Proteins AP Biology Proteins Multipurpose molecules AP Biology

Proteins AP Biology
Proteins Multipurpose molecules AP Biology 2008 -2009

Why are Proteins Most structurally & functionally diverse group? Function: involved in almost everything u u u u AP Biology enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (hemoglobin, aquaporin) cell communication signals (insulin & other hormones) receptors defense (antibodies) movement (actin & myosin) storage (bean seed proteins)
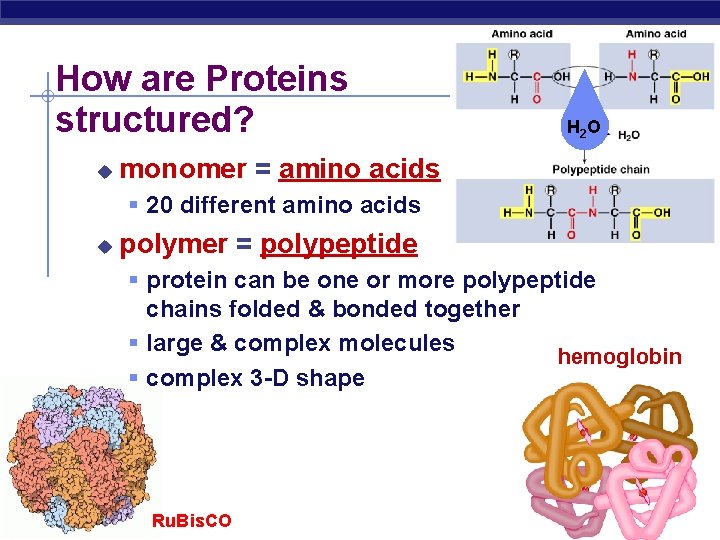
How are Proteins structured? u H 2 O monomer = amino acids 20 different amino acids u polymer = polypeptide protein can be one or more polypeptide chains folded & bonded together large & complex molecules hemoglobin complex 3 -D shape AP Biology Ru. Bis. CO
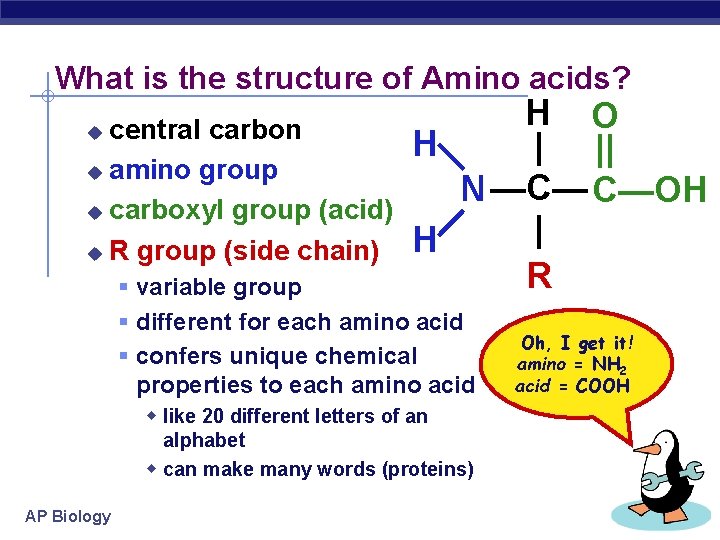
What is the structure of Amino acids? central carbon u amino group u carboxyl group (acid) u R group (side chain) u H O H | || —C— C—OH —N— | H R variable group different for each amino acid confers unique chemical properties to each amino acid w like 20 different letters of an alphabet w can make many words (proteins) AP Biology Oh, I get it! amino = NH 2 acid = COOH
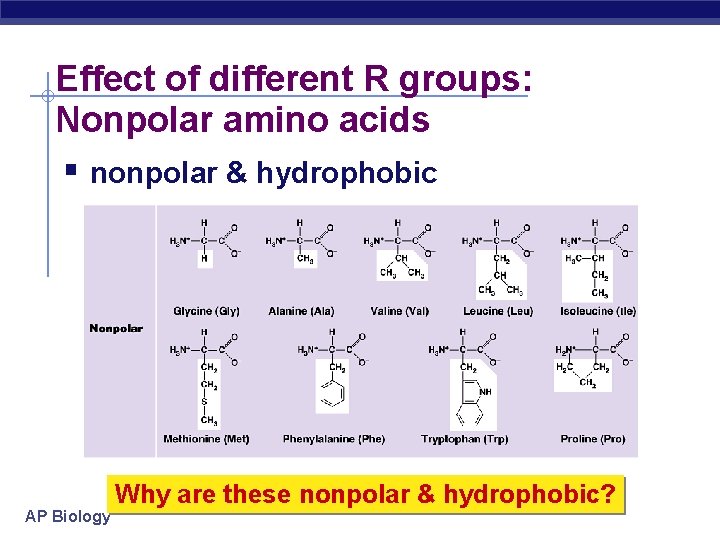
Effect of different R groups: Nonpolar amino acids nonpolar & hydrophobic AP Biology Why are these nonpolar & hydrophobic?

