Proteins by Sakvinder S Khalsa WE SHALL LOOK

Proteins by Sakvinder S Khalsa

• WE SHALL LOOK AT PROTEIN SYNTHESIS. • CONSIDER PROTEIN STRUCTURE AT THE MOLECULAR LEVEL. • DISCUSS DIFERENT USES OF PROTEINS. • BREIFLY LOOK AT ENZYMES.

Protein synthesis • Protein synthesis is the making of proteins, using the information that is found in DNA CELL (Chromosomes). chro
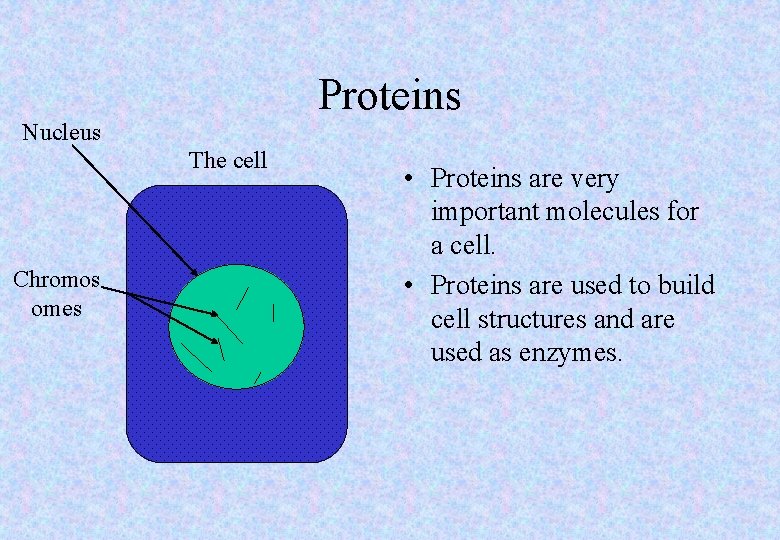
Proteins Nucleus The cell Chromos omes • Proteins are very important molecules for a cell. • Proteins are used to build cell structures and are used as enzymes.

Proteins • Proteins are long chains of small molecules called amino acids. • Different proteins are made using different sequences of amino acids. • The pieces of information in DNA are called genes. • Genes describe how to make proteins by putting the correct amino acids into a long chain in the correct order.
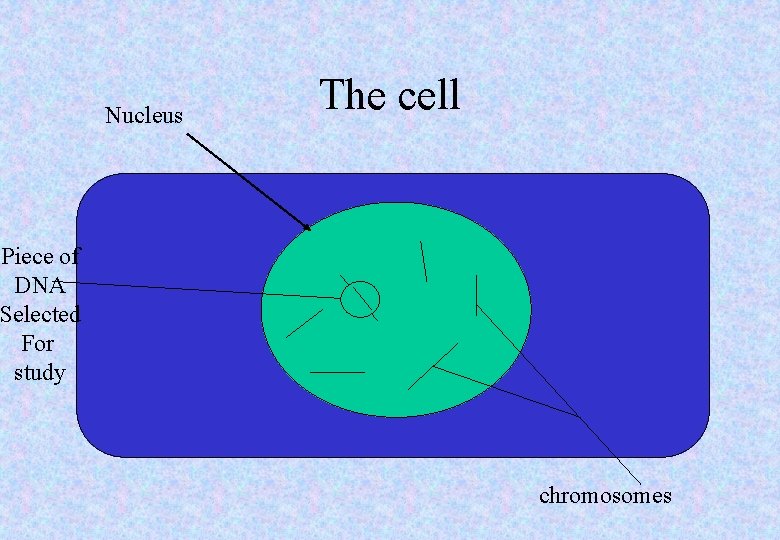
Nucleus The cell Piece of DNA Selected For study chromosomes
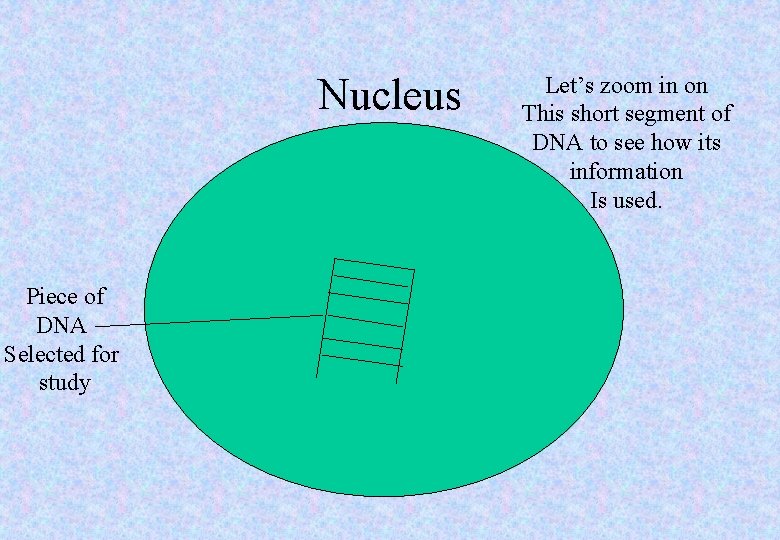
Nucleus Piece of DNA Selected for study Let’s zoom in on This short segment of DNA to see how its information Is used.
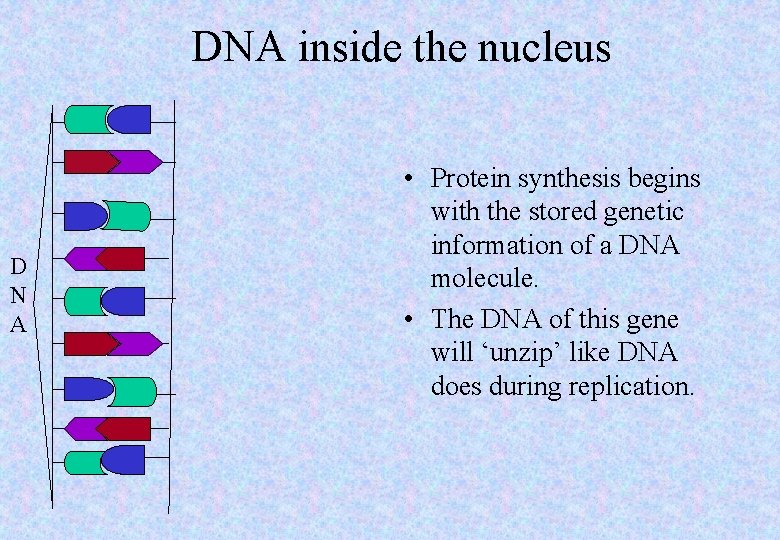
DNA inside the nucleus D N A • Protein synthesis begins with the stored genetic information of a DNA molecule. • The DNA of this gene will ‘unzip’ like DNA does during replication.

