Proteins in Food BY DR GHULAM ABBAS PROTEINS

Proteins in Food BY DR. GHULAM ABBAS

PROTEINS • Proteins from Greek proteios meaning first, are a class of organic compounds which are present in and vital to every living cell. • In the form of skin , hair, callus, cartilage, muscles, tendons and ligaments , protein hold together , protect , and provide structure to the body of a multi-celled organism. • Proteins are generally beneficial, and are a necessary part of the diet of all animals. • Humans can become seriously ill if they do not eat enough suitable proteins, the disease kwashiorkor being an extreme from of protein deficiency.

Function of Proteins • Antibodies which are made of proteins , help to resist diseases and infections. we warm and protect our bodies with clothing and shoes that are often protein in nature (e. g. wool, silk and leather). • Each day our body losses protein in the form of hair, skin, and nails, We also use up proteins in all the activities of running and maintaining our body. • If you eat protein daily , it is supplied to your body tissue to replenish any loss and repair • any injury. • For growing infants, children, and teens, protein along with sufficient calories is necessary for growth of the entire body.

Function of Proteins • Pregnant women need adequate protein for their fetus, maternal tissues support, and for milk production after delivery. • If you have a cut, undergo surgery, or have an injury or illness, you need proteins to recover and to rebuilt your body.

Amino acids • Proteins are natural polymers molecules consisting of amino acid units. • Number of amino acid in proteins may range from fifty to several thousands. • Natural alpha- amino acid: Hydrolysis of proteins by boiling with aqueous acid or base yields a mixture of small molecules identified as α- amino carboxylic acid. • More than twenty such components have been isolated and most common are listed in the table. General structure Natural alpha- amino acid
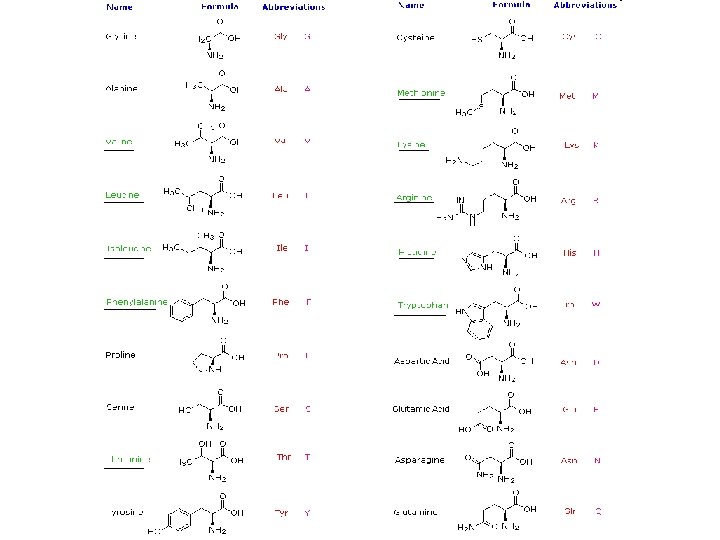

Amino Acid Table • Those amino acids having their names underlined are essential diet components, since they are not synthesized by human metabolic processes. • The best food source of these nutrients is proteins, but it is important to recognize that not all proteins have equal nutritional value. • For example, peanuts have a higher weight contents of protein than fish or eggs, but the proportion of essential amino acids in peanut protein is only one third of that from the two other sources. It will become evident when discussing the structures of these proteins and peptides.
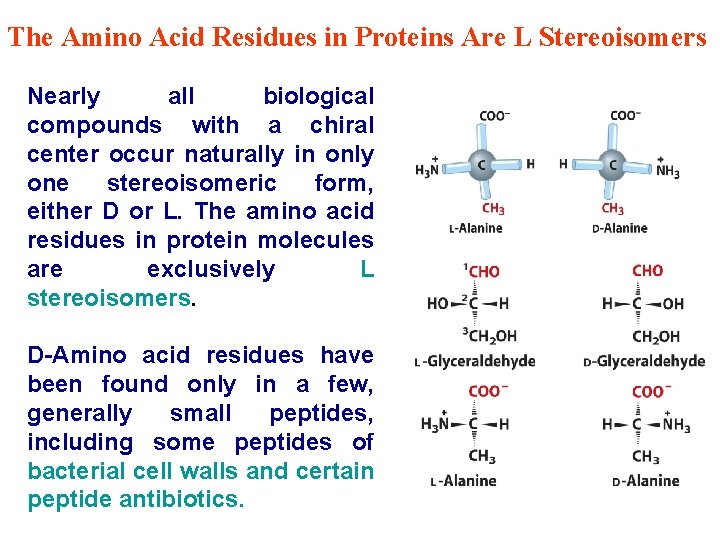
The Amino Acid Residues in Proteins Are L Stereoisomers Nearly all biological compounds with a chiral center occur naturally in only one stereoisomeric form, either D or L. The amino acid residues in protein molecules are exclusively L stereoisomers. D-Amino acid residues have been found only in a few, generally small peptides, including some peptides of bacterial cell walls and certain peptide antibiotics.

