Proteins as Pathogens Stanley B Prusiner MD The

Proteins as Pathogens Stanley B. Prusiner, MD The Nobel Prize in Physiology or Medicine 1997 Presented by Shannon S. Rickner-Schmidt
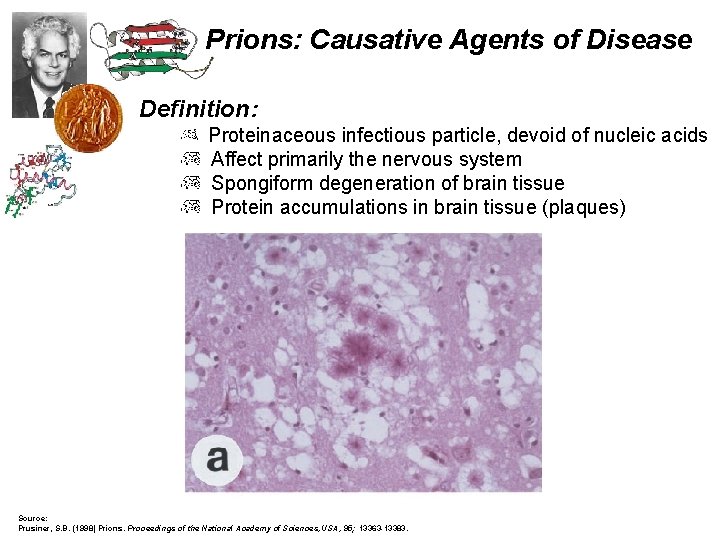
Prions: Causative Agents of Disease Definition: Proteinaceous infectious particle, devoid of nucleic acids Affect primarily the nervous system Spongiform degeneration of brain tissue Protein accumulations in brain tissue (plaques) Source: Prusiner, S. B. (1998) Prions. Proceedings of the National Academy of Sciences, USA, 95; 13363 -13383.
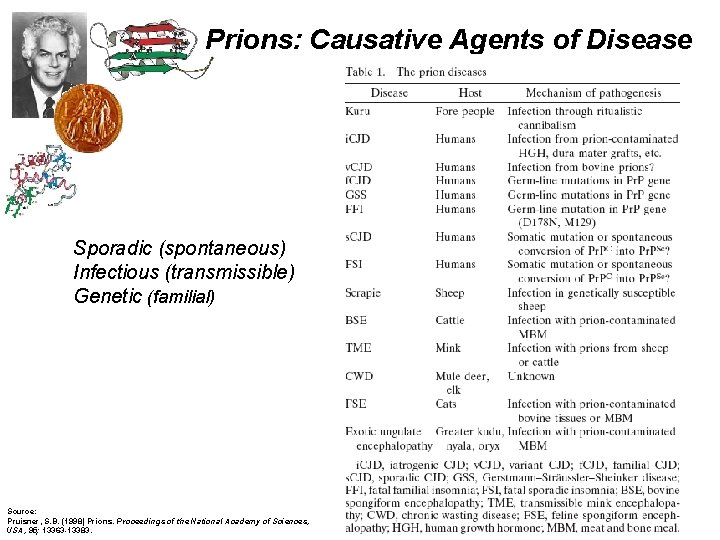
Prions: Causative Agents of Disease Sporadic (spontaneous) Infectious (transmissible) Genetic (familial) Source: Pruisner, S. B. (1998) Prions. Proceedings of the National Academy of Sciences, USA, 95; 13363 -13383.