Unu
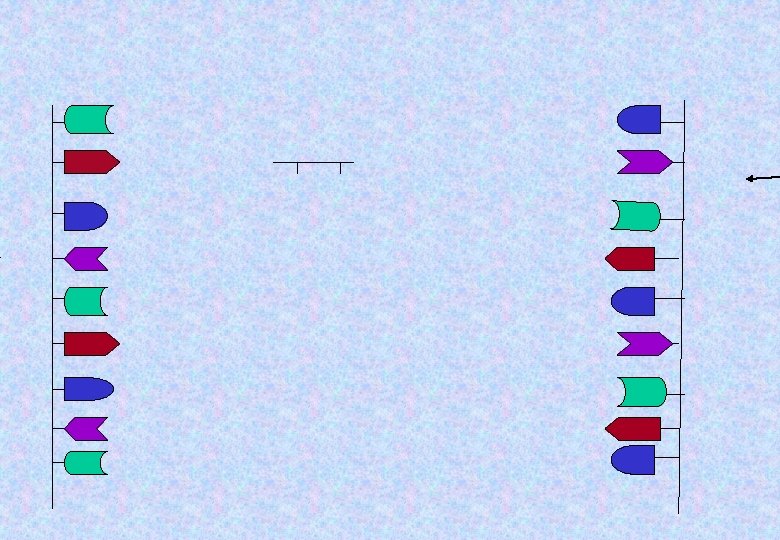
d
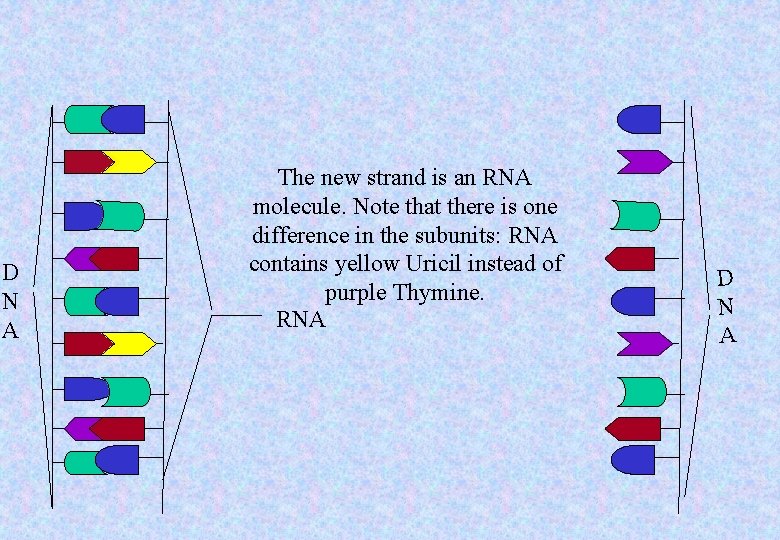
D N A The new strand is an RNA molecule. Note that there is one difference in the subunits: RNA contains yellow Uricil instead of purple Thymine. RNA D N A
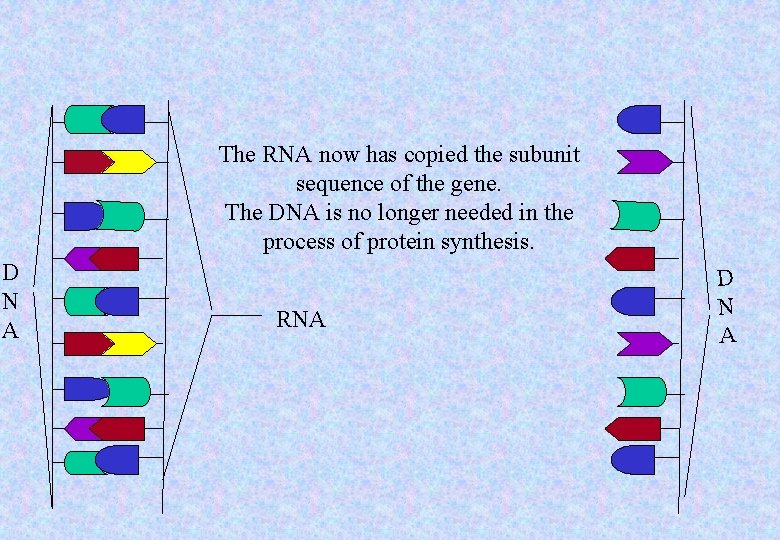
The RNA now has copied the subunit sequence of the gene. The DNA is no longer needed in the process of protein synthesis. D N A RNA D N A
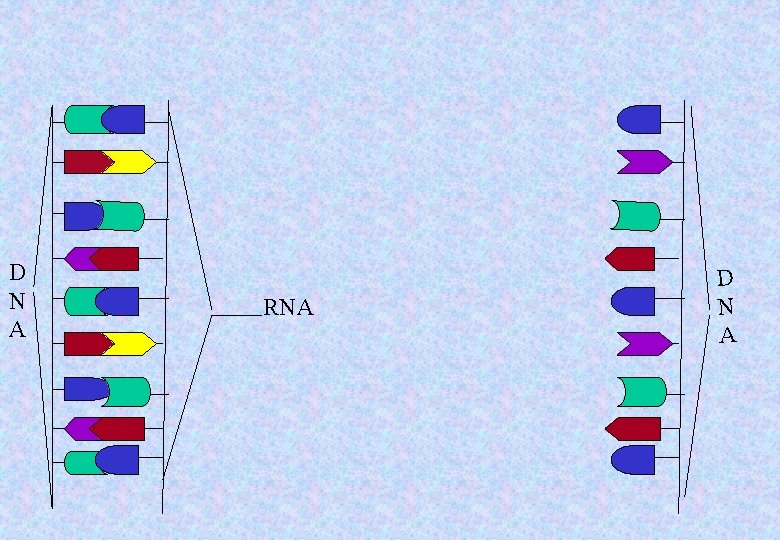
D N A RNA D N A
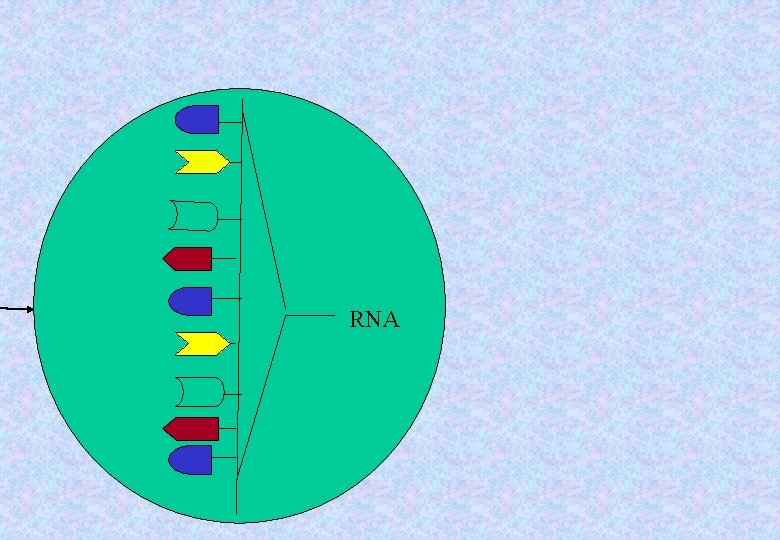
RNA
![A] ion will rotein At the ribosome m. RNA At the ribosome A] ion will rotein At the ribosome m. RNA At the ribosome](http://slidetodoc.com/presentation_image_h/11fffa8f817c08888993bf2785bacd10/image-15.jpg)
A] ion will rotein At the ribosome m. RNA At the ribosome