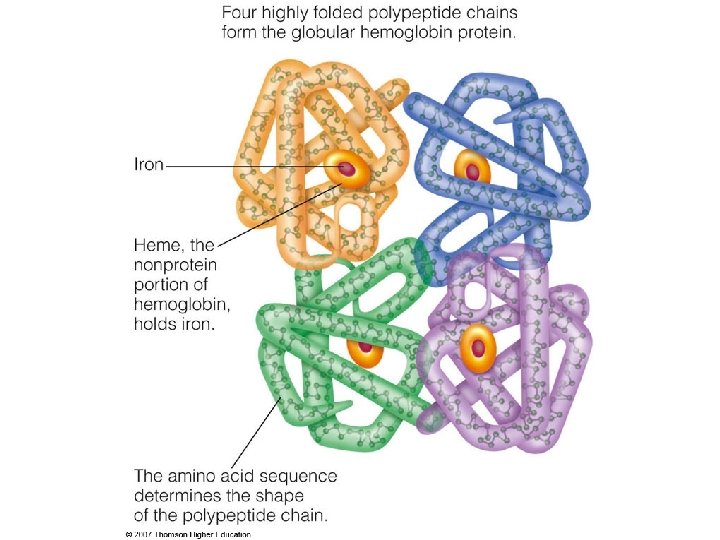
There Are Several Levels of Protein Structure The primary structure consists of a sequence of amino acids linked together by peptide bonds and includes disulfide bonds. The resulting polypeptide can be coiled into units of secondary structure, such as an helix. The helix is a part of the tertiary structure of the folded polypeptide, which is itself one of the subunits that make up the quaternary structure of the multisubunit protein, in this case hemoglobin.
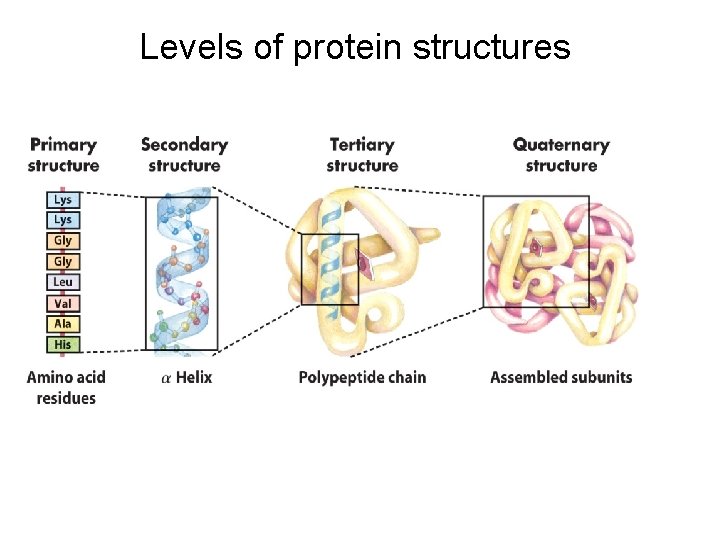
Levels of protein structures
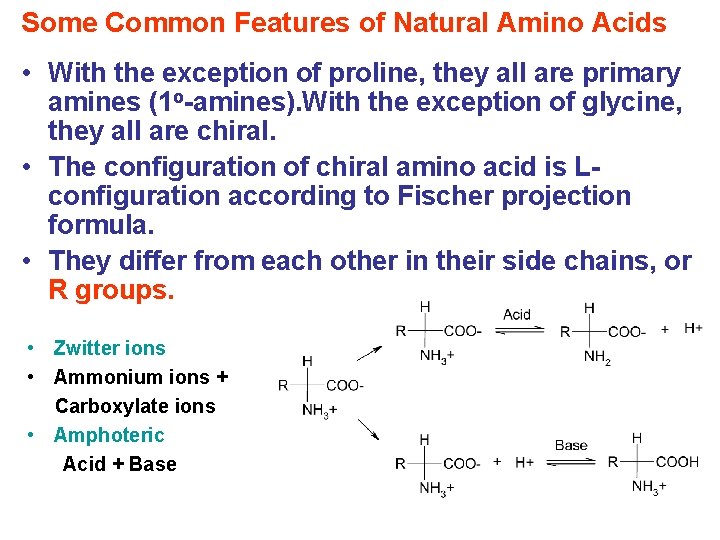
Some Common Features of Natural Amino Acids • With the exception of proline, they all are primary amines (1 o-amines). With the exception of glycine, they all are chiral. • The configuration of chiral amino acid is Lconfiguration according to Fischer projection formula. • They differ from each other in their side chains, or R groups. • Zwitter ions • Ammonium ions + Carboxylate ions • Amphoteric Acid + Base

