Proteins AP Biology Proteins Most structurally and functionally
Proteins AP Biology

Proteins § Most structurally and functionally diverse group of biomolecules § Function Involved in almost everything Enzymes Structure (keratin, collagen) Carriers and transport (membrane channels) Receptors and binding (defense) Contraction ( actin and myosin) Signaling (hormones) Storage (bean seed proteins) AP Biology

Proteins § Structure Monomer = amino acids 20 different amino acids Polymer = polypeptide Protein can be one or more polypeptide chains folded and bonded together Large and complex molecules Complex 3 D shape AP Biology
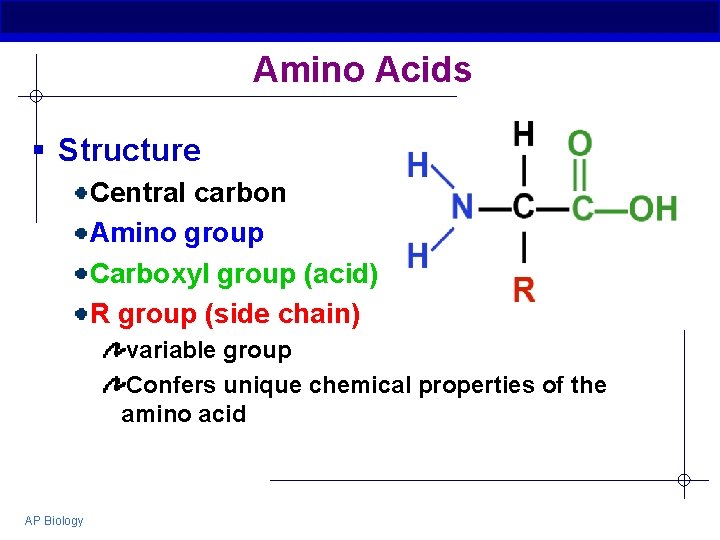
Amino Acids § Structure Central carbon Amino group Carboxyl group (acid) R group (side chain) variable group Confers unique chemical properties of the amino acid AP Biology

Nonpolar amino acids § Nonpolar and hydrophobic AP Biology