At the ribosome • The genetic information is interpreted and used to assemble a protein. • We should remember, the m. RNA is a sequence of subunits (like a chain) that tells how to build a protein • A protein is a sequence of subunits – a chain of amino acids.
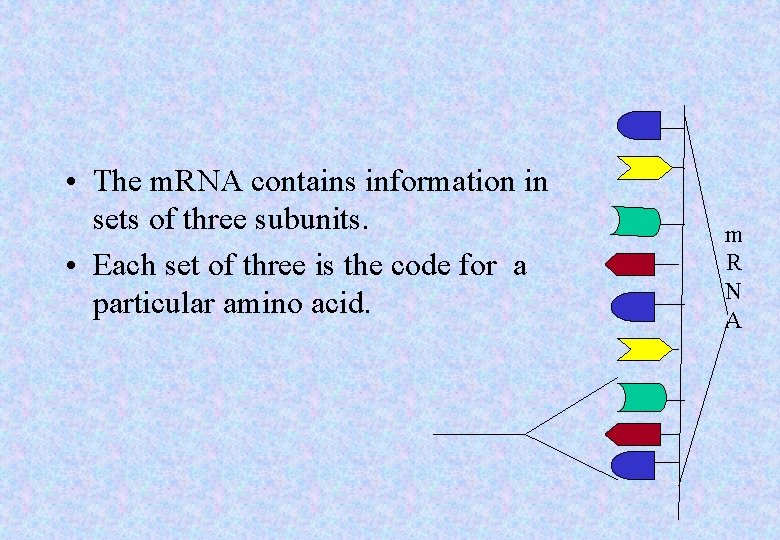
• The m. RNA contains information in sets of three subunits. • Each set of three is the code for a particular amino acid. m R N A
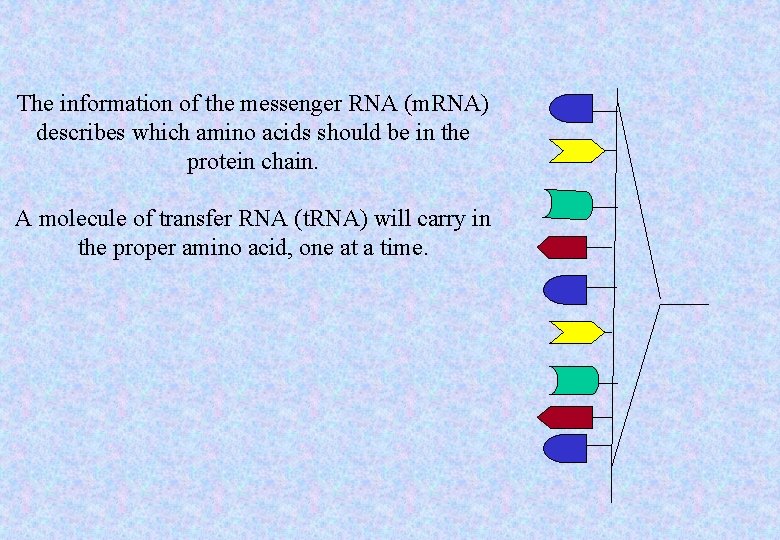
The information of the messenger RNA (m. RNA) describes which amino acids should be in the protein chain. A molecule of transfer RNA (t. RNA) will carry in the proper amino acid, one at a time.
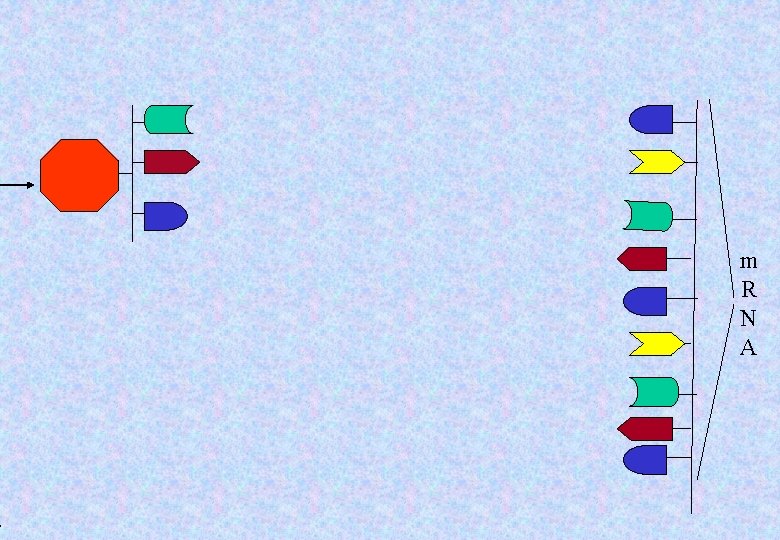
. m R N A
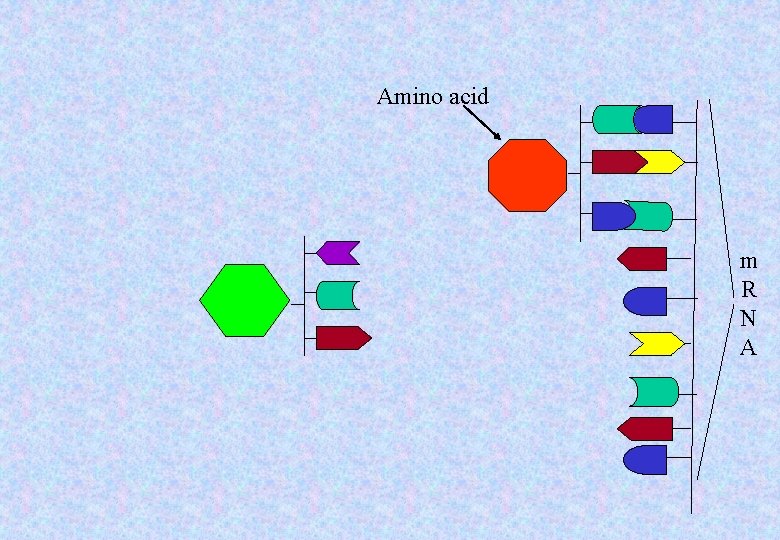
Amino acid m R N A