Ninhydrin reaction of Amino Acids Ninhydrin (2, 2 -Dihydroxyindane-1, 3 -dione or triketohydrindene hydrate ) is a chemical used to detect primary and secondary amines and carboxylic acid groups on proteins and peptides. When reacting with these free amines, a deep blue or purple colour known as Ruhemann's purple is produced. Ninhydrin is also used in amino acid analysis of proteins. Most of the amino acids (except proline) are hydrolyzed and react with ninhydrin. Among the products of this unusual reaction is a purple coloured imino derivative, which provides a useful colour test of the amino acids most of which are colourless.

Ninhydrin reaction of Amino Acids -H 2 O Purple Colored product

Peptide Bond • Peptide Bond: If the amino acid and carboxylic acid functional groups in amino acid join together to form amino bonds, chain of amino acid units (peptide) is formed. • By convention an amino acid with NH 2 group is drawn at the left hand end (N-terminus) of the peptide chain and amino acid containing the free carboxylic acid is drawn on the right (Cterminus). • Free amino acid and carboxylic acid functions on peptide chain form zwiterionic structure at their isoelectric point. Isoelectric point is the p. H at which a particular molecule carries no net charge.
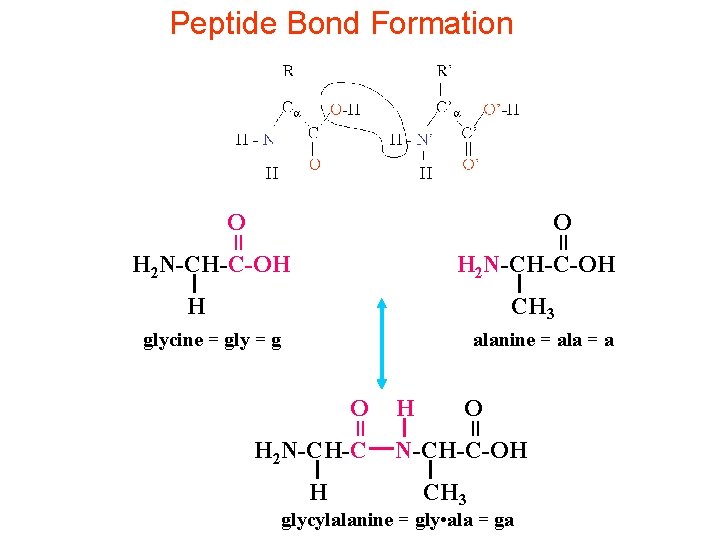
Peptide Bond Formation O = = O H 2 N-CH-C-OH H CH 3 glycine = gly = g alanine = ala = a H 2 N-CH-C H H O = = O N-CH-C-OH CH 3 glycylalanine = gly • ala = ga
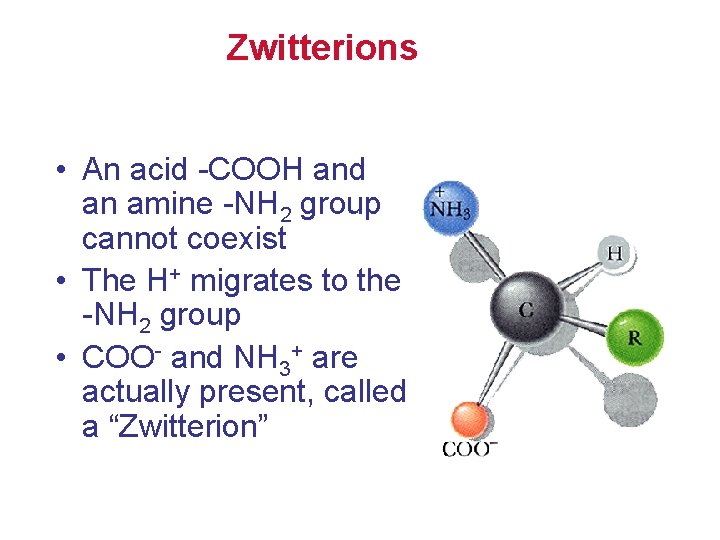
Zwitterions • An acid -COOH and an amine -NH 2 group cannot coexist • The H+ migrates to the -NH 2 group • COO- and NH 3+ are actually present, called a “Zwitterion”
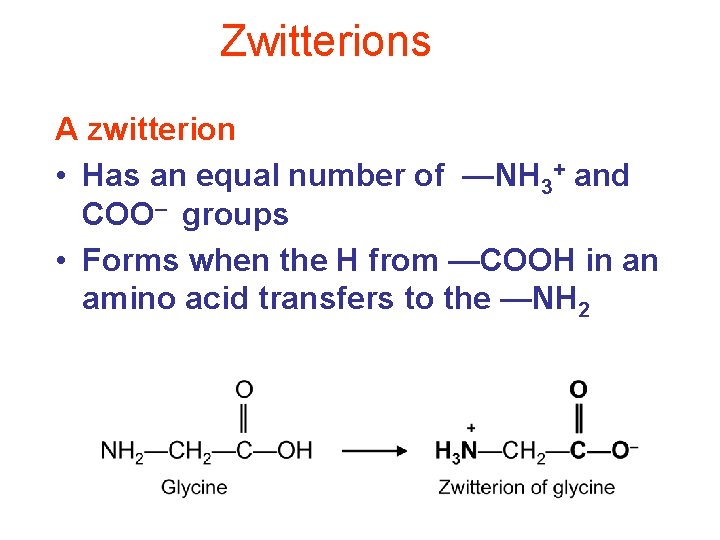
Zwitterions A zwitterion • Has an equal number of —NH 3+ and COO– groups • Forms when the H from —COOH in an amino acid transfers to the —NH 2

