Proteins Building Blocks of Life By Robert Ramirez

Proteins: Building Blocks of Life By: Robert Ramirez, David Flores, Eduardo Becerra, Edward Zhu, Aram Gebretensae, and Thu Tran
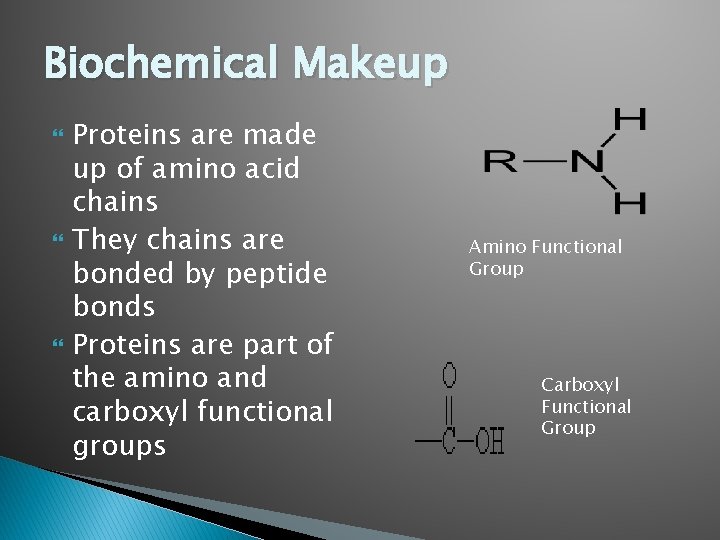
Biochemical Makeup Proteins are made up of amino acid chains They chains are bonded by peptide bonds Proteins are part of the amino and carboxyl functional groups Amino Functional Group Carboxyl Functional Group
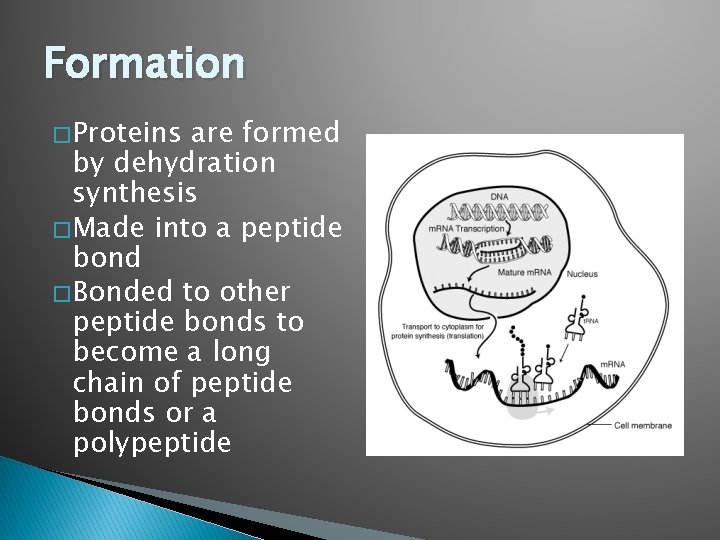
Formation � Proteins are formed by dehydration synthesis � Made into a peptide bond � Bonded to other peptide bonds to become a long chain of peptide bonds or a polypeptide
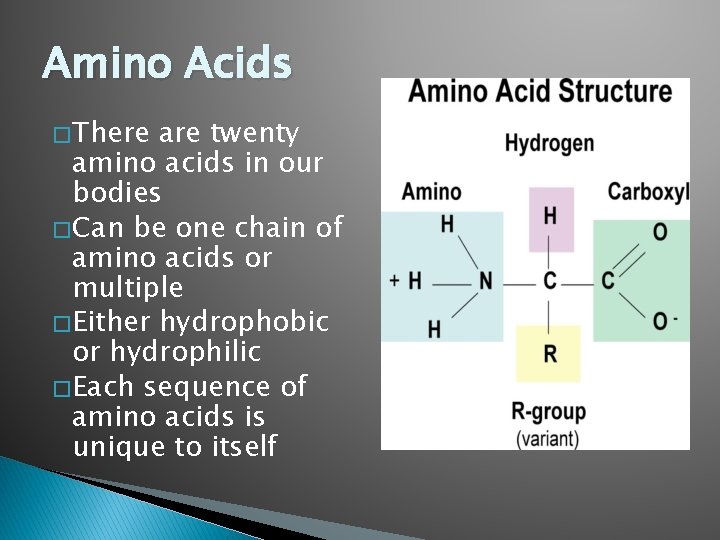
Amino Acids � There are twenty amino acids in our bodies � Can be one chain of amino acids or multiple � Either hydrophobic or hydrophilic � Each sequence of amino acids is unique to itself
