline (Pro or P)
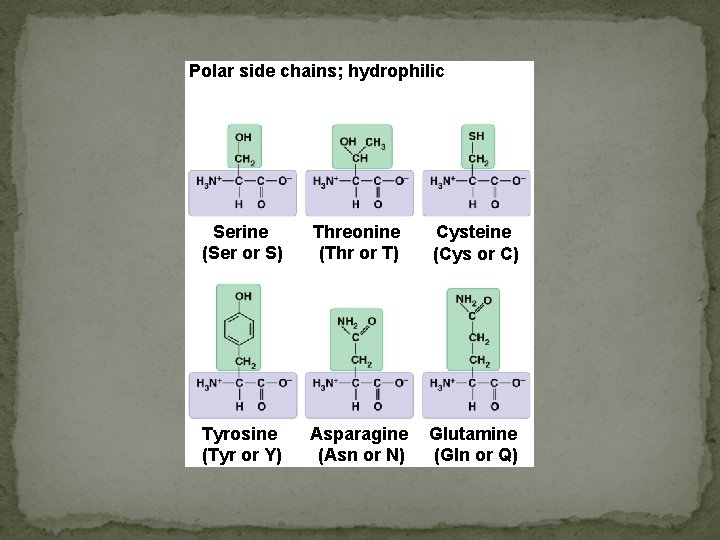
Polar side chains; hydrophilic Serine (Ser or S) Threonine (Thr or T) Cysteine (Cys or C) Tyrosine (Tyr or Y) Asparagine (Asn or N) Glutamine (Gln or Q)
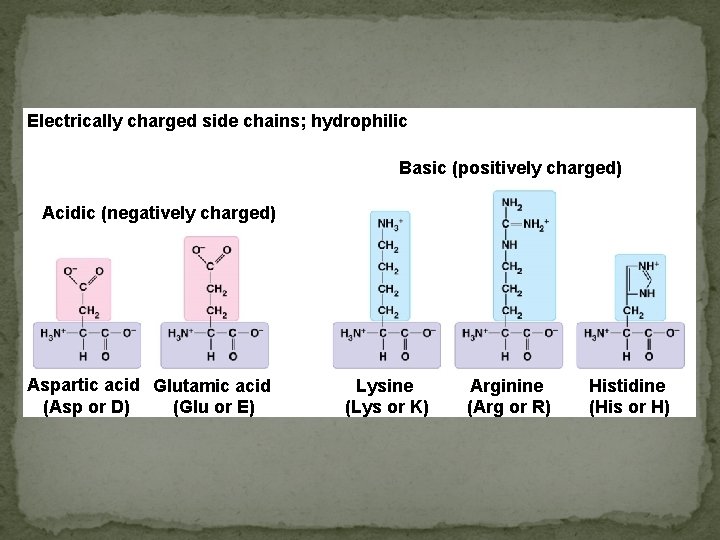
Electrically charged side chains; hydrophilic Basic (positively charged) Acidic (negatively charged) Aspartic acid Glutamic acid (Asp or D) (Glu or E) Lysine (Lys or K) Arginine (Arg or R) Histidine (His or H)
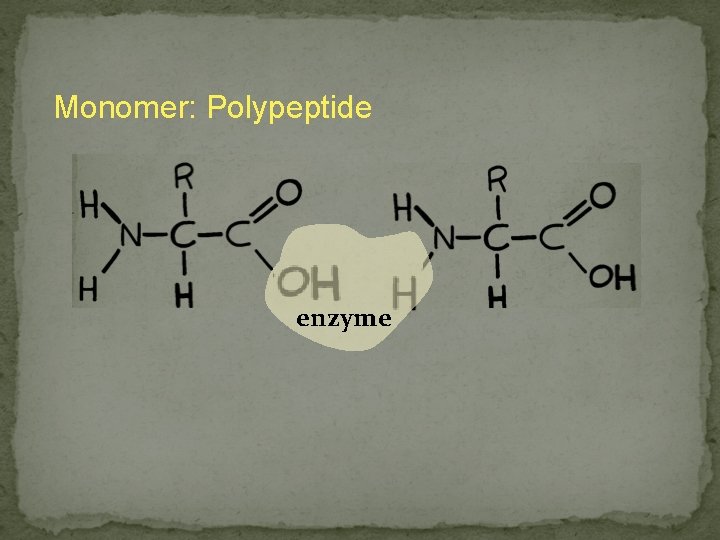
Monomer: Polypeptide enzyme
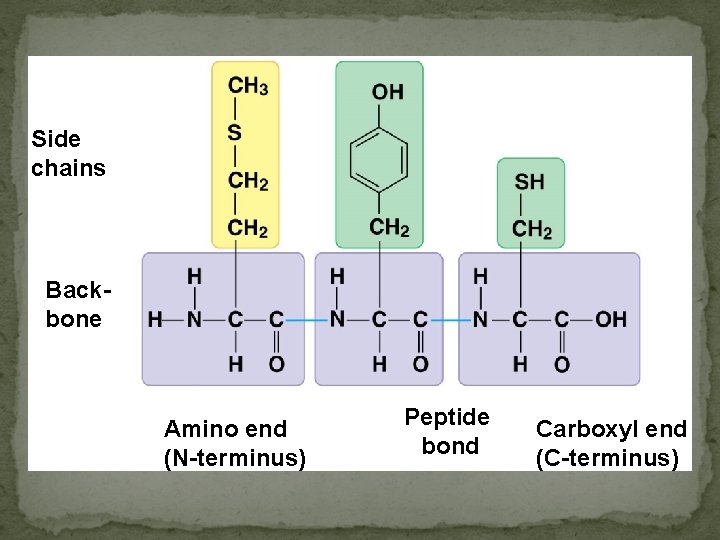
Side chains Backbone Amino end (N-terminus) Peptide bond Carboxyl end (C-terminus)

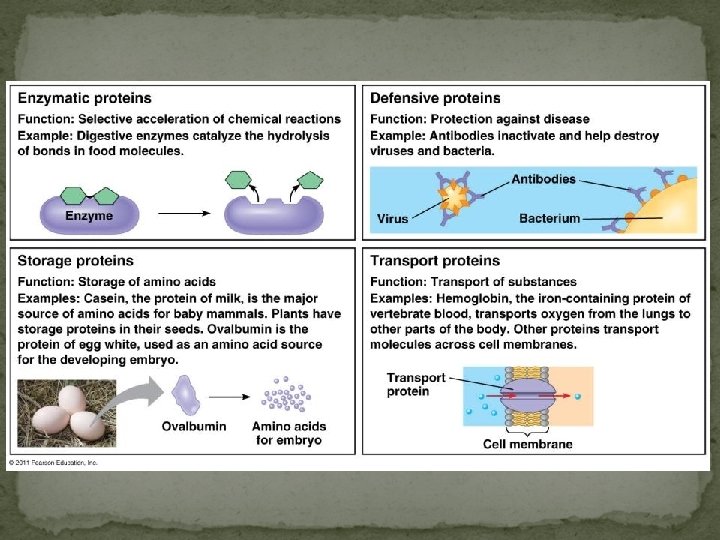