Zwitterions in Acidic Solutions In solutions that are more acidic than the p. I, the COO– in the zwitterion accepts a proton the amino acid has a positive charge. Glycine, with a p. I of 6. 0, has a 1+ charge in solutions that have a p. H below p. H 6. 0. 18

Zwitterions in Basic Solutions In solutions that are more basic than the p. I, the NH 3+ in the zwitterion loses a proton the amino acid has a negative charge Glycine, with a p. I of 6. 0, has a 1– charge in solutions that have a p. H above p. H 6. 0. 19

Digestion and Absorption of Protein • Stomach acid and enzymes facilitate the digestion of protein. • It is first denatured, then broken down to polypeptides. • The small intestine continues to break down protein into smaller peptides and amino acids so it can be absorbed.

Digestion and Absorption of Protein • Protein Digestion – In the Small Intestine • Proteases hydrolyze protein into short peptide chains called oligopeptides, which contain four to nine amino acids. • Peptidases split proteins into amino acids.

Digestion and Absorption of Protein • Protein Absorption – Used by intestinal cells for energy or synthesis of necessary compounds – Transported to the liver – Taking enzyme supplements or consuming predigested proteins is unnecessary.

Proteins in the Body • Proteins are versatile and unique. The synthesis of protein is determined by genetic information. • Protein is constantly being broken down and synthesized in the body. • Researchers measure nitrogen balance to study synthesis, degradation and excretion of protein. • Protein has many important functions in the body. • Protein can be used for energy if needed; its excesses are stored as fat. • The study of proteins is called proteomics.
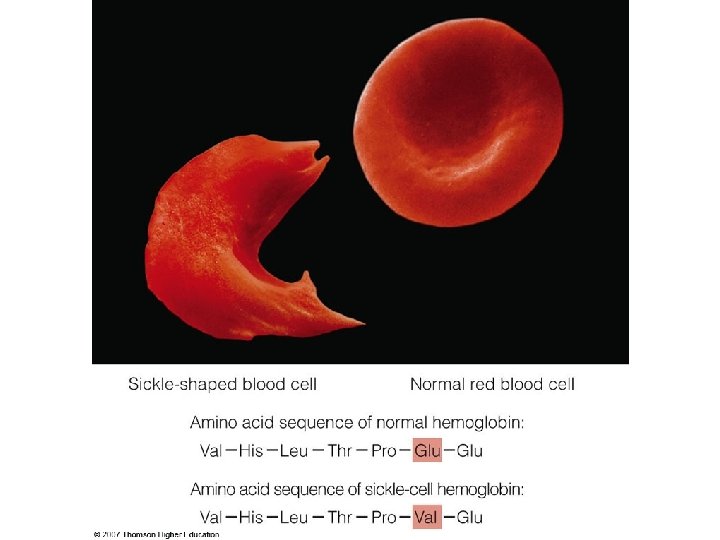

Denaturation of protein • The natural structure of proteins may be altered and their biological activity changed or destroyed by treatment with different agents without disrupting the primary structure. The denaturation is often done deliberately in the course of separating and purification of proteins. • For example many soluble globular proteins precipitate if the p. H of the solution is set at the isoelectric point of the protein.

Following denaturation, some proteins will return to their natural structure under proper conditions. However extreme treatments such as strong heating, usually causes irreversible changes. Not all proteins are easily denatured. Fibrous proteins such as keratins, collagens and elastins are robust, relatively insoluble, quaternary structured proteins that play important roles in the physical structure of organisms. Some treatments known to denature proteins are listed in the following table.