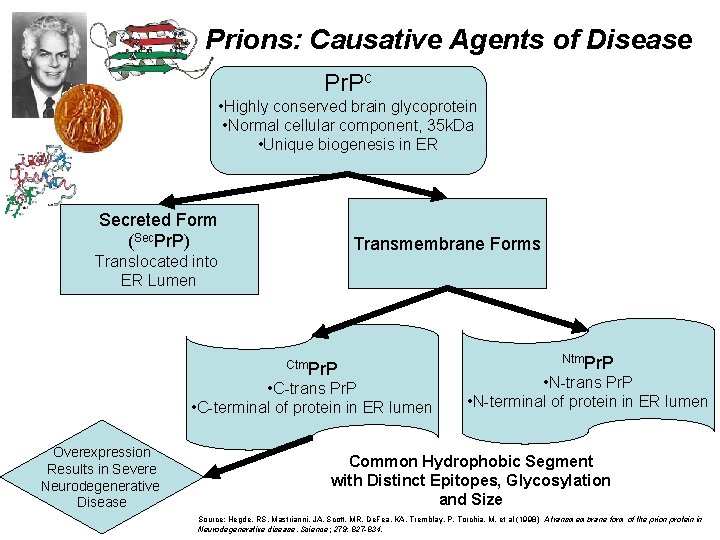
Prions: Causative Agents of Disease Pr. Pc • Highly conserved brain glycoprotein • Normal cellular component, 35 k. Da • Unique biogenesis in ER Secreted Form (Sec. Pr. P) Transmembrane Forms Translocated into ER Lumen Ctm. Pr. P • C-trans Pr. P • C-terminal of protein in ER lumen Overexpression Results in Severe Neurodegenerative Disease Ntm. Pr. P • N-trans Pr. P • N-terminal of protein in ER lumen Common Hydrophobic Segment with Distinct Epitopes, Glycosylation and Size Source: Hegde, RS, Mastrianni, JA, Scott, MR, De. Fea, KA, Tremblay, P, Torchia, M, et al (1998) A transmembrane form of the prion protein in Neurodegenerative disease. Science; 279: 827 -834.

Function Follows Form: Isoforms Pr. PSc: Prion Protein Scrapie- Infectious form When exposed to Pr. PSc, normal constituent of mammalian cells (Ctm. Pr. P) becomes infectious form through a structural change Hypothesis: Ability of host to make the Ctm. Pr. P form determines effectiveness of Pr. PSc in causing neurodegenerative disease

Designing the Experiment Mutant mice that do not produce Ctm. Pr. P (FVB/Pmp 0/0) Create transgenic (Tg) lines by introducing either mutated or normal hamster genes (SHa. Pr. P) Correlate neurodegeneration with the Pr. P form expressed Transgenic Line Expression (KH→II)H High Ctm. Pr. P (KH→II)M Medium Ctm. Pr. P (A 117 V)H High Ctm. Pr. P (N 108 I)H High Ctm. Pr. P (ΔSTE)H Sec. Pr. P (KH→II)L Low Ctm. Pr. P (A 117 V)L Low Ctm. Pr. P (N 108 I)L Low Ctm. Pr. P only
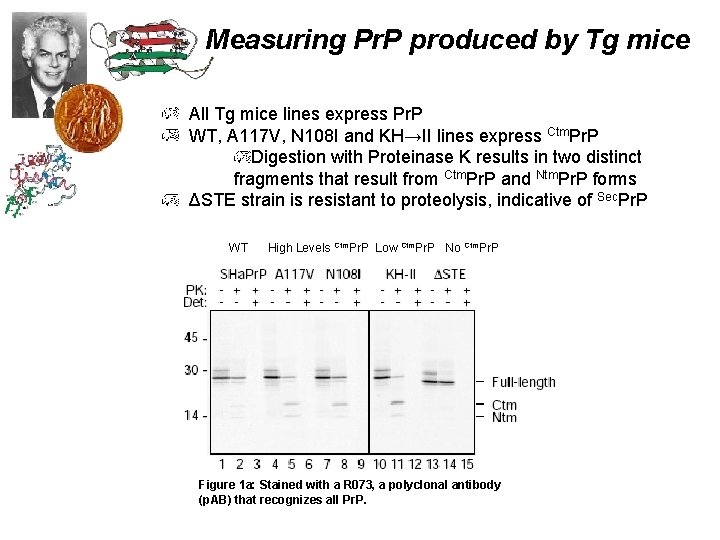
Measuring Pr. P produced by Tg mice All Tg mice lines express Pr. P WT, A 117 V, N 108 I and KH→II lines express Ctm. Pr. P Digestion with Proteinase K results in two distinct fragments that result from Ctm. Pr. P and Ntm. Pr. P forms ΔSTE strain is resistant to proteolysis, indicative of Sec. Pr. P WT High Levels Ctm. Pr. P Low Ctm. Pr. P No Ctm. Pr. P Figure 1 a: Stained with a R 073, a polyclonal antibody (p. AB) that recognizes all Pr. P.