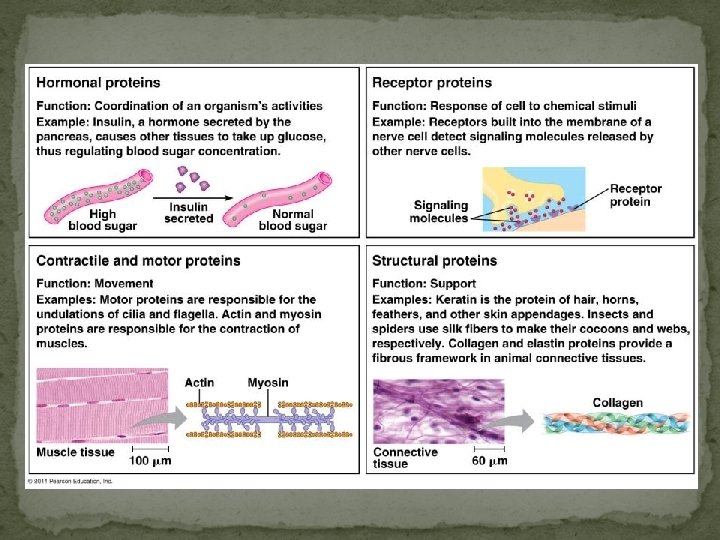
Proteins Most structurally & functionally diverse group Function: involved in almost everything that goes on in a living thing! (50% dry mass of cells enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (hemoglobin, aquaporin) cell communication signals (insulin & other hormones) receptors defense (antibodies) movement (actin & myosin) storage (bean seed proteins)
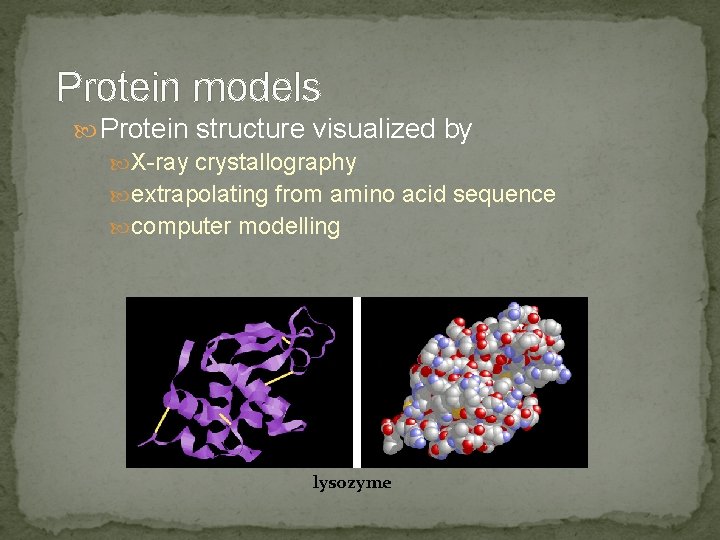
Protein models Protein structure visualized by X-ray crystallography extrapolating from amino acid sequence computer modelling lysozyme
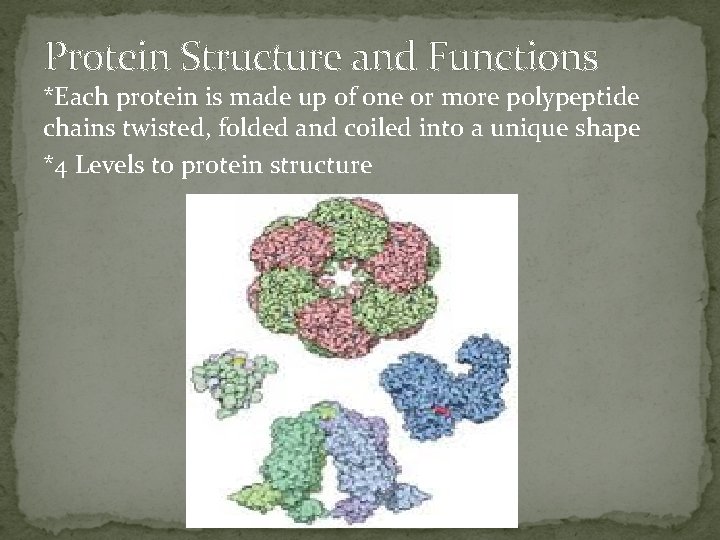