S. No Denaturing Action Mechanism of Operation 1 Heat Hydrogen bonds are broken by increased translational and vibrational energy. (Coagulation of egg white albumin on frying) 2 Ultraviolet Radiation Similar to heat (sunburn) 3 Strong Acids or Bases Salt formation, disruption of hydrogen bonds. (skin blisters and burns, protein precipitation) 4 Urea solution Competition for hydrogen bonds. (precipitation of soluble proteins) 5 Some organic solvents such as ethanol and acetone Change in dielectric constant and hydration of ionic groups. (disinfectant action and precipitation of proteins) 6 Agitation Shearing of hydrogen bonds. (beating egg white albumin into meringue).

Protein in Diet • Protein containing foods are grouped as either complete or incomplete proteins. • Complete proteins contain all nine essential amino acids. • Complete proteins are found in animal foods such as meat, fish, poultry, eggs, milk, and milk products such as yogurt and cheese. • Soybeans are the only plant protein considered to be complete protein.

• Incomplete proteins lack one or more of essential amino acids. Sources of incomplete protein include beans, peas, nuts seeds, and grains. A small amount of incomplete protein is also found in vegetables. • Plant proteins can be combined to provide all of the essential amino acids and form a complete protein. Examples of combined complete proteins are rice and beans, wheat cereals and corn and beans.

How much protein do we need • Proteins needs are determined by your age, sex, weight (whether pregnant), lactating or intense sports training. • The below chart indicates the recommended daily amounts for different types of peoples. • The amount of proteins one needs daily may be calculated by multiplying your weight in pounds by the number that corresponds to one’s situation as shown here. This will give the recommended gram of protein per pound of body weight per day that is appropriate for people of depending on their sex, age/and activity level.
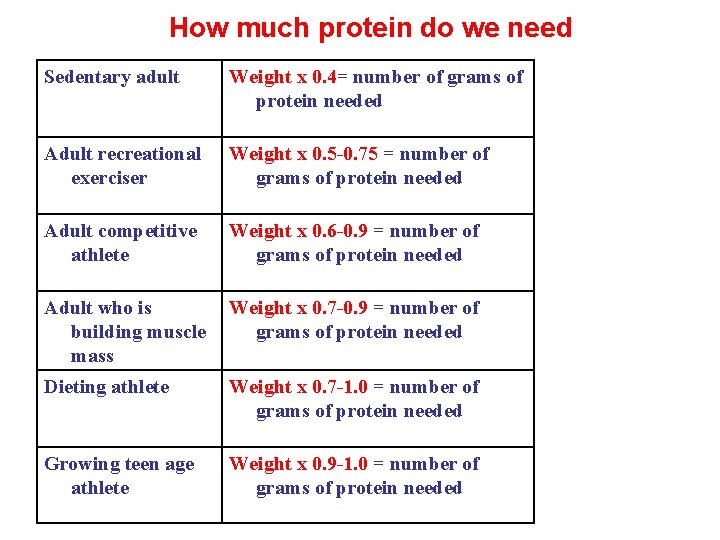
How much protein do we need Sedentary adult Weight x 0. 4= number of grams of protein needed Adult recreational exerciser Weight x 0. 5 -0. 75 = number of grams of protein needed Adult competitive athlete Weight x 0. 6 -0. 9 = number of grams of protein needed Adult who is building muscle mass Weight x 0. 7 -0. 9 = number of grams of protein needed Dieting athlete Weight x 0. 7 -1. 0 = number of grams of protein needed Growing teen age athlete Weight x 0. 9 -1. 0 = number of grams of protein needed