Effect of different R groups: Polar amino acids polar or charged & hydrophilic AP Biology Why are these polar & hydrophillic?

. Ionize in cellular waters by donating H+ AP Biology

Ionizing in cellular waters AP Biology H+ acceptors Acid/Base
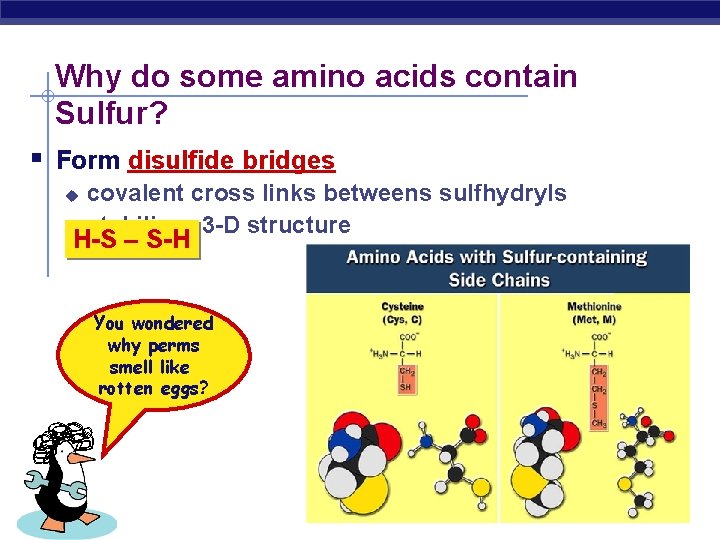
Why do some amino acids contain Sulfur? Form disulfide bridges u u covalent cross links betweens sulfhydryls stabilizes 3 -D structure H-S – S-H You wondered why perms smell like rotten eggs? AP Biology
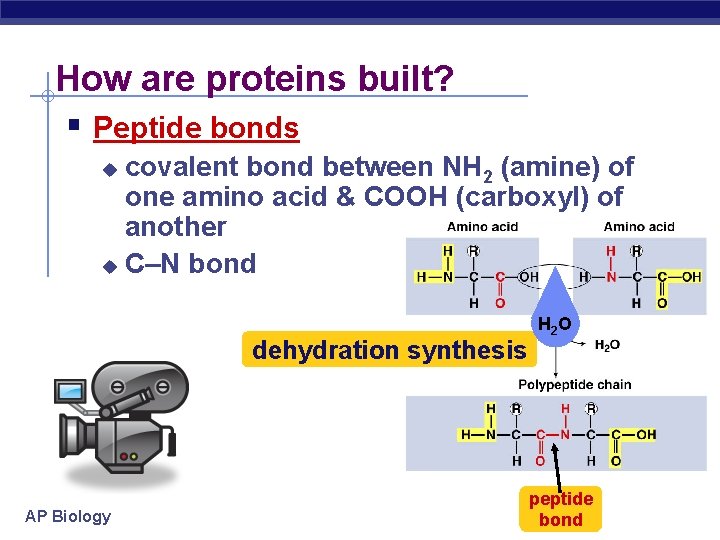
How are proteins built? Peptide bonds covalent bond between NH 2 (amine) of one amino acid & COOH (carboxyl) of another u C–N bond u dehydration synthesis AP Biology H 2 O peptide bond
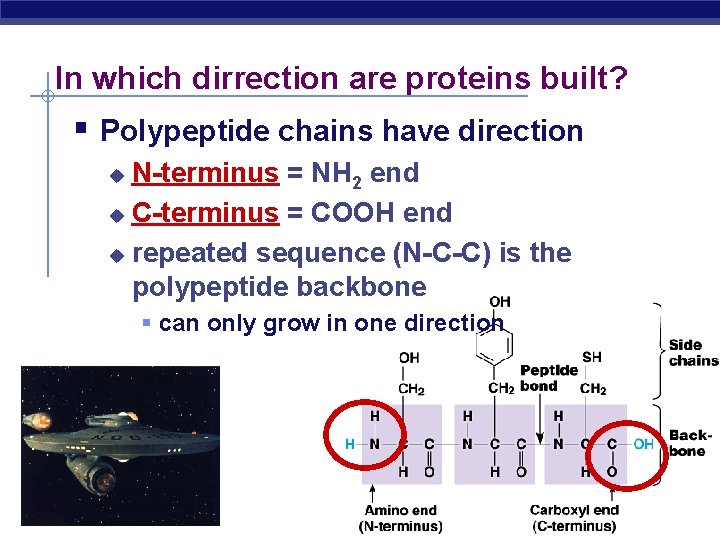