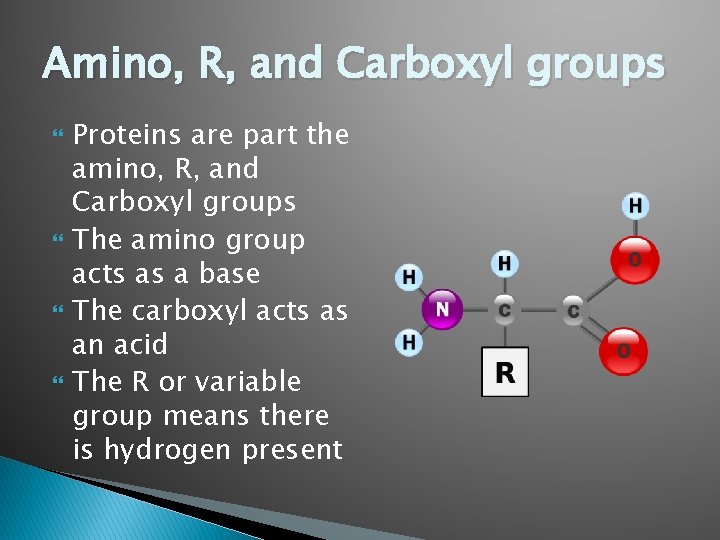
Amino, R, and Carboxyl groups Proteins are part the amino, R, and Carboxyl groups The amino group acts as a base The carboxyl acts as an acid The R or variable group means there is hydrogen present
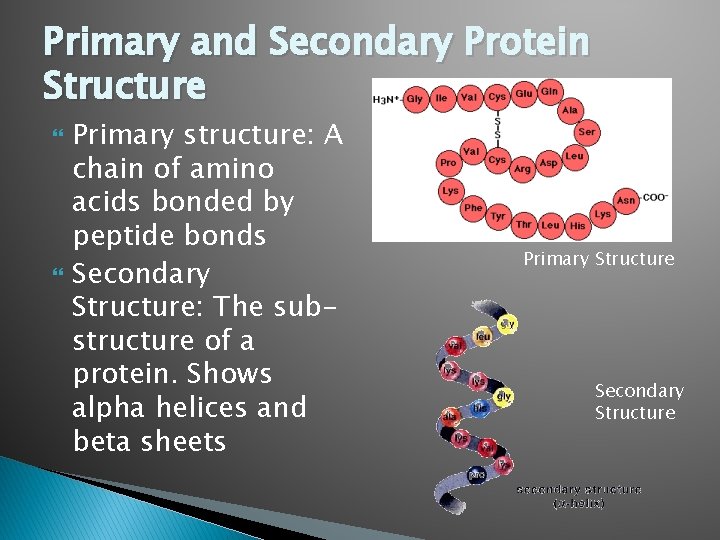
Primary and Secondary Protein Structure Primary structure: A chain of amino acids bonded by peptide bonds Secondary Structure: The substructure of a protein. Shows alpha helices and beta sheets Primary Structure Secondary Structure
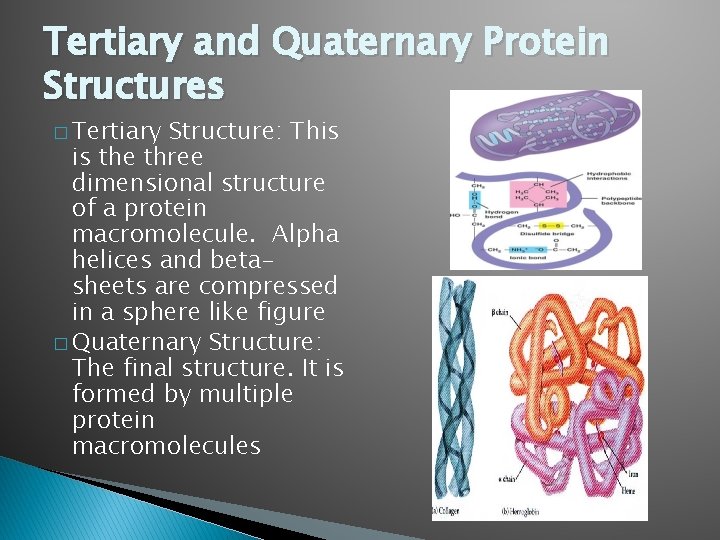
Tertiary and Quaternary Protein Structures � Tertiary Structure: This is the three dimensional structure of a protein macromolecule. Alpha helices and betasheets are compressed in a sphere like figure � Quaternary Structure: The final structure. It is formed by multiple protein macromolecules