Polar amino acids § Polar or charged and hydrophilic AP Biology
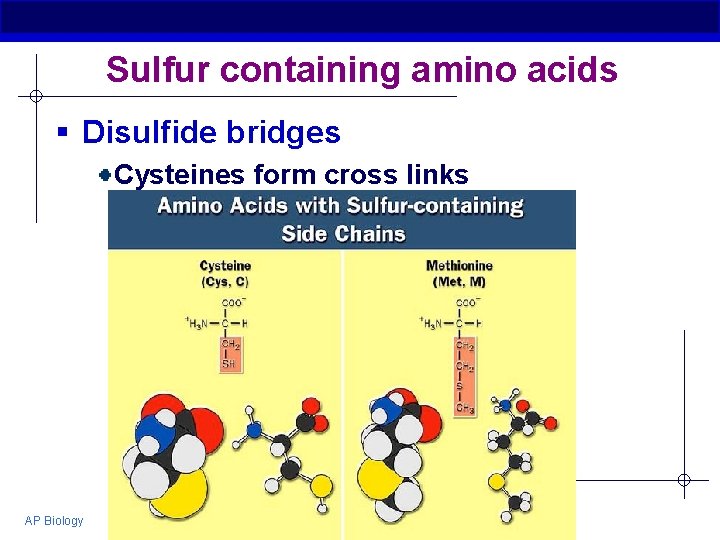
Sulfur containing amino acids § Disulfide bridges Cysteines form cross links AP Biology
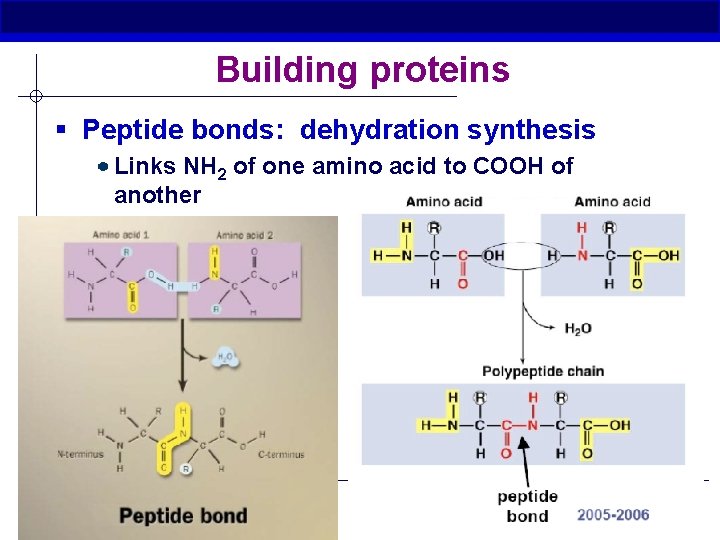
Building proteins § Peptide bonds: dehydration synthesis Links NH 2 of one amino acid to COOH of another C-N bond AP Biology Minzenmayer 2005 -2006
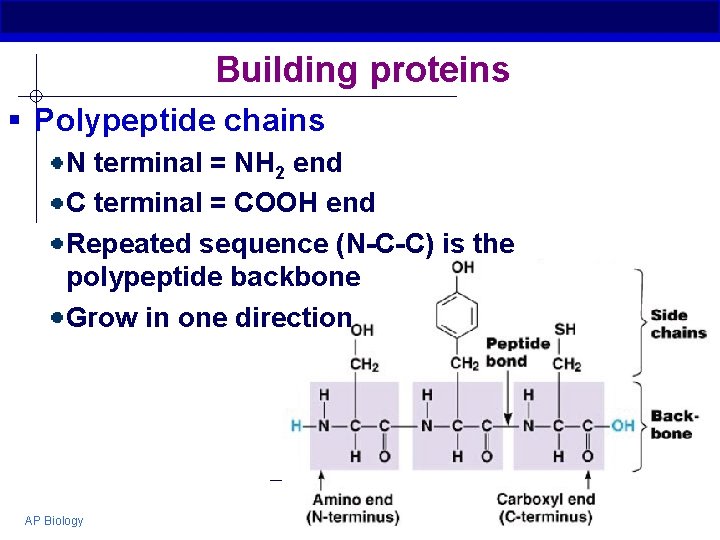
Building proteins § Polypeptide chains N terminal = NH 2 end C terminal = COOH end Repeated sequence (N-C-C) is the polypeptide backbone Grow in one direction AP Biology
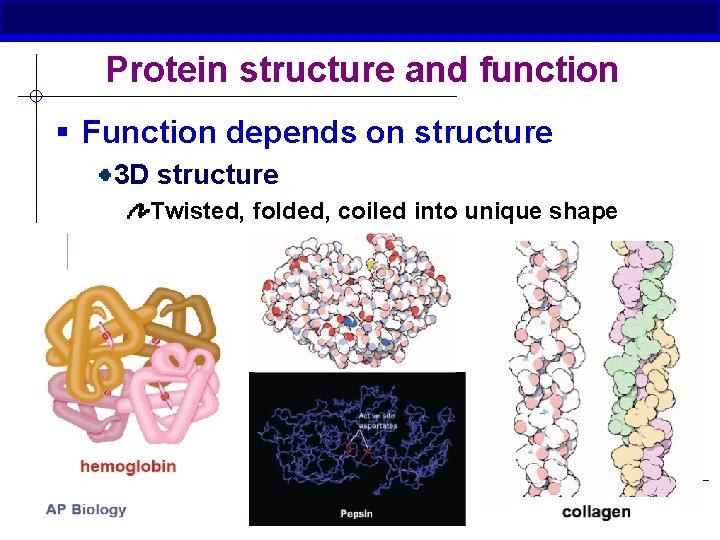
Protein structure and function § Function depends on structure 3 D structure Twisted, folded, coiled into unique shape AP Biology
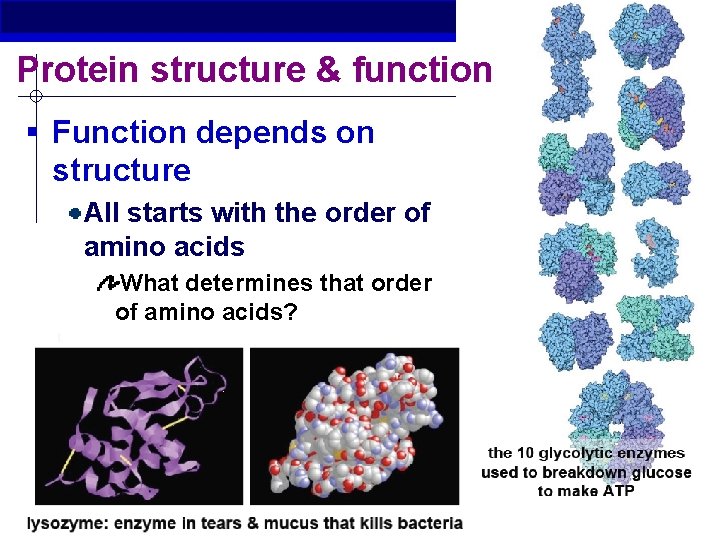
Protein structure & function § Function depends on structure All starts with the order of amino acids What determines that order of amino acids? AP Biology