In which dirrection are proteins built? Polypeptide chains have direction N-terminus = NH 2 end u C-terminus = COOH end u repeated sequence (N-C-C) is the polypeptide backbone u can only grow in one direction AP Biology
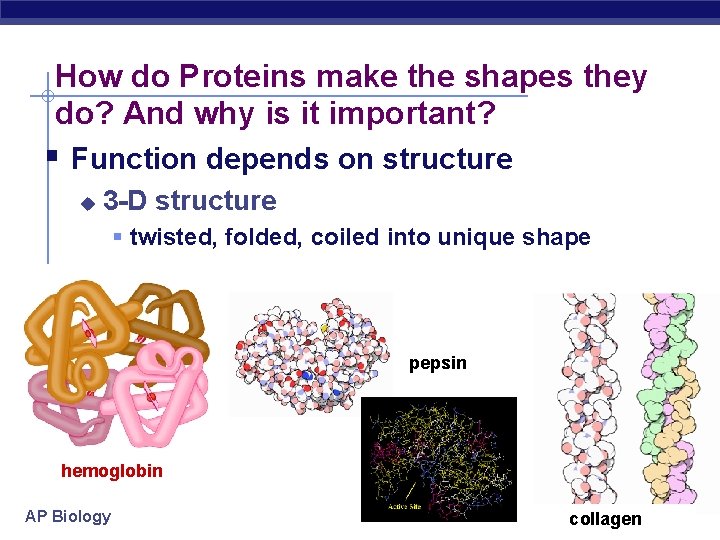
How do Proteins make the shapes they do? And why is it important? Function depends on structure u 3 -D structure twisted, folded, coiled into unique shape pepsin hemoglobin AP Biology collagen
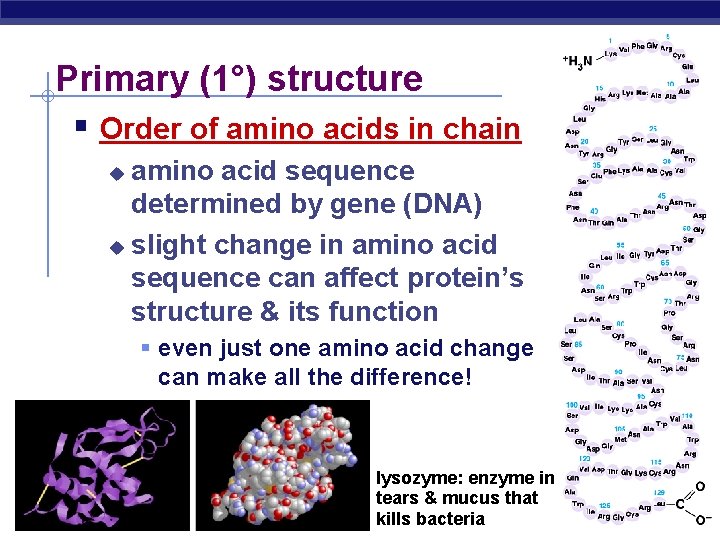
Primary (1°) structure Order of amino acids in chain amino acid sequence determined by gene (DNA) u slight change in amino acid sequence can affect protein’s structure & its function u even just one amino acid change can make all the difference! AP Biology lysozyme: enzyme in tears & mucus that kills bacteria