Protein Structure and Functions *Each protein is made up of one or more polypeptide chains twisted, folded and coiled into a unique shape *4 Levels to protein structure
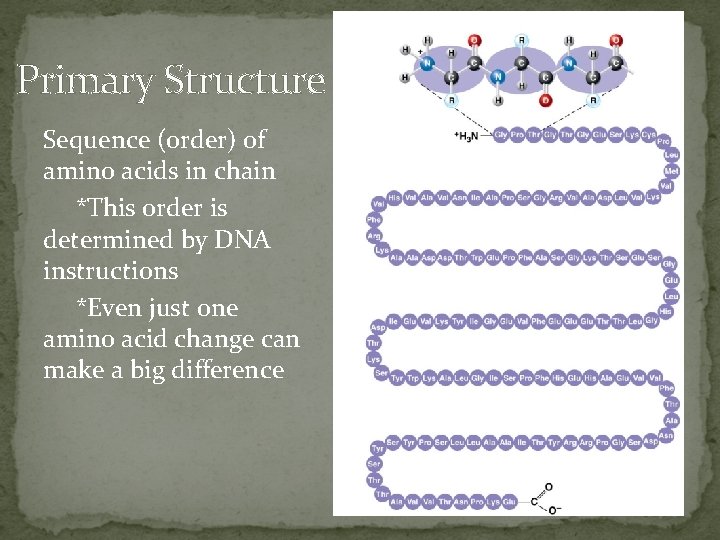
Primary Structure Sequence (order) of amino acids in chain *This order is determined by DNA instructions *Even just one amino acid change can make a big difference
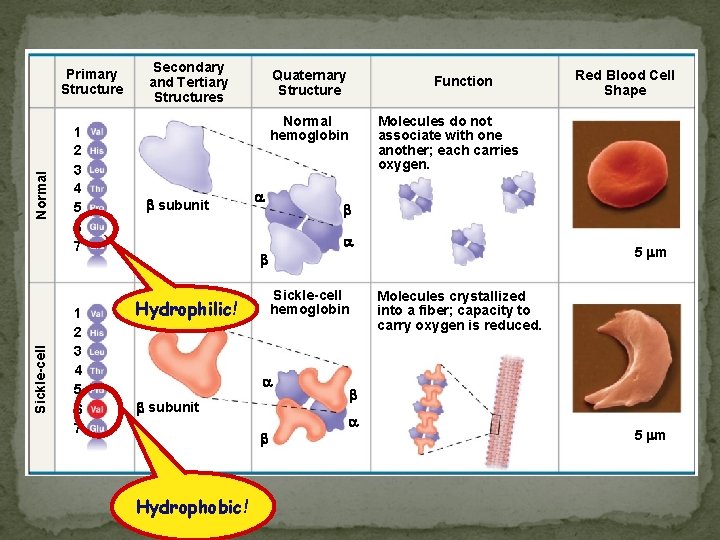
Sickle-cell Normal Primary Structure 1 2 3 4 5 6 7 Secondary and Tertiary Structures Quaternary Structure Function Normal hemoglobin subunit Molecules do not associate with one another; each carries oxygen. 5 m Exposed hydrophobic region Hydrophilic! Sickle-cell hemoglobin subunit Hydrophobic! Red Blood Cell Shape Molecules crystallized into a fiber; capacity to carry oxygen is reduced. 5 m
