Proteins 2 Biological importance of proteins Required for

Proteins 2

Biological importance of proteins • Required for building and repair of body tissues (including muscle) • Enzymes, hormones, and many immune molecules are proteins • Essential body processes such as water balancing, nutrient transport, and muscle contractions require protein to function. • Protein is a source of energy. • Protein helps keep skin, hair, and nails healthy. • Protein, like most other essential nutrients, is absolutely crucial for overall good health.

Biological importance of proteins • Antibodies - are specialized proteins involved in defending the body from antigens (foreign invaders). Contractile Proteins - are responsible for movement. Examples include actin and myosin. These proteins are involved in muscle contraction and movement. Enzymes - are proteins that facilitate biochemical reactions. They are often referred to as catalysts because they speed up chemical reactions.

Biological importance of proteins • Hormonal Proteins - are messenger proteins which help to coordinate certain bodily activities. Structural Proteins - are fibrous and stringy and provide support. Examples include keratin, collagen, and elastin. Keratins strengthen protective coverings such as hair, quills, feathers, horns, and beaks. Collagens and elastin provide support for connective tissues such as tendons and ligaments. Storage Proteins - store amino acids. Examples include ovalbumin and casein.

1 - Simple proteins These are proteins which on hydrolysis give rise to amino acids only. 2 - Conjugated proteins 3 - Derived proteins
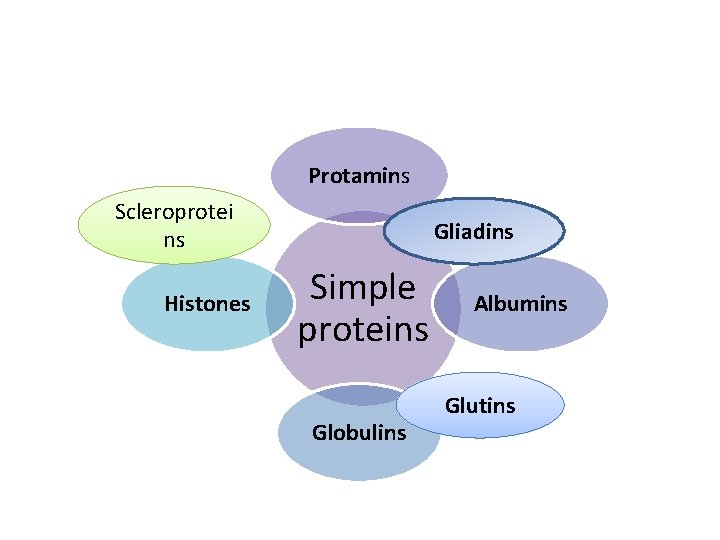
Protamins Scleroprotei ns Histones Gliadins Simple proteins Globulins Albumins Glutins

Albumins Albumin is widely distributed in nature. They are usually associated with globulins. It gives positive heat coagulation test. It is soluble in distilled water, also soluble in very diluted salt solutions, alkalies and acids. • Albumins are proteins of high biological value, missing non of the essential AA. • Ex: serum albumin, lactalbumin of milk, ovalbumin of egg. • •

• Conjugated proteins are characterised by giving free amino acids and nonproteinic groups (prosthetic group). On hydrolysis. • The prosthetic groups are different and accirding to this difference proteins may be divided into: • Phosphoproteins: Phosphoric acid • Glycoproteins: Carbohydrates • Chromoproteins: Pigments

• Phospholipids are conjugated proteins in which phosphourus is present in a combined form. • The combination is usually in the ester form, the phosphoric acid combines with the OH group of serine (phoserine). - Phosphoproteins are insoluble in distilled water, saline , dil acids, but soluble in dilute alkaline solution.
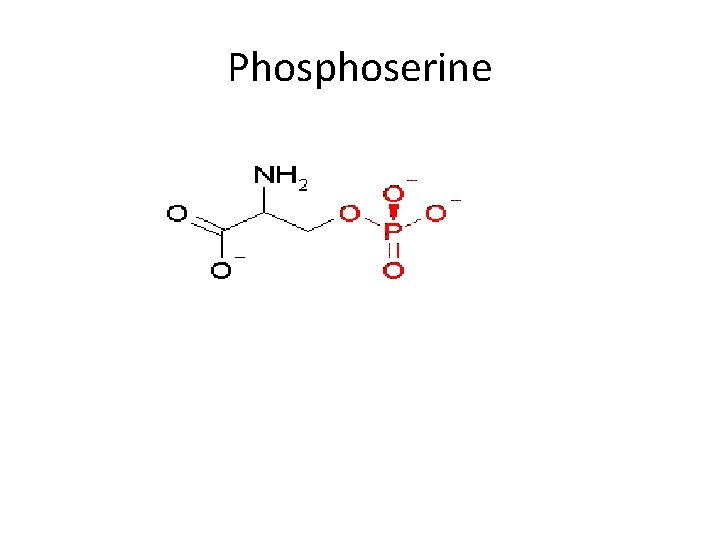
Phosphoserine

Derived proteins • The derived proteins result from the denaturation or cleavage of the native proteins by acids, alkalies or enzymes. • Derived proteins include: • Proteans • Denaturated proteins • Metaproteins • Proteases • Peptones

Derived Proteins • Proteans: they are insoluble intial decompostition products of proteins. • They result from the action of various reagents such as diluted acids or enzymes on proteins. • Peptone: It is the end products of digestion of proteins by pepsin of the stomach. • Peptones are soluble in water, saline, diluted acids and diluted alkalies.

