Proteins Protein Basics leucine valine glycine alanine leucine
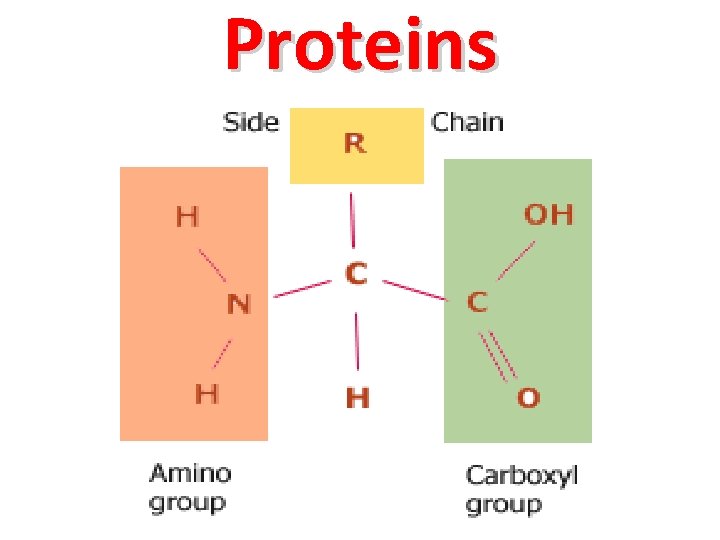
Proteins

Protein Basics leucine valine glycine alanine leucine histidine asparagine serine proline = Protein A valine leucine glycine alanine leucine histidine asparagine serine proline = Protein B leucine valine = Protein C Amino acids (monomer) glycine alanine leucine • Used in variety of cellular functions • Made of smaller amino acids – Monomer: Amino acid – Polymer: Polypeptide (Protein) • Only 20 amino acids… but thousands of proteins – Exact arrangement of amino acids determines the protein
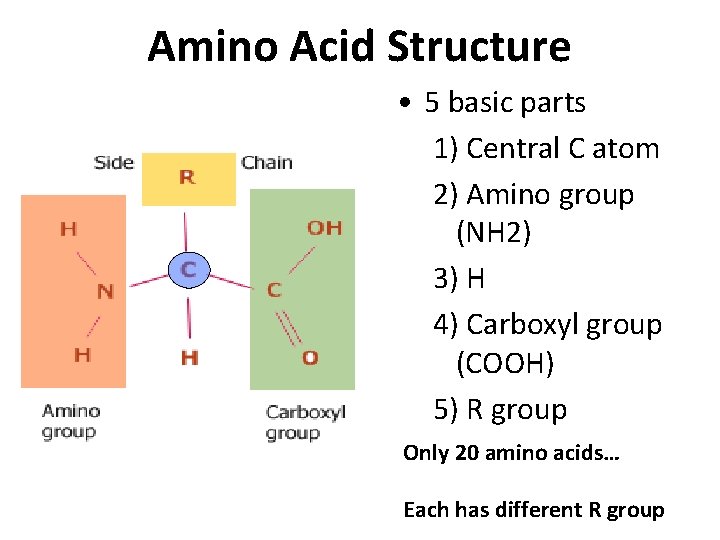
Amino Acid Structure • 5 basic parts 1) Central C atom 2) Amino group (NH 2) 3) H 4) Carboxyl group (COOH) 5) R group Only 20 amino acids… Each has different R group
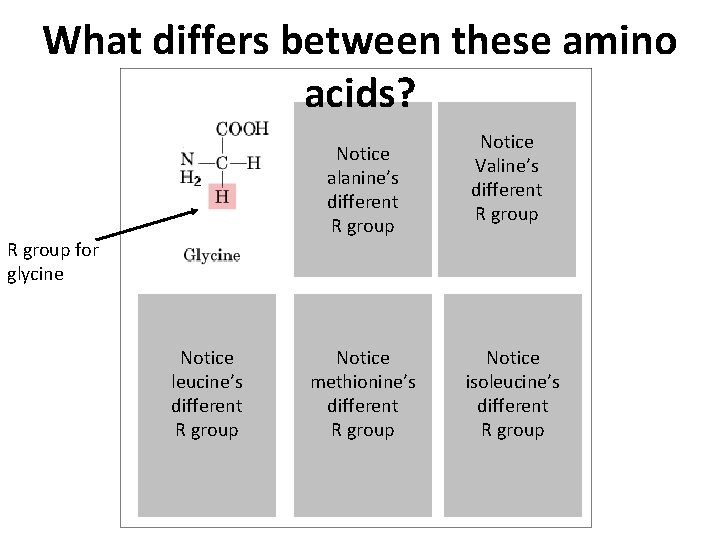
What differs between these amino acids? Notice alanine’s different R group Notice Valine’s different R group for glycine Notice leucine’s different R group Notice methionine’s different R group Notice isoleucine’s different R group
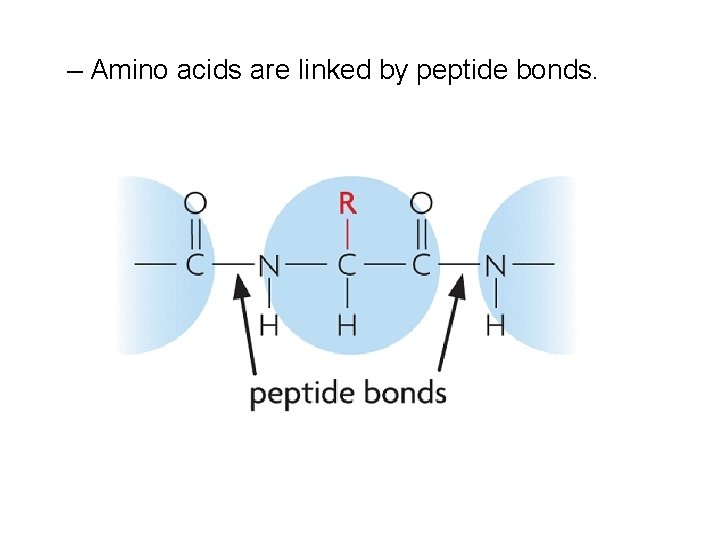
– Amino acids are linked by peptide bonds.