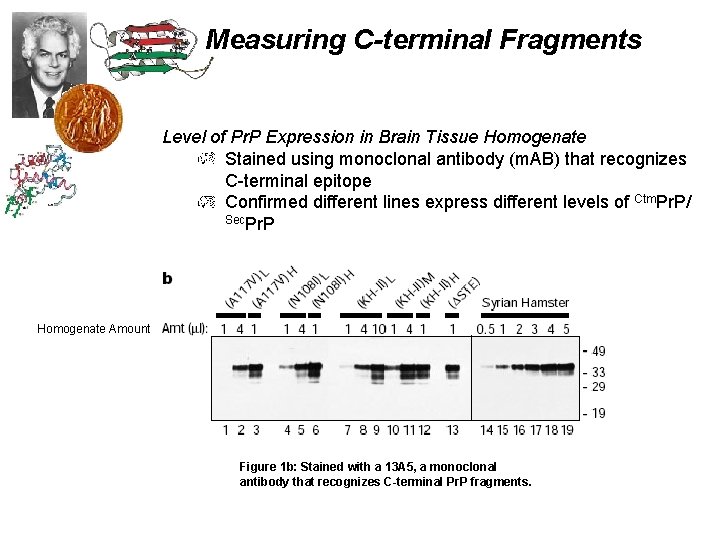
Measuring C-terminal Fragments Level of Pr. P Expression in Brain Tissue Homogenate Stained using monoclonal antibody (m. AB) that recognizes C-terminal epitope Confirmed different lines express different levels of Ctm. Pr. P/ Sec. Pr. P Homogenate Amount Figure 1 b: Stained with a 13 A 5, a monoclonal antibody that recognizes C-terminal Pr. P fragments.
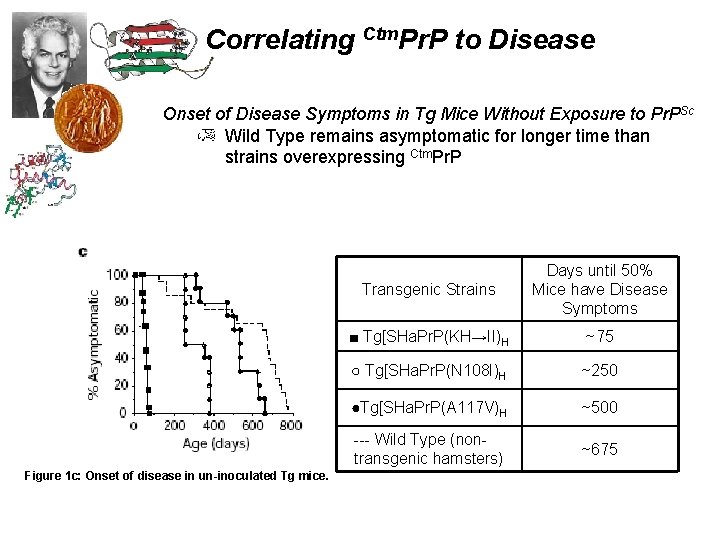
Correlating Ctm. Pr. P to Disease Onset of Disease Symptoms in Tg Mice Without Exposure to Pr. PSc Wild Type remains asymptomatic for longer time than strains overexpressing Ctm. Pr. P Figure 1 c: Onset of disease in un-inoculated Tg mice. Transgenic Strains Days until 50% Mice have Disease Symptoms ■ Tg[SHa. Pr. P(KH→II)H ~75 ○ Tg[SHa. Pr. P(N 108 I)H ~250 ●Tg[SHa. Pr. P(A 117 V)H ~500 --- Wild Type (nontransgenic hamsters) ~675
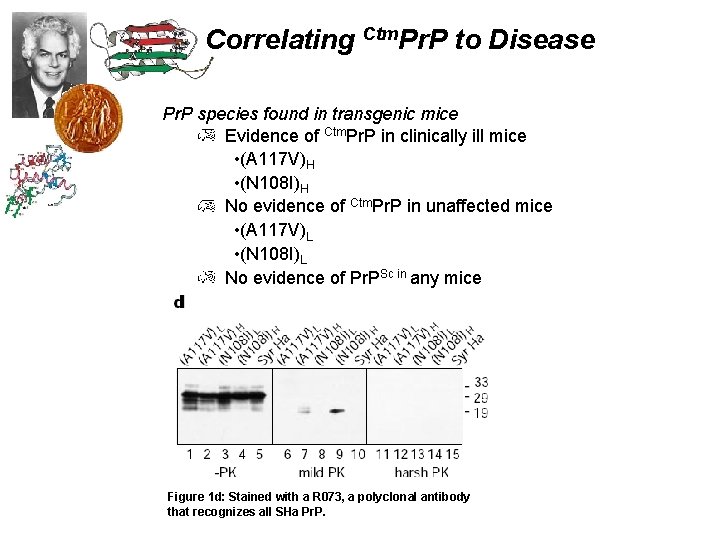
Correlating Ctm. Pr. P to Disease Pr. P species found in transgenic mice Evidence of Ctm. Pr. P in clinically ill mice • (A 117 V)H • (N 108 I)H No evidence of Ctm. Pr. P in unaffected mice • (A 117 V)L • (N 108 I)L No evidence of Pr. PSc in any mice Figure 1 d: Stained with a R 073, a polyclonal antibody that recognizes all SHa Pr. P.