Recommended serving sizes for proteins • 2 to 3 ounces of cooked lean meat, poultry Or fish (a portion about the size of a deck of playing cards) • ½ cup of cooked dried beans. • 1 egg, 2 table spoons of peanut butter, or 1 ounce of cheese. • Recent research indicates that such combination needed not to be eaten at the same meal. If they are consumed over a period of a day, the necessary building of muscle and body tissue will occur.
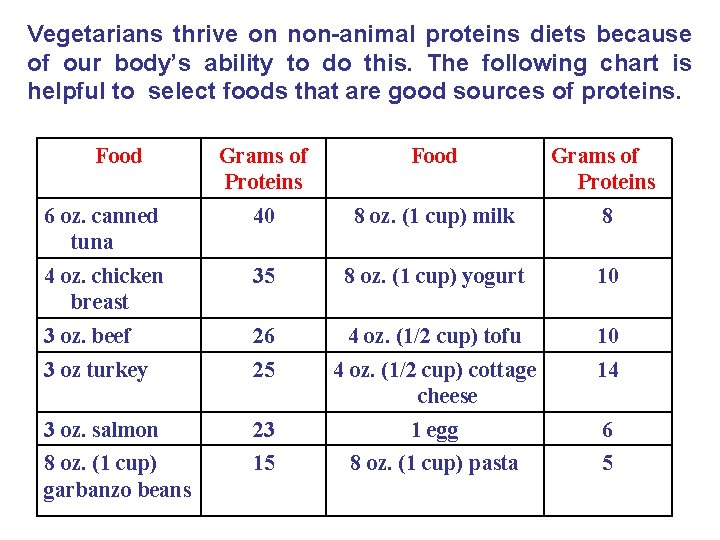
Vegetarians thrive on non-animal proteins diets because of our body’s ability to do this. The following chart is helpful to select foods that are good sources of proteins. Food Grams of Proteins 6 oz. canned tuna 40 8 oz. (1 cup) milk 8 4 oz. chicken breast 35 8 oz. (1 cup) yogurt 10 3 oz. beef 26 4 oz. (1/2 cup) tofu 10 3 oz turkey 25 4 oz. (1/2 cup) cottage cheese 14 3 oz. salmon 23 1 egg 6 8 oz. (1 cup) garbanzo beans 15 8 oz. (1 cup) pasta 5

Proteins Rich Foods • Common proteins rich foods can include: • Milk, soy milk, cheese, yogurt, peanut butter • Lean meat, fish, and pultry, eggs • Beans, tofu, lentils and other legumes. • Grains including bread and pasta • Nuts and seeds. Side Effects of Overeating of proteins: A diet high in meat can contribute to high cholesterol levels or other diseases such as gout (Inflammatory arthritis). A high-protein diet may also out a strain on the kidneys.

Health Effects and Recommended Intakes of Protein – Marasmus • Infancy, 6 to 18 months of age • Severe deprivation or impaired absorption of protein, energy, vitamins and minerals • Develops slowly • Severe weight loss and muscle wasting, including the heart • < 60% weight-for-age • Anxiety and apathy • Good appetite is possible • Hair and skin problems

• Health Effects of Protein – Heart Disease • Foods high in animal protein also tend to be high in saturated fat. • Homocysteine levels increase cardiac risks. • Arginine may protect against cardiac risks. – Cancer • A high intake of animal protein is associated with some cancers. – Adult Bone Loss (Osteoporosis) • High protein intake associated with increased calcium excretion. • Inadequate protein intake affects bone health also.

Proteins Rich Foods Canned tuna tofu Lean meat Salmon soy milk THE END lentils
- Slides: 38