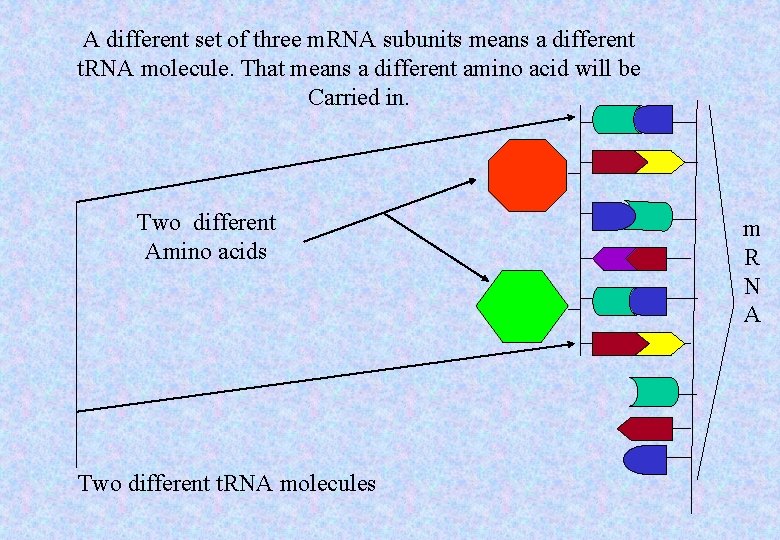
A different set of three m. RNA subunits means a different t. RNA molecule. That means a different amino acid will be Carried in. Two different Amino acids Two different t. RNA molecules m R N A
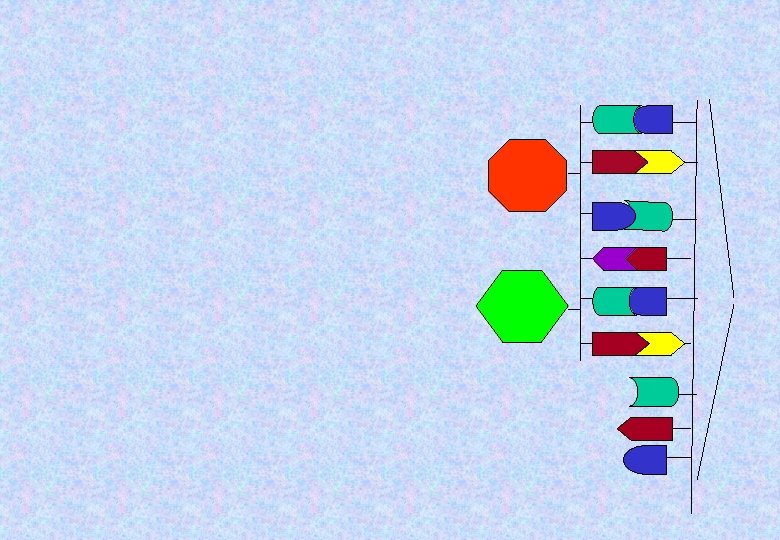
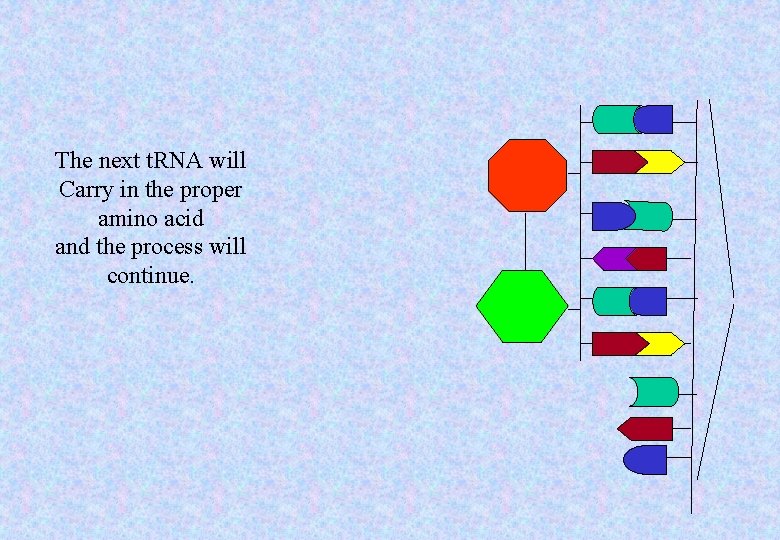
The next t. RNA will Carry in the proper amino acid and the process will continue.
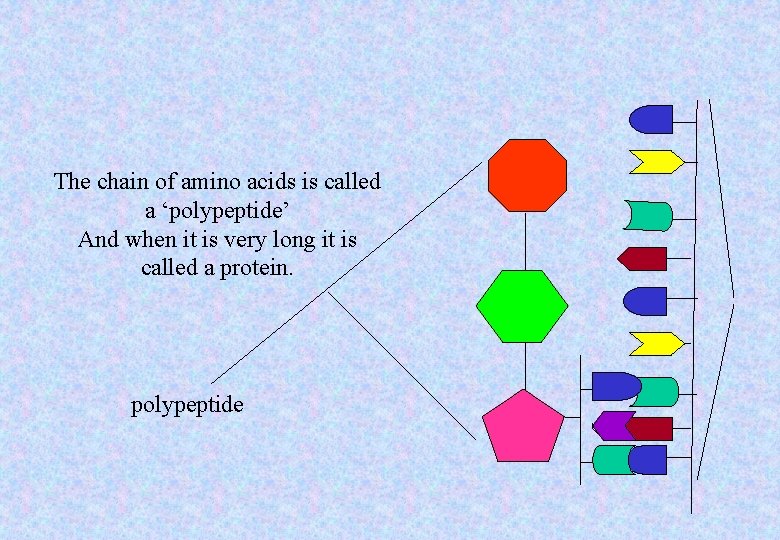
The chain of amino acids is called a ‘polypeptide’ And when it is very long it is called a protein. polypeptide
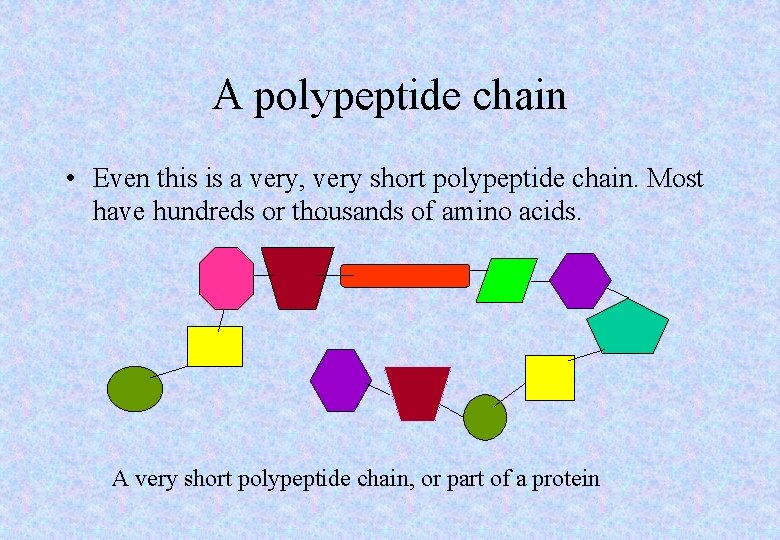
A polypeptide chain • Even this is a very, very short polypeptide chain. Most have hundreds or thousands of amino acids. A very short polypeptide chain, or part of a protein