Types of Proteins � Enzymes � Amino Acids � Hormones � Structural proteins � Storage proteins � Transport � Signaling � Transport � Antibodies � Motor � Receptors Testosterone Antibodie s
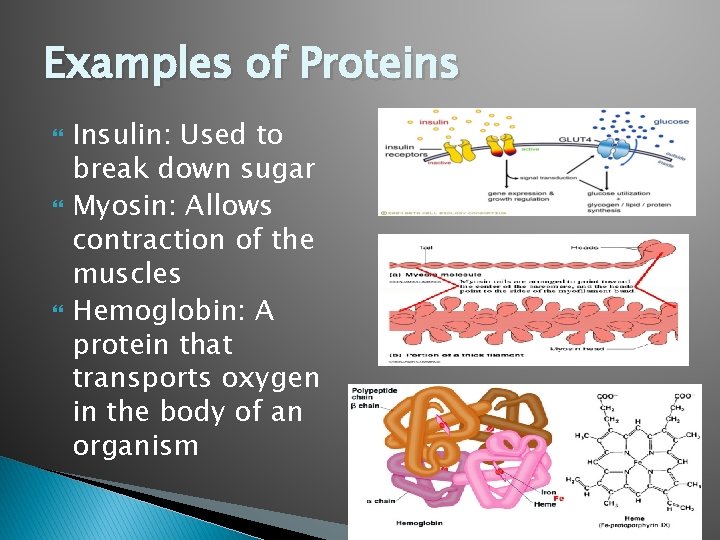
Examples of Proteins Insulin: Used to break down sugar Myosin: Allows contraction of the muscles Hemoglobin: A protein that transports oxygen in the body of an organism

The role of protein in Human life Building Blocks of Life Very important to overall health Build and repair body tissues Key part of metabolism Action proteins help humans contract muscles Create muscle mass Repair muscles after rigorous workouts
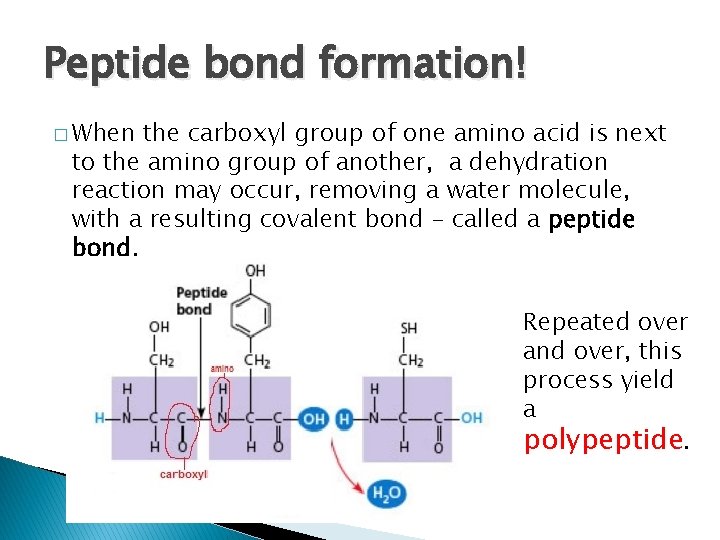
Peptide bond formation! � When the carboxyl group of one amino acid is next to the amino group of another, a dehydration reaction may occur, removing a water molecule, with a resulting covalent bond – called a peptide bond. Repeated over and over, this process yield a polypeptide.