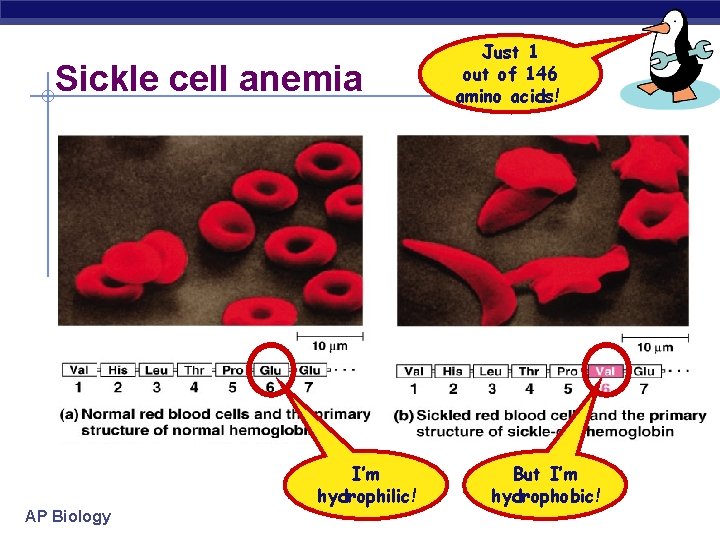
Sickle cell anemia I’m hydrophilic! AP Biology Just 1 out of 146 amino acids! But I’m hydrophobic!
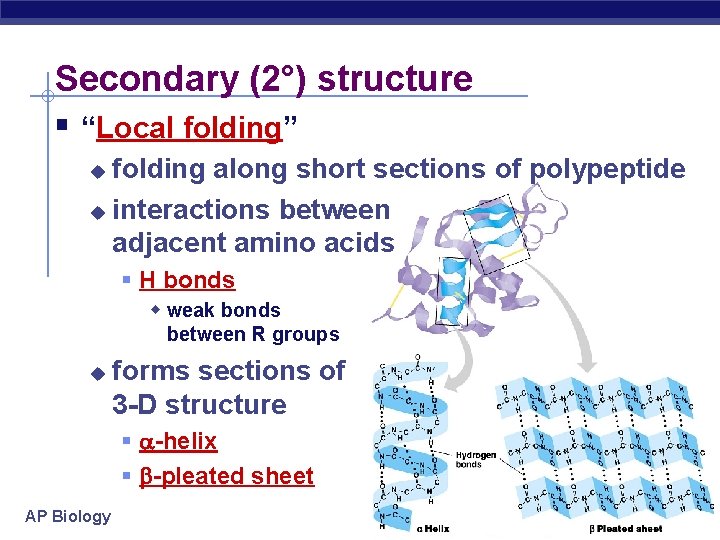
Secondary (2°) structure “Local folding” folding along short sections of polypeptide u interactions between adjacent amino acids u H bonds w weak bonds between R groups u forms sections of 3 -D structure -helix -pleated sheet AP Biology
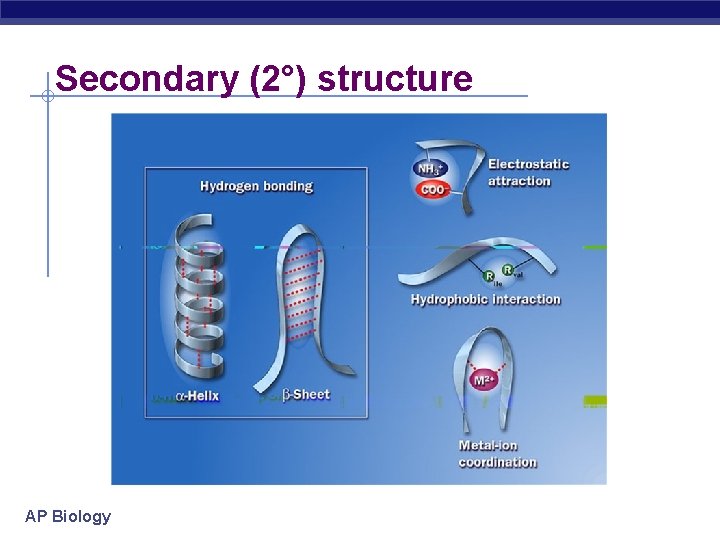
Secondary (2°) structure AP Biology
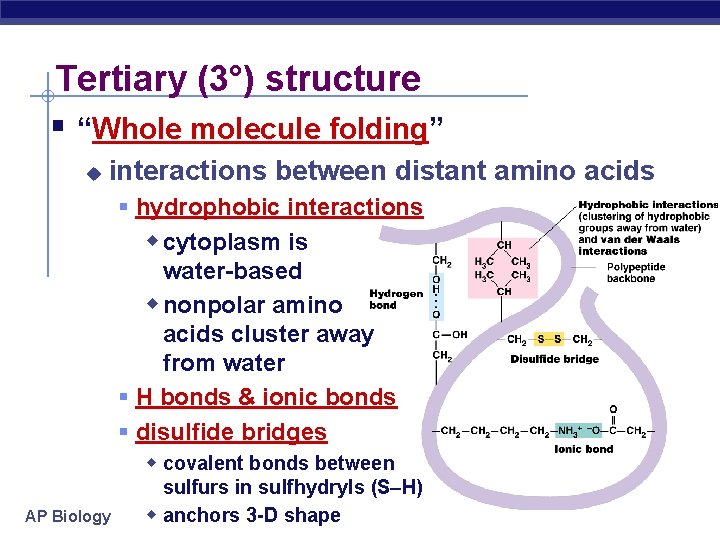
Tertiary (3°) structure “Whole molecule folding” u interactions between distant amino acids hydrophobic interactions w cytoplasm is water-based w nonpolar amino acids cluster away