The end of protein synthesis at the ribosome.
PROTEIN STRUCTURE • We will look at the main elements found in proteins. • Recall how proteins are constructed. • Look at the structure of proteins. • Overview the major functions of proteins.
The building blocks of proteins • Like carbohydrate and lipid molecules proteins contain the elements : Oxygen(O), Carbon(C), and Hydrogen(H) • In addition they always contain the element Nitrogen(N).

• Before we can understand how proteins are constructed, the structure of amino acids needs to be considered.
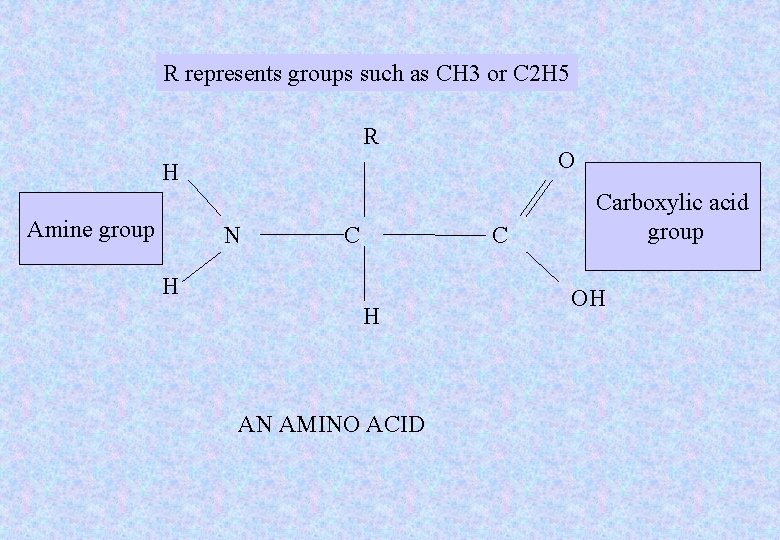
R represents groups such as CH 3 or C 2 H 5 R O H Amine group N C C H H AN AMINO ACID Carboxylic acid group OH
How are proteins constructed • First the Amino acids bond together. • They are joined together by what is known as a peptide bond.
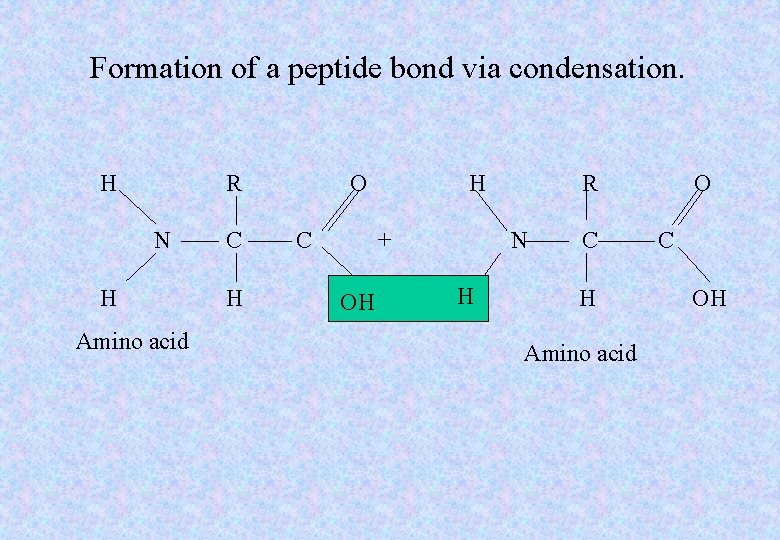
Formation of a peptide bond via condensation. H R N H Amino acid C H O C H + OH R N H C H Amino acid O C OH
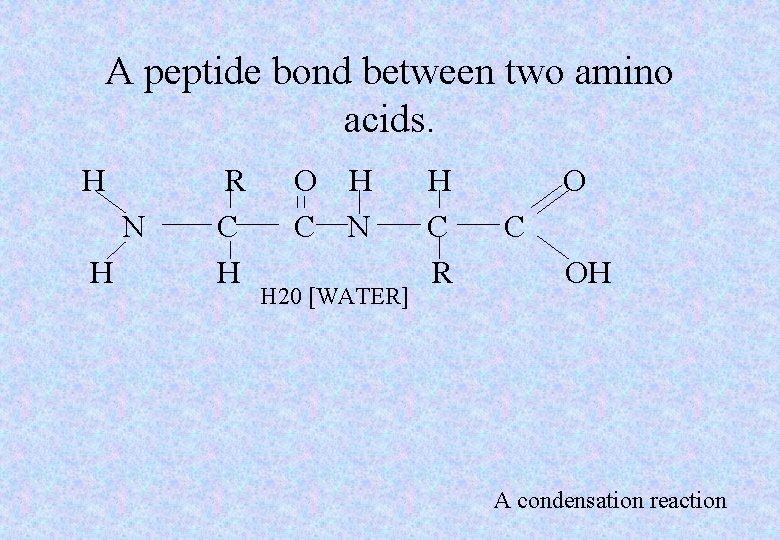
A peptide bond between two amino acids. H N H R C H O H C N H 20 [WATER] H C R O C OH A condensation reaction