Conformation � *Reminder: A functional protein is not just a polypeptide chain, but one or more polypeptides twisted, and folded into a uniquely shaped molecule. Conformation: three-dimensional arrangement of side groups on a molecule which can freely rotate into different positions without breaking any bonds. � A protein’s specific conformation determines how it works! � In almost every case, the function of a protein depends on its ability to bind to some other molecule. � � � Like antibodies -> bind to foreign substances in body Enzymes bind to substrates.

Denaturation Protein conformation also depends on the physical and chemical conditions of the protein’s environment. If aspects of the environment are altered, like the p. H, salt concentration, temperature, the protein may misshape, and untangle. Denatured protein are biologically dormant : o
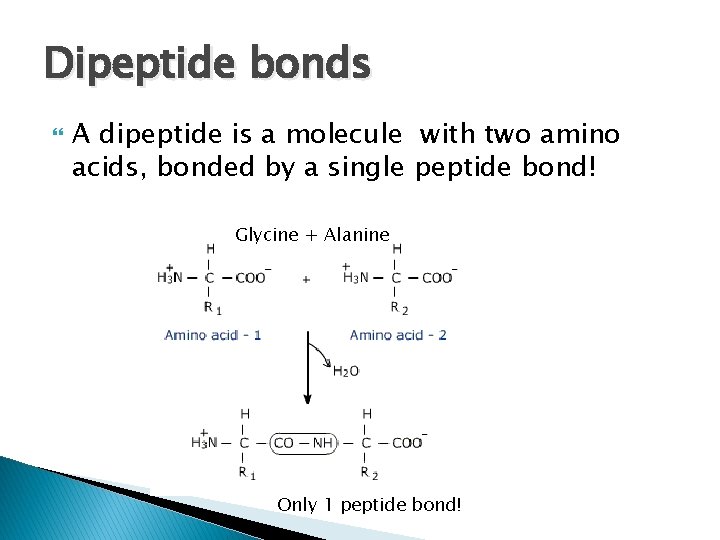
Dipeptide bonds A dipeptide is a molecule with two amino acids, bonded by a single peptide bond! Glycine + Alanine Only 1 peptide bond!
Chemical Formulas � The chemical formulas for proteins contains many hydrogen atoms and carbon atoms � There are 3 common roots found in the formulas: � Gly, ser, and ala
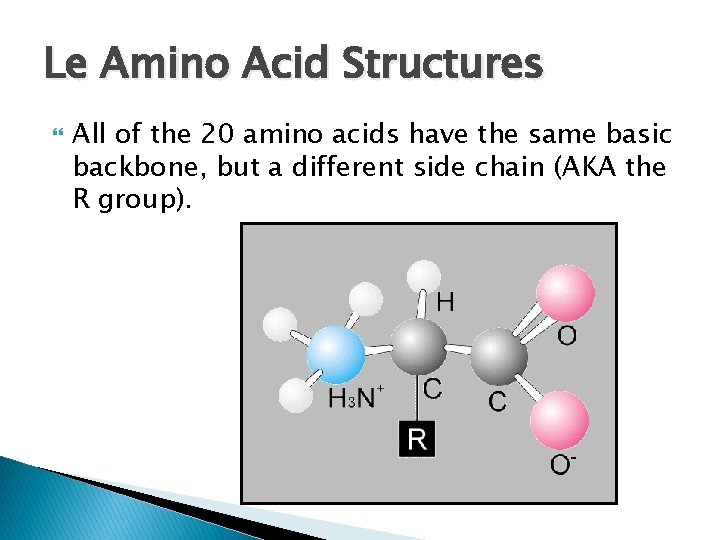
Le Amino Acid Structures All of the 20 amino acids have the same basic backbone, but a different side chain (AKA the R group).