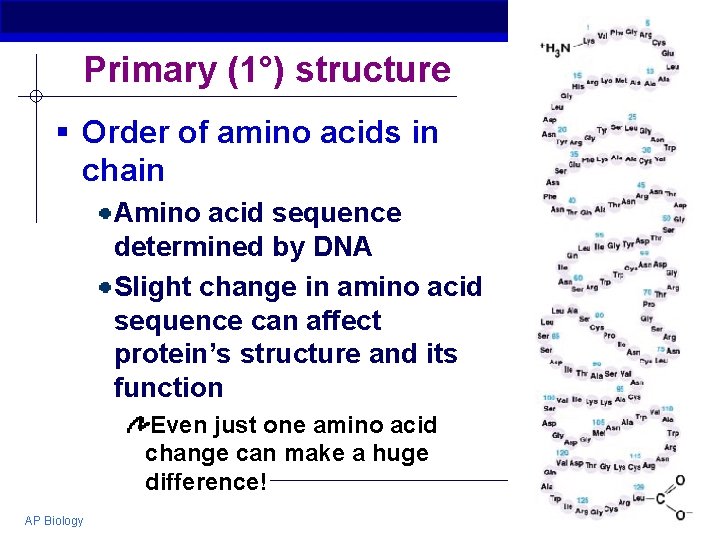
Primary (1°) structure § Order of amino acids in chain Amino acid sequence determined by DNA Slight change in amino acid sequence can affect protein’s structure and its function Even just one amino acid change can make a huge difference! AP Biology
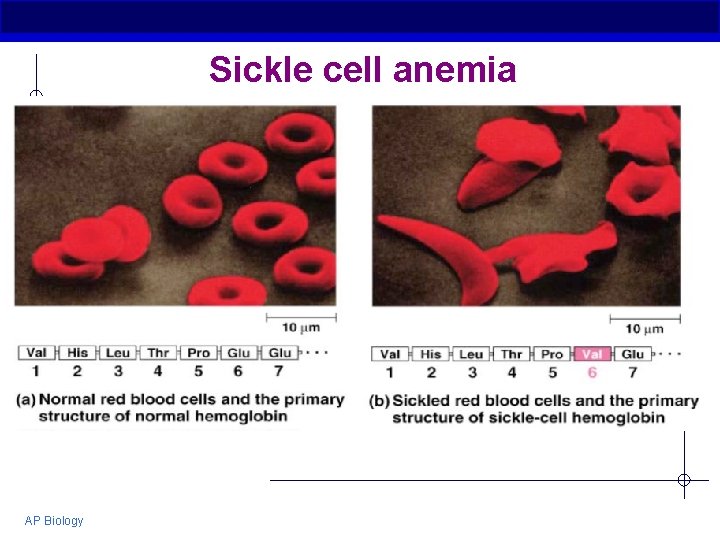
Sickle cell anemia AP Biology
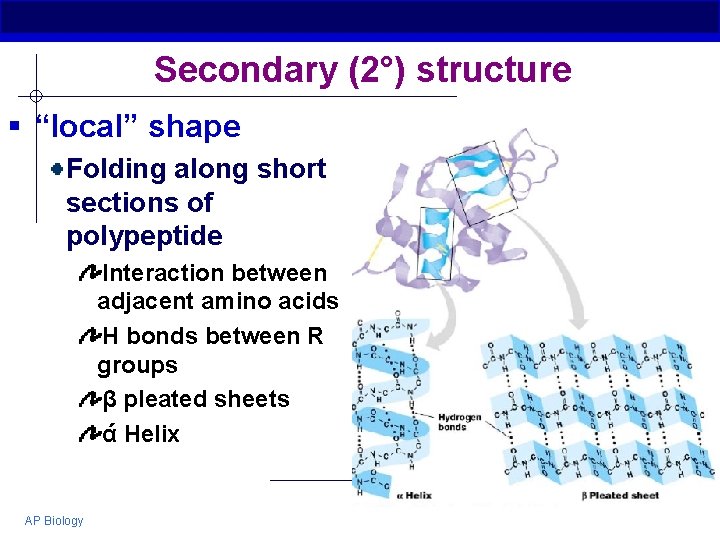
Secondary (2°) structure § “local” shape Folding along short sections of polypeptide Interaction between adjacent amino acids H bonds between R groups β pleated sheets ά Helix AP Biology

Fig. 5 -21 d Abdominal glands of the spider secrete silk fibers made of a structural protein containing pleated sheets. The radiating strands, made of dry silk fibers, maintain the shape of the web. The spiral strands (capture strands) are elastic, stretching in response to wind, rain, and the touch of insects.