Protein construction • When two amino acids join together they form a dipeptide. • When many amino acids are joined together a long-chain polypeptide is formed. • Organisms join amino acids in different linear sequences to form a variety of polypeptides in to complex molecules, the proteins.
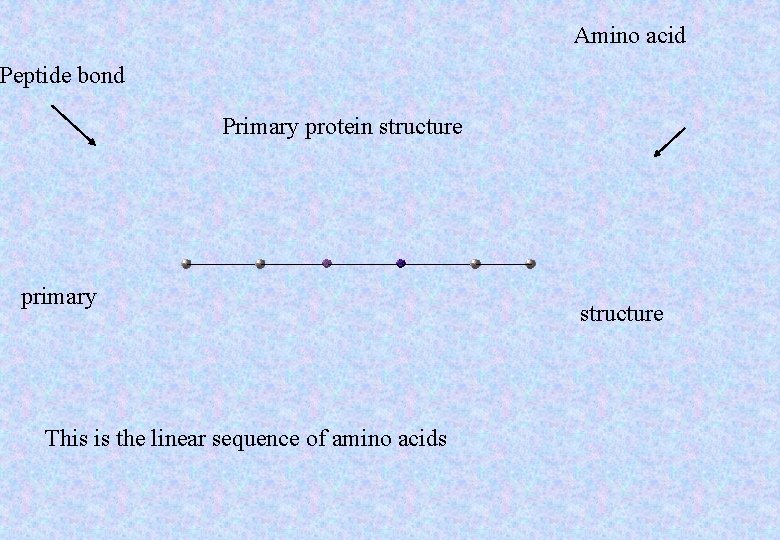
Amino acid Peptide bond Primary protein structure primary This is the linear sequence of amino acids structure
Secondary protein structure Polypeptides become twisted or coiled. These shapes are known as the secondary Structure. There are two common secondary structures The alpha-helix and the beta-pleated sheet.
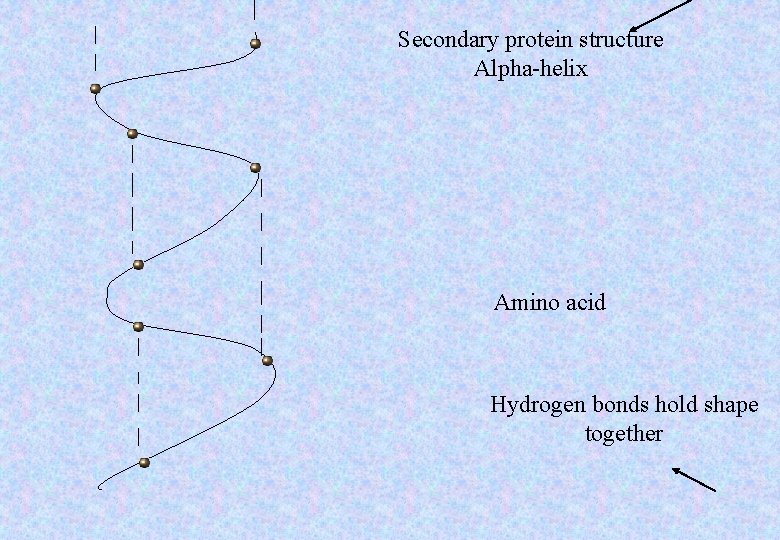
Secondary protein structure Alpha-helix Amino acid Hydrogen bonds hold shape together
![Secondary Protein structure [The beta pleated sheet] Amino acid Secondary Protein structure [The beta pleated sheet] Amino acid](http://slidetodoc.com/presentation_image_h/11fffa8f817c08888993bf2785bacd10/image-38.jpg)
Secondary Protein structure [The beta pleated sheet] Amino acid

Hydrogen bonds The polypeptides are held in position by hydrogen bonds. In both alpha-helices and beta pleated sheets the C=O of one amino acid bonds to the H-N of an adjacent amino acid. As below: C=O----H-N

Secondary structures • Both secondary structures give additional strength to proteins. The alpha-helix helps make fibres like in your nails, e. g. Keratin. • The beta pleated-sheet helps make the strength giving protein in silk, fibroin. • Many proteins are made from both alpha-helix and beta-pleated sheet.