from water H bonds & ionic bonds disulfide bridges w covalent bonds between AP Biology sulfurs in sulfhydryls (S–H) w anchors 3 -D shape
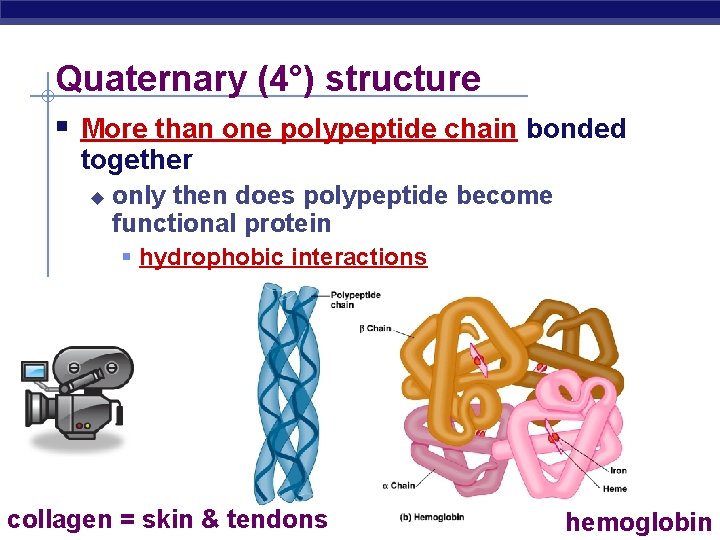
Quaternary (4°) structure More than one polypeptide chain bonded together u only then does polypeptide become functional protein hydrophobic interactions AP Biology = skin & tendons collagen hemoglobin
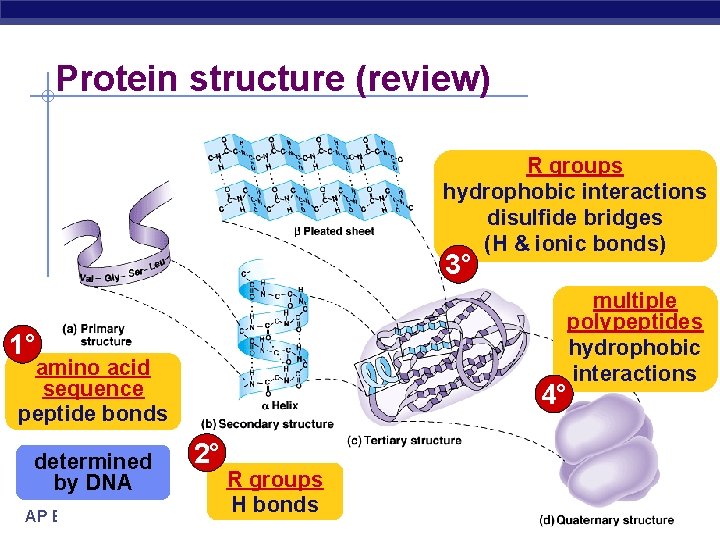
Protein structure (review) R groups hydrophobic interactions disulfide bridges (H & ionic bonds) 3° multiple polypeptides hydrophobic interactions 1° amino acid sequence peptide bonds determined by DNA AP Biology 4° 2° R groups H bonds