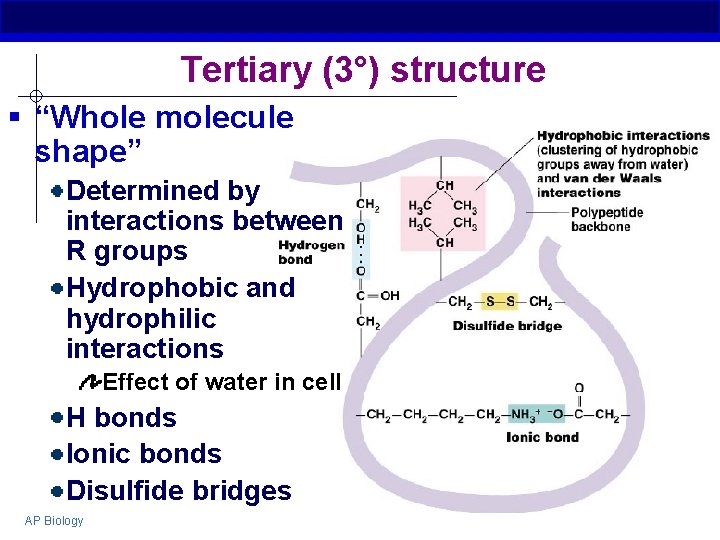
Tertiary (3°) structure § “Whole molecule shape” Determined by interactions between R groups Hydrophobic and hydrophilic interactions Effect of water in cell H bonds Ionic bonds Disulfide bridges AP Biology
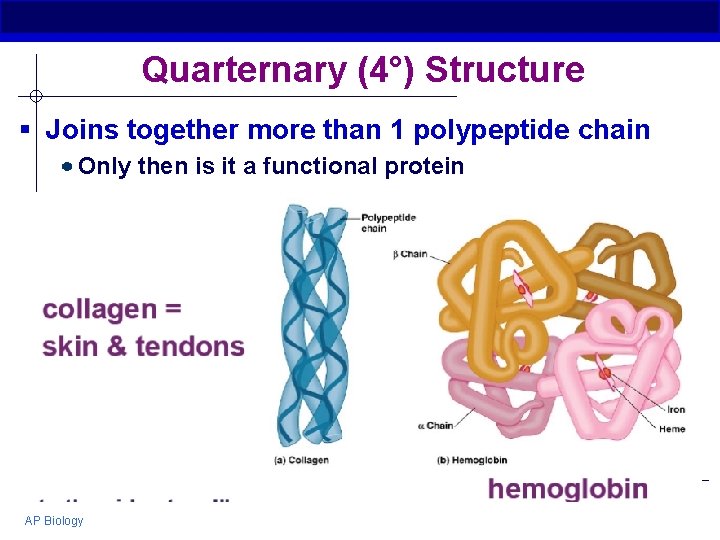
Quarternary (4°) Structure § Joins together more than 1 polypeptide chain Only then is it a functional protein AP Biology
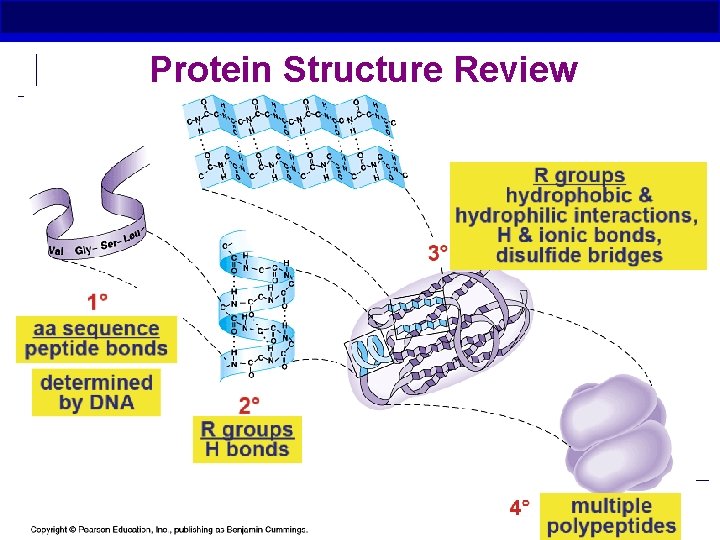
Protein Structure Review AP Biology
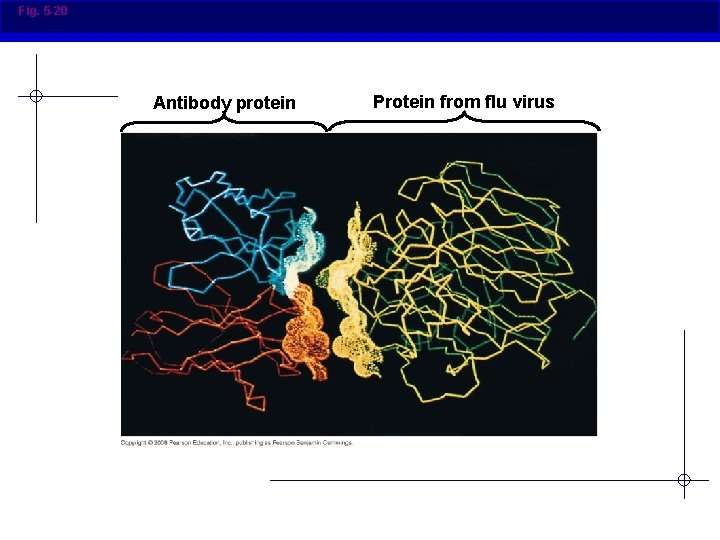
Fig. 5 -20 Antibody protein Protein from flu virus
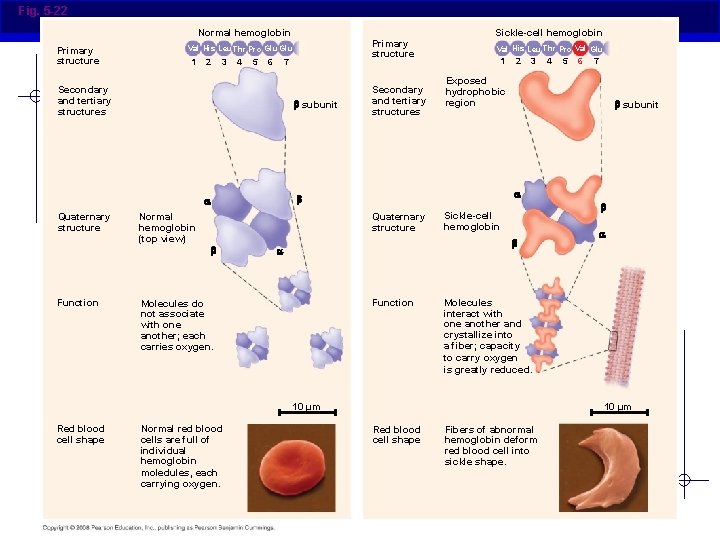
Fig. 5 -22 Normal hemoglobin Primary structure 1 2 3 4 5 6 7 Secondary and tertiary structures subunit Function Normal hemoglobin (top view) Secondary and tertiary structures Val His Leu Thr Pro Val Glu 1 2 3 Normal red blood cells are full of individual hemoglobin moledules, each carrying oxygen. 6 7 subunit Sickle-cell hemoglobin Function Molecules interact with one another and crystallize into a fiber; capacity to carry oxygen is greatly reduced. 10 µm Red blood cell shape 5 Exposed hydrophobic region Molecules do not associate with one another; each carries oxygen. 4 Quaternary structure Sickle-cell hemoglobin Quaternary structure Primary structure Val His Leu Thr Pro Glu 10 µm Red blood cell shape Fibers of abnormal hemoglobin deform red blood cell into sickle shape.