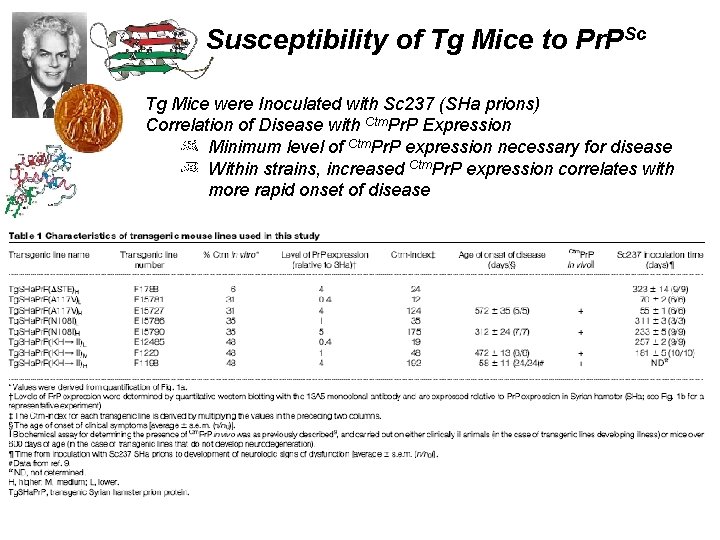
Susceptibility of Tg Mice to Pr. PSc Tg Mice were Inoculated with Sc 237 (SHa prions) Correlation of Disease with Ctm. Pr. P Expression Minimum level of Ctm. Pr. P expression necessary for disease Within strains, increased Ctm. Pr. P expression correlates with more rapid onset of disease
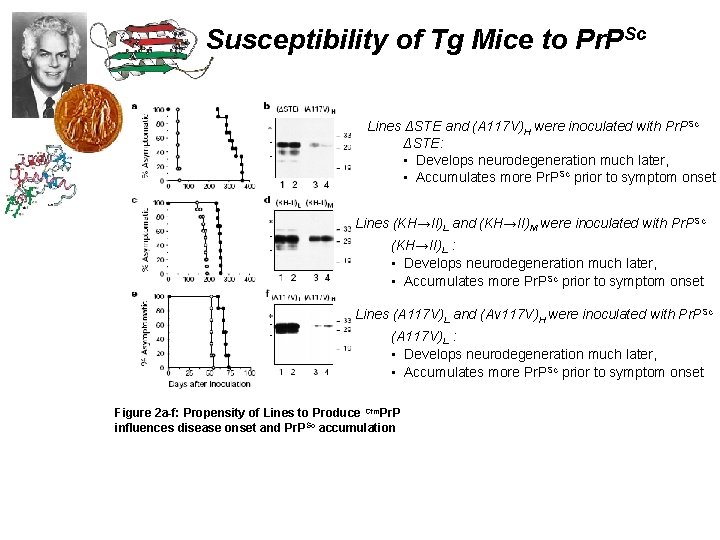
Susceptibility of Tg Mice to Pr. PSc Lines ΔSTE and (A 117 V)H were inoculated with Pr. PSc ΔSTE: • Develops neurodegeneration much later, • Accumulates more Pr. PSc prior to symptom onset Lines (KH→II)L and (KH→II)M were inoculated with Pr. PSc (KH→II)L : • Develops neurodegeneration much later, • Accumulates more Pr. PSc prior to symptom onset Lines (A 117 V)L and (Av 117 V)H were inoculated with Pr. PSc (A 117 V)L : • Develops neurodegeneration much later, • Accumulates more Pr. PSc prior to symptom onset Figure 2 a-f: Propensity of Lines to Produce Ctm. Pr. P influences disease onset and Pr. PSc accumulation