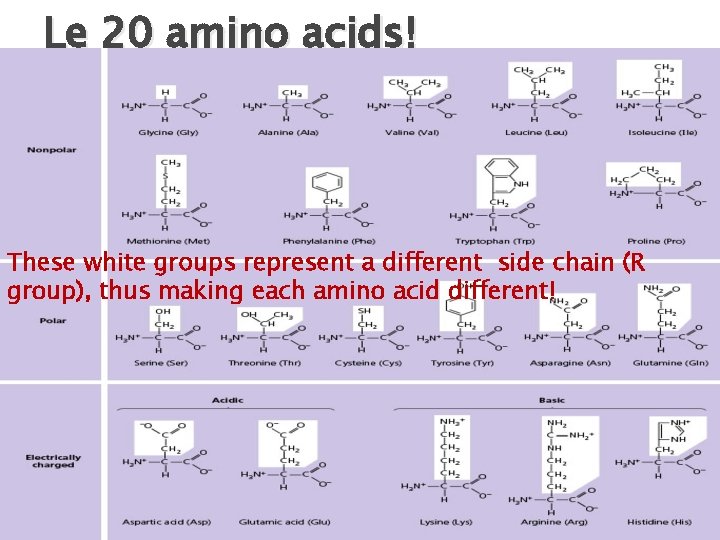
Le 20 amino acids! These white groups represent a different side chain (R group), thus making each amino acid different!

Citations for pictures http: //ca. wikipedia. org/wiki/Fitxer: Aminogroup-primary-2 D-flat. png http: //www. 3 rd 1000. com/chem 301/chem 30 1 a. htm http: //en. wikipedia. org/wiki/File: MRNAinteraction. png http: //www. proprofs. com/flashcards/cardsho wall. php? title=biological-molecules-aqaproteins

Citations for pictures (continued) � http: //en. wikipedia. org/wiki/File: Amino. Acidball. svg � http: //users. rcn. com/jkimball. ma. ultranet/Biology. Pag es/P/Primary. Structure. html � http: //chsweb. lr. k 12. nj. us/mstanley/outlines/organic AP/aporgchem. html � http: //kentsimmons. uwinnipeg. ca/cm 1504/proteins. htm � http: //www. google. com/imgres? q=protein+quaterna ry+structure&hl=en&safe=off&sa=X&gbv=2&biw=16 00&bih=809&tbm=isch&tbnid=U 8 t_m. Pkm 3 ms. KHM: & imgrefurl=http: //fc. sjsd. net/~gvalentim/FOV 100043 AE 4/FOV 1000488 C 8/Biochemistry. pptx%3 FFCItem. ID%3 DS 0239 30 F 1%26 Plugin%3 DLoft&docid=2 nh. Zuozs 7 Fbie. M&w= 800&h=531&ei=_QGVTtr. PEY 39 s. QKZt-Xv. AQ&zoom=1

Citations for pictures (continued) http: //www. worldofmolecules. com/emotions /testosterone. htm http: //www. abpro-labs. com/ http: //happyhealthybalance. blogspot. com/20 10/05/insulin-sugar-fat. html http: //www. unm. edu/~jimmy/myosin. jpg http: //chemistry. wiki. elanco. net/Element+Inf ormation http: //www. inchem. org/documents/ehc/ v 219 eh 02. gif
- Slides: 20