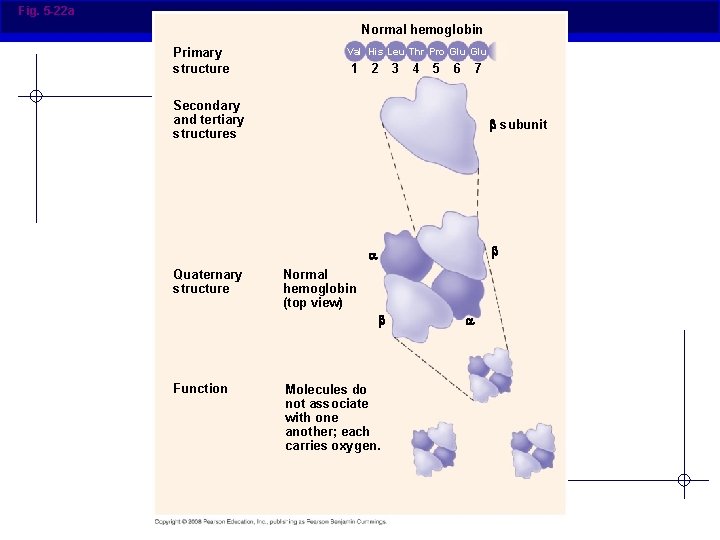
Fig. 5 -22 a Normal hemoglobin Primary structure Val His Leu Thr Pro Glu 1 2 3 4 5 7 6 Secondary and tertiary structures subunit Quaternary structure Normal hemoglobin (top view) Function Molecules do not associate with one another; each carries oxygen.
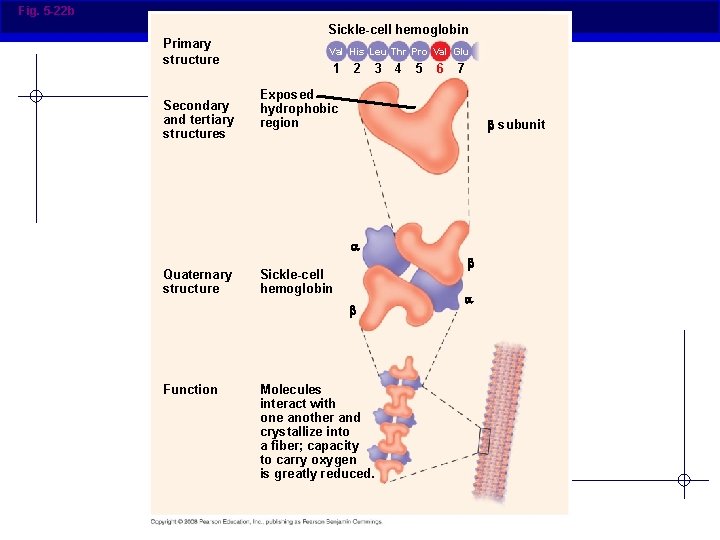
Fig. 5 -22 b Primary structure Secondary and tertiary structures Sickle-cell hemoglobin Val His Leu Thr Pro Val Glu 1 2 3 4 5 6 7 Exposed hydrophobic region subunit Quaternary structure Sickle-cell hemoglobin Function Molecules interact with one another and crystallize into a fiber; capacity to carry oxygen is greatly reduced.