Fibrous proteins • A fibrous protein only achieves a secondary structure. • The simple alpha-helix polypeptides do not undergo further folding.

d elix ure Structure of a fibrous protein
Tertiary protein structure • This is when a polypeptide is folded into a precise shape. • The polypeptide is held in ‘bends’ and ‘tucks’ in a permanent shape by a range of bonds including: • Disulphide bridges [sulphur-sulphur bonds] • Hydrogen bonds • Ionic bonds.
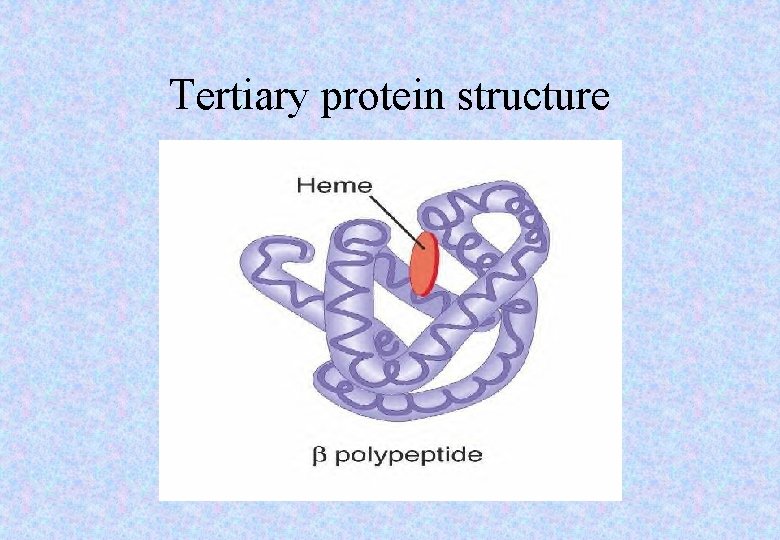
Tertiary protein structure

Quaternary protein structure • Some proteins consist of different polypeptides bonded together to form extremely intricate shapes. • A haemoglobin molecule is formed for separate polypeptide chains. • It also has a haem group, which contains iron. • The inorganic group is known as the prosthetic group. • In haemoglobin it aids oxygen transport.
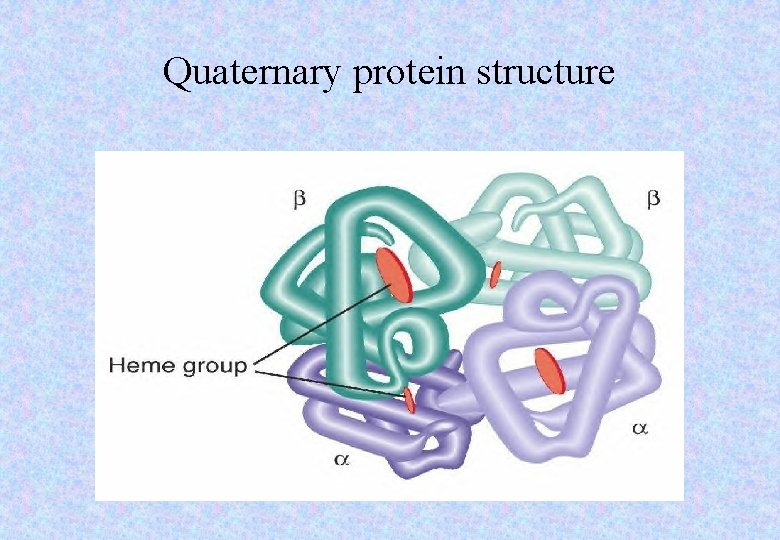
Quaternary protein structure

How useful are proteins? • Cell membrane proteins: Transport substances across the membrane for processes such as facilitated diffusion and active transport. • Enzymes: Catalyse biochemical reactions, e. g. pepsin breaks down protein in to polypeptides.

• Hormones: are passed through the blood and trigger reactions in other parts of the body e. g. insulin regulates blood sugar. • Immuno-proteins: e. g. antibodies are made by lymphocytes and act against antigenic sites on microbes. • Structural proteins: give strength to organs, e. g. collagen makes tendons tough.

• Transport proteins: e. g. haemoglobin transports oxygen in the blood. • Contractile proteins: e. g. actin and myosin help muscles shorten during contraction • Storage proteins: e. g. aleurone in seeds helps germination, and casein in milk helps supply valuable protein to babies. • Buffer proteins: e. g. blood proteins, due to their high charge, help maintain the p. H of plasma.

Enzymes • Living cells carry out many biochemical reactions. • These reactions take place rapidly due to enzymes. • All enzymes consist of globular proteins.