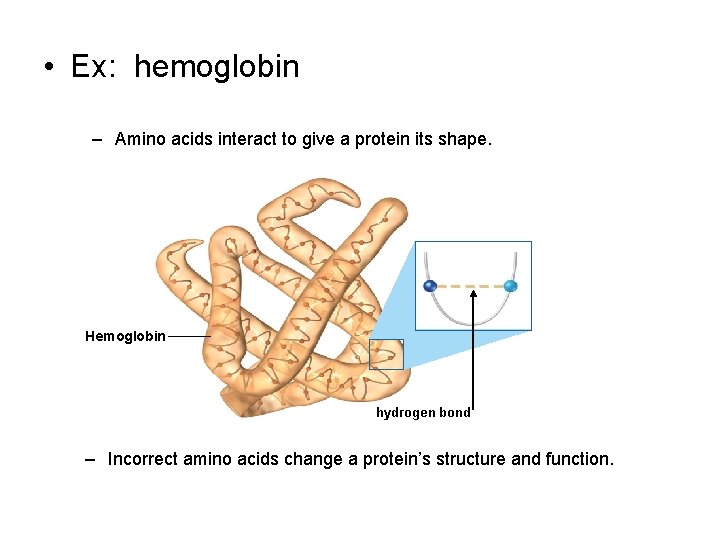
• Ex: hemoglobin – Amino acids interact to give a protein its shape. Hemoglobin hydrogen bond – Incorrect amino acids change a protein’s structure and function.
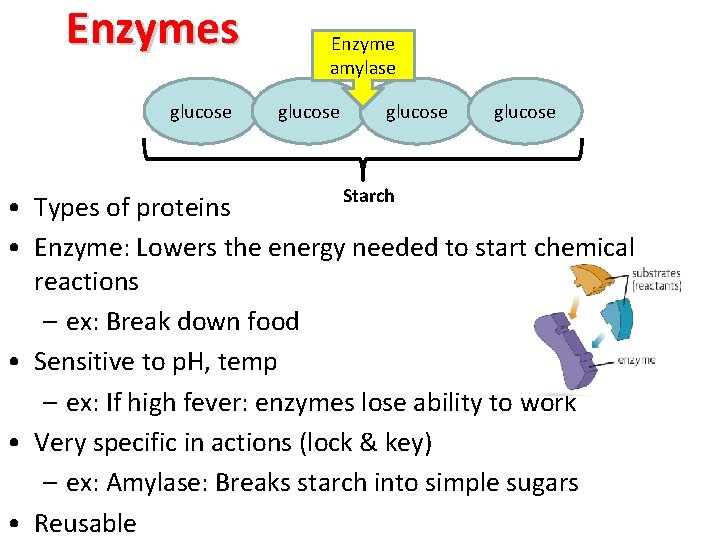
Enzymes glucose Enzyme amylase glucose Starch • Types of proteins • Enzyme: Lowers the energy needed to start chemical reactions – ex: Break down food • Sensitive to p. H, temp – ex: If high fever: enzymes lose ability to work • Very specific in actions (lock & key) – ex: Amylase: Breaks starch into simple sugars • Reusable
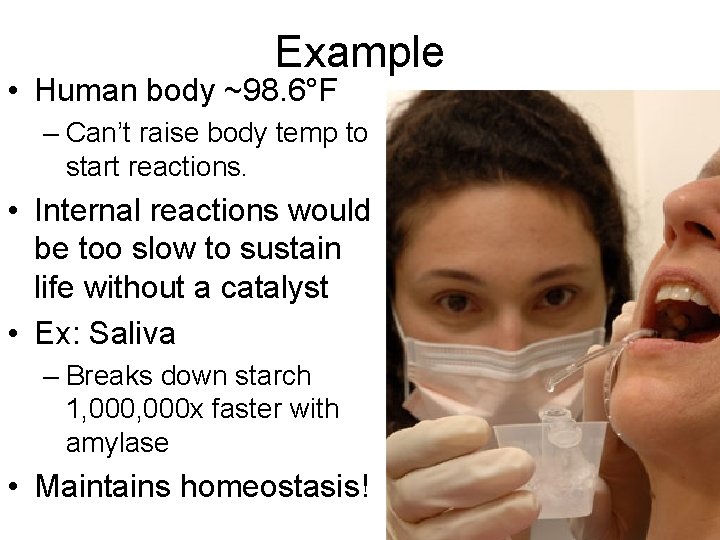
Example • Human body ~98. 6°F – Can’t raise body temp to start reactions. • Internal reactions would be too slow to sustain life without a catalyst • Ex: Saliva – Breaks down starch 1, 000 x faster with amylase • Maintains homeostasis!

Recap 1) What are the smaller monomers that make proteins called? 2) How many different amino acids exist? 3) How does each amino acid differ? 4) The NH 2 part of the amino acid is called the ____ group. 5) The COOH part of the amino acid is called the ____ group. 6) Which group of proteins help to start chemical reactions? 7) What can cause an enzyme to denature? 8) Explain the lock and key analogy as it relates to enzymes.
- Slides: 9