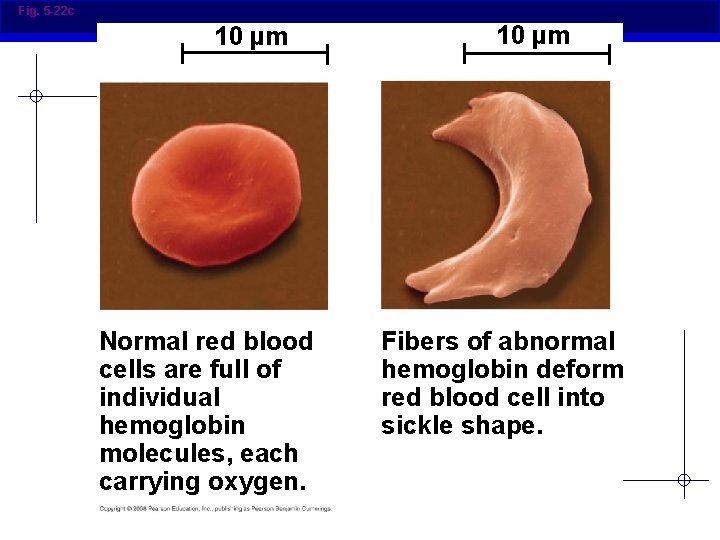
Fig. 5 -22 c 10 µm Normal red blood cells are full of individual hemoglobin molecules, each carrying oxygen. 10 µm Fibers of abnormal hemoglobin deform red blood cell into sickle shape.
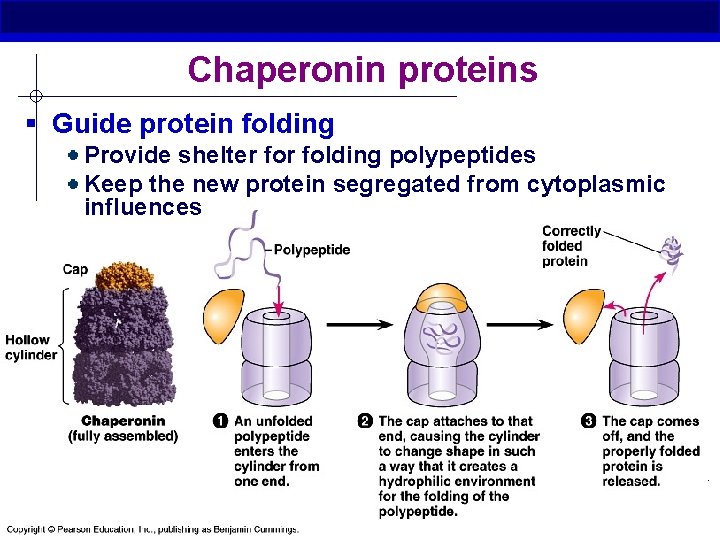
Chaperonin proteins § Guide protein folding Provide shelter folding polypeptides Keep the new protein segregated from cytoplasmic influences AP Biology
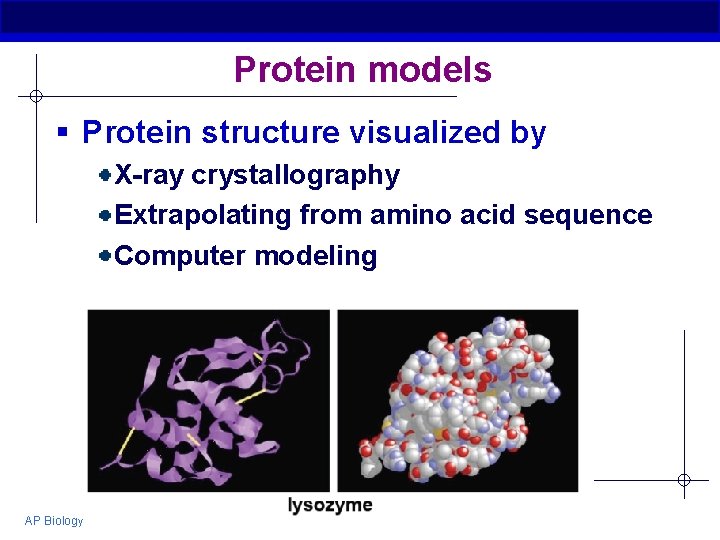
Protein models § Protein structure visualized by X-ray crystallography Extrapolating from amino acid sequence Computer modeling AP Biology

Denaturing Proteins § Disrupt 3° structure p. H Temperature Salt § Unravel or denature protein § Disrupts H bonds, ionic bonds and disulfide bridges § Some proteins can return to their functional shape after denaturation, many cannot AP Biology
- Slides: 26