Enzymes • The tertiary folding of polypeptides are responsible for the special shape of the ‘active’ site. • Some enzymes require additional nonprotein groups to enable them to work efficiently. e. g the enzyme dehydrogenase needs coenzyme NAD to function.
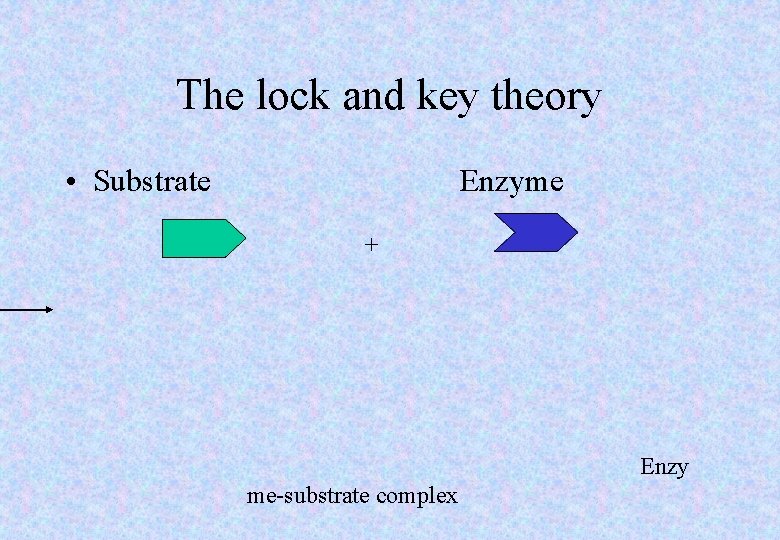
The lock and key theory • Substrate Enzyme + Enzy me-substrate complex
![oducts A catabolic reaction [substrate broken down] • • enzyme-substrate complex oducts A catabolic reaction [substrate broken down] • • enzyme-substrate complex](http://slidetodoc.com/presentation_image_h/11fffa8f817c08888993bf2785bacd10/image-53.jpg)
oducts A catabolic reaction [substrate broken down] • • enzyme-substrate complex
![An anabolic reaction [substrates used to build a new molecule] substrate An anabolic reaction [substrates used to build a new molecule] substrate](http://slidetodoc.com/presentation_image_h/11fffa8f817c08888993bf2785bacd10/image-54.jpg)
An anabolic reaction [substrates used to build a new molecule] substrate

Anabolic reaction continued • Enzyme substrate complex

Metabolic reactions • Metabolic reactions = anabolic reaction + catabolic reaction. • Metabolism is a summary of build up and break down reactions.

Induced fit theory • The active site is a cavity of a particular shape. • Initially the active site is not the correct shape in which to fit the substrate. • As the substrate approaches the active site, the site changes and this results in it being a perfect fit. • After the reaction has taken place, and the products have gone, the active site returns to its normal shape.
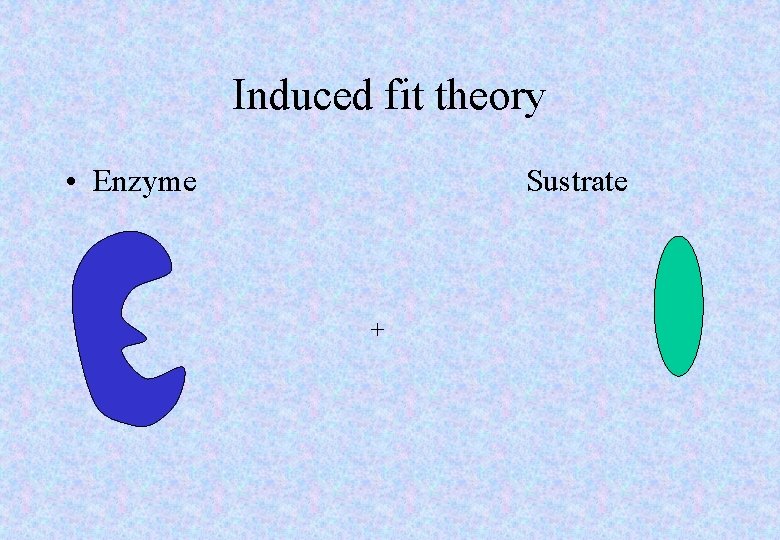
Induced fit theory • Enzyme Sustrate +

Induced fit continued In Enzyme-substrate complex

pr enzyme
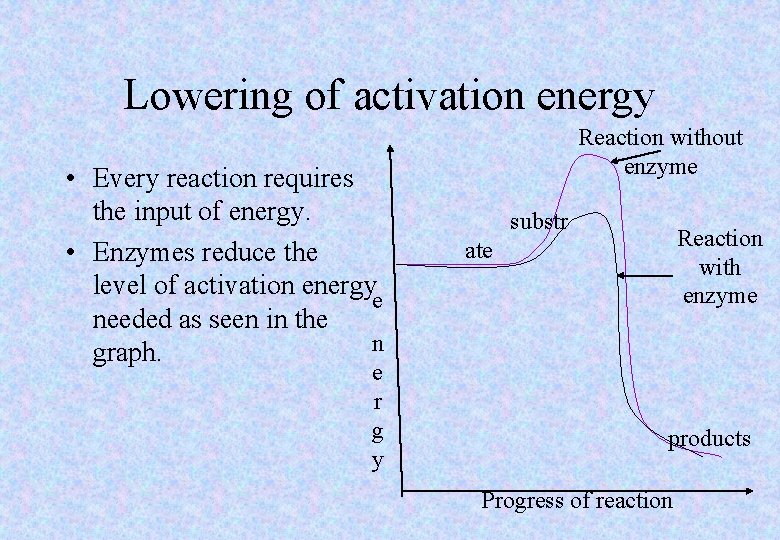
Lowering of activation energy • Every reaction requires the input of energy. • Enzymes reduce the level of activation energye needed as seen in the n graph. e r g y Reaction without enzyme substr Reaction with enzyme ate products Progress of reaction

Two minute summary • Now you have seen the presentation ! • Summarise the most important points of this presentation. • What was the ‘muddiest’ point in the presentation? • Hand in your paper to the teacher before you leave the classroom.
![END OF PRESENTATION by S S Khalsa [science PGCE] END OF PRESENTATION by S S Khalsa [science PGCE]](http://slidetodoc.com/presentation_image_h/11fffa8f817c08888993bf2785bacd10/image-63.jpg)
END OF PRESENTATION by S S Khalsa [science PGCE]

This powerpoint was kindly donated to www. worldofteaching. com http: //www. worldofteaching. com is home to over a thousand powerpoints submitted by teachers. This is a completely free site and requires no registration. Please visit and I hope it will help in your teaching.
- Slides: 64