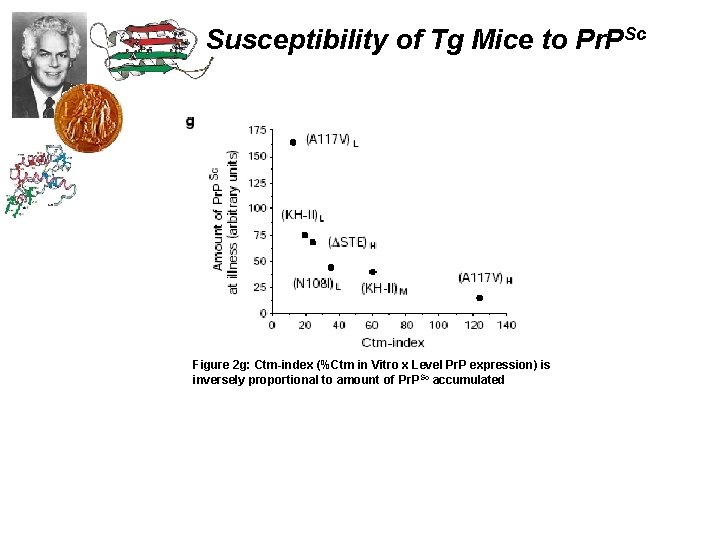
Susceptibility of Tg Mice to Pr. PSc Figure 2 g: Ctm-index (%Ctm in Vitro x Level Pr. P expression) is inversely proportional to amount of Pr. PSc accumulated
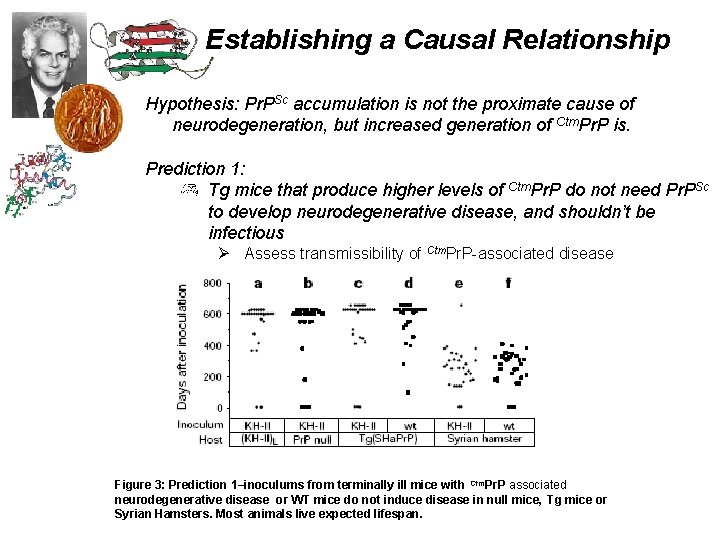
Establishing a Causal Relationship Hypothesis: Pr. PSc accumulation is not the proximate cause of neurodegeneration, but increased generation of Ctm. Pr. P is. Prediction 1: Tg mice that produce higher levels of Ctm. Pr. P do not need Pr. PSc to develop neurodegenerative disease, and shouldn’t be infectious Ø Assess transmissibility of Ctm. Pr. P-associated disease Figure 3: Prediction 1–inoculums from terminally ill mice with Ctm. Pr. P associated neurodegenerative disease or WT mice do not induce disease in null mice, Tg mice or Syrian Hamsters. Most animals live expected lifespan.