Protein denaturation Unfolding a protein u In Biology, size doesn’t matter, SHAPE matters! conditions that disrupt H bonds, ionic bonds, disulfide bridges temperature p. H salinity u alter 2° & 3° structure alter 3 -D shape u destroys functionality some proteins can return to their functional shape after denaturation, many cannot AP Biology

EAT Let’s build X some Proteins! AP Biology 2008 -2009

Ghosts of Lectures Past (storage) AP Biology 2007 -2008
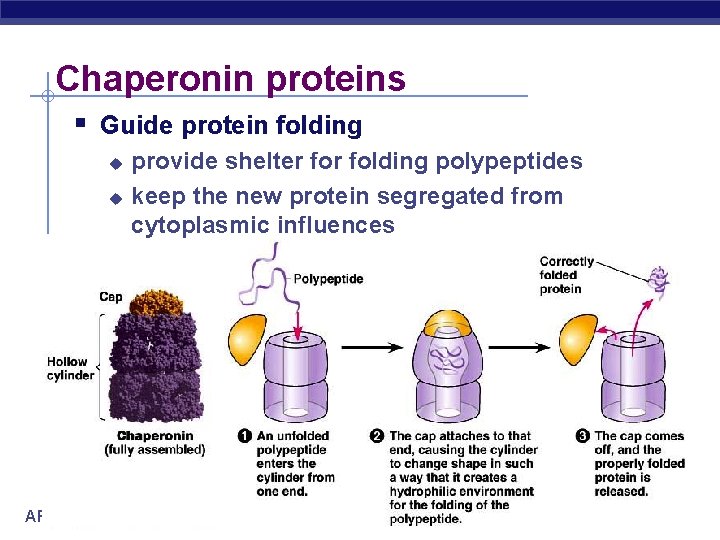
Chaperonin proteins Guide protein folding u u AP Biology provide shelter folding polypeptides keep the new protein segregated from cytoplasmic influences
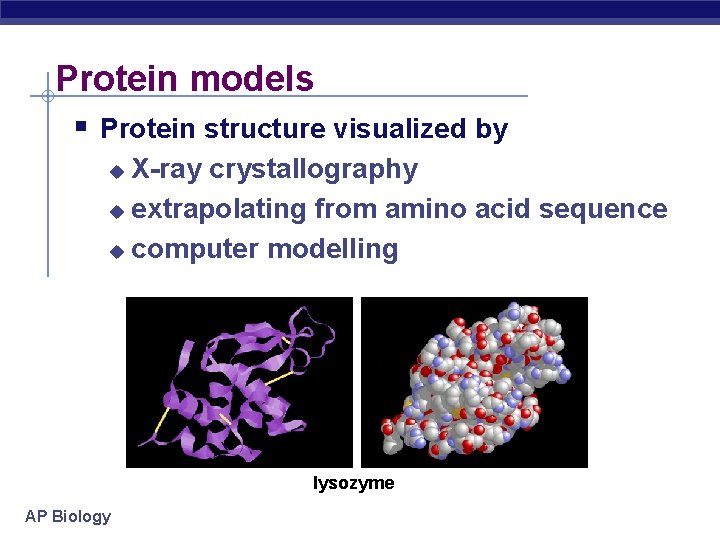
Protein models Protein structure visualized by X-ray crystallography u extrapolating from amino acid sequence u computer modelling u lysozyme AP Biology
- Slides: 25