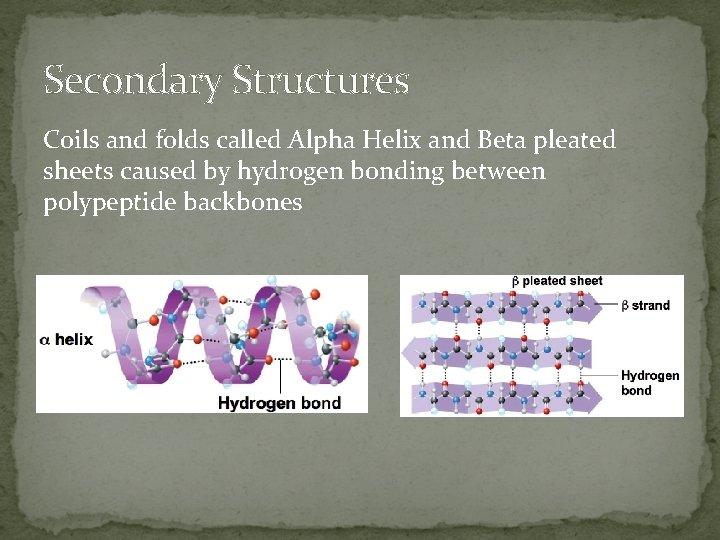
Secondary Structures Coils and folds called Alpha Helix and Beta pleated sheets caused by hydrogen bonding between polypeptide backbones
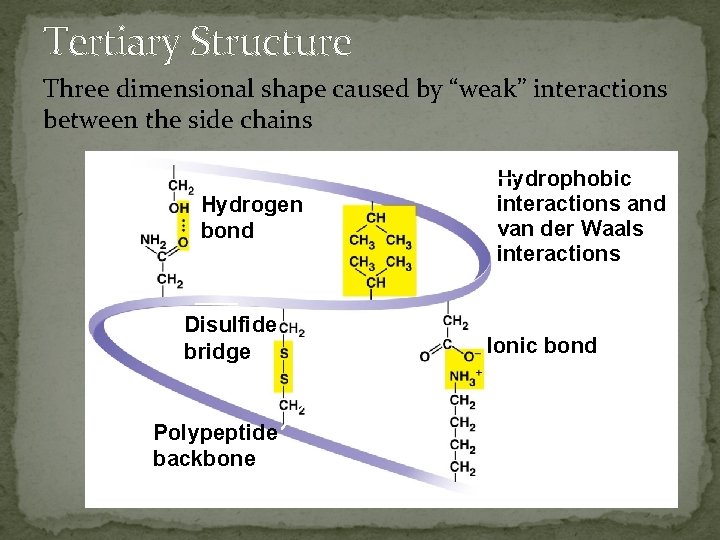
Tertiary Structure Three dimensional shape caused by “weak” interactions between the side chains Hydrogen bond Disulfide bridge Polypeptide backbone Hydrophobic interactions and van der Waals interactions Ionic bond
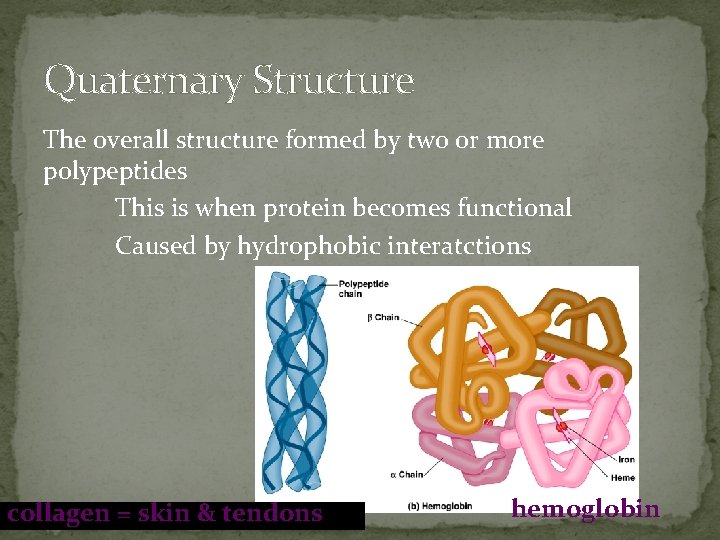
Quaternary Structure The overall structure formed by two or more polypeptides This is when protein becomes functional Caused by hydrophobic interatctions collagen = skin & tendons hemoglobin