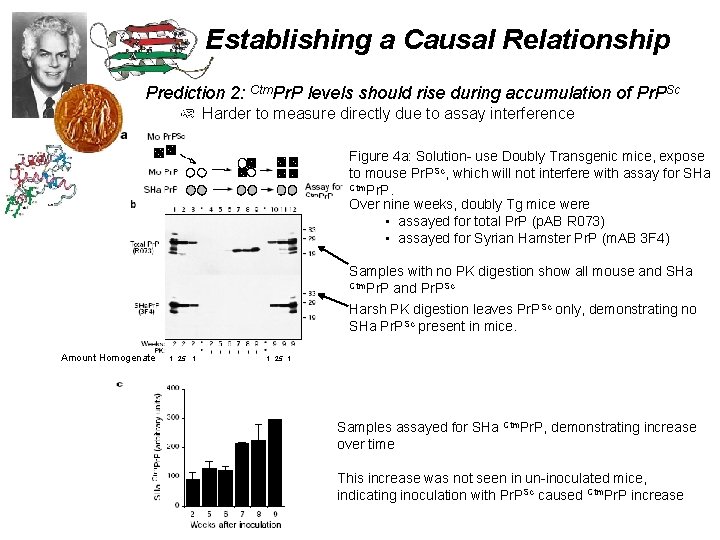
Establishing a Causal Relationship Prediction 2: Ctm. Pr. P levels should rise during accumulation of Pr. PSc Harder to measure directly due to assay interference Figure 4 a: Solution- use Doubly Transgenic mice, expose to mouse Pr. PSc, which will not interfere with assay for SHa Ctm. Pr. P. Over nine weeks, doubly Tg mice were • assayed for total Pr. P (p. AB R 073) • assayed for Syrian Hamster Pr. P (m. AB 3 F 4) Samples with no PK digestion show all mouse and SHa Ctm. Pr. P and Pr. PSc Harsh PK digestion leaves Pr. PSc only, demonstrating no SHa Pr. PSc present in mice. Amount Homogenate: 1. 25. 1 . 1. 25 1 Samples assayed for SHa Ctm. Pr. P, demonstrating increase over time This increase was not seen in un-inoculated mice, indicating inoculation with Pr. PSc caused Ctm. Pr. P increase
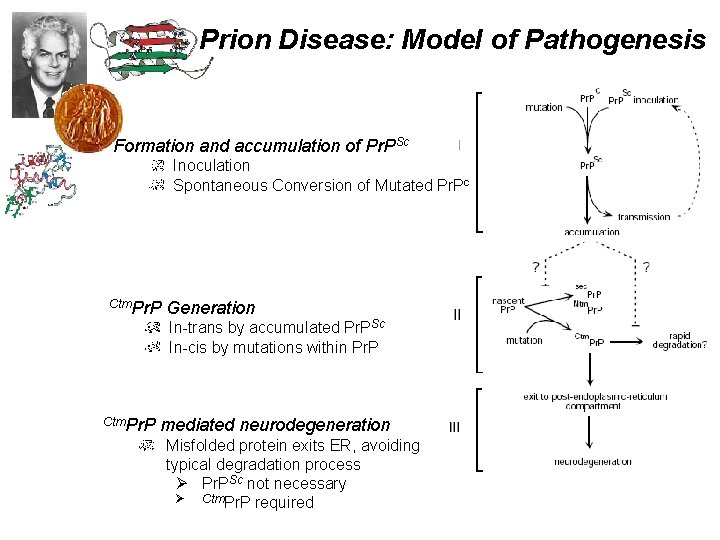
Prion Disease: Model of Pathogenesis Formation and accumulation of Pr. PSc Inoculation Spontaneous Conversion of Mutated Pr. Pc Ctm. Pr. P Generation In-trans by accumulated Pr. PSc In-cis by mutations within Pr. P mediated neurodegeneration Misfolded protein exits ER, avoiding typical degradation process Ø Pr. PSc not necessary Ø Ctm. Pr. P required
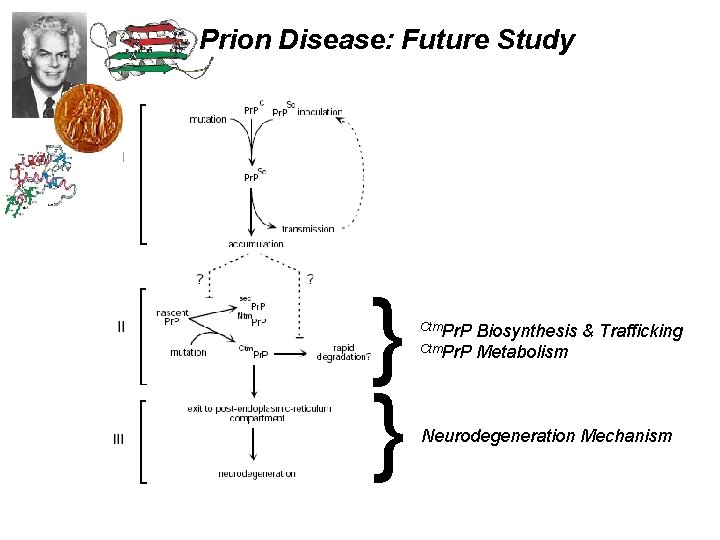
Prion Disease: Future Study } } Ctm. Pr. P Biosynthesis & Trafficking Ctm. Pr. P Metabolism Neurodegeneration Mechanism

Prion Disease: Suggested Readings Horwich, A. L. & Weissman, J. S. (1997). Deadly Conformations. Protein Misfolding in Prion Disease. Cell; 89: 499 -510. Prusiner, S. B. (1998). Prions. Proceedings of the National Academy of Sciences, USA; 95: 13363 -13383.

- Slides: 19