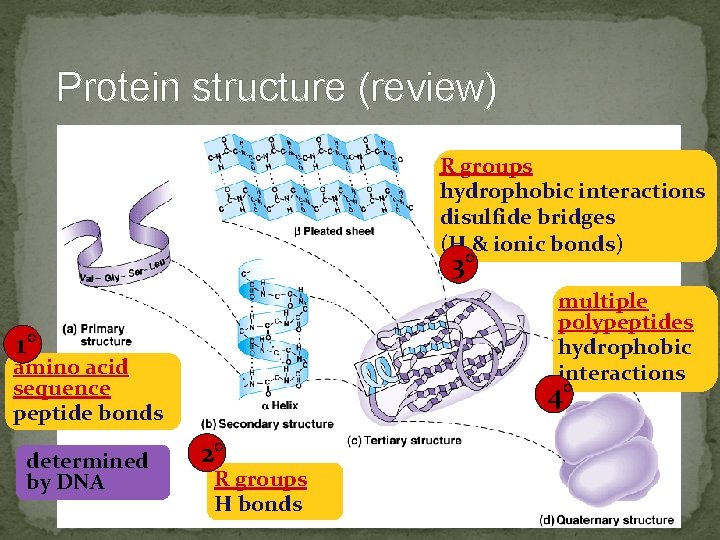
Protein structure (review) R groups hydrophobic interactions disulfide bridges (H & ionic bonds) 3° multiple polypeptides hydrophobic interactions 1° amino acid sequence peptide bonds determined by DNA 4° 2° R groups H bonds
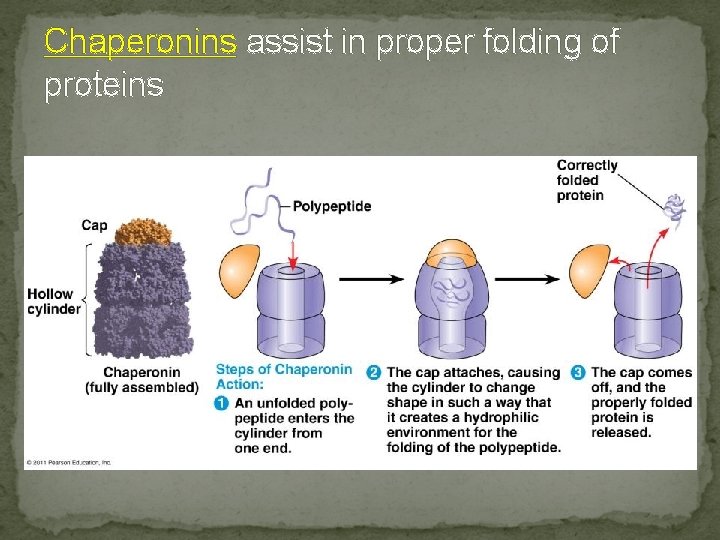
Chaperonins assist in proper folding of proteins

Protein denaturation Denaturation is the unfolding of a protein Conditions that disrupt H bonds, ionic bonds, disulfide bridges *Temperature *p. H *salinity Result in changes in secondary and tertiary structures Wrong shape = no function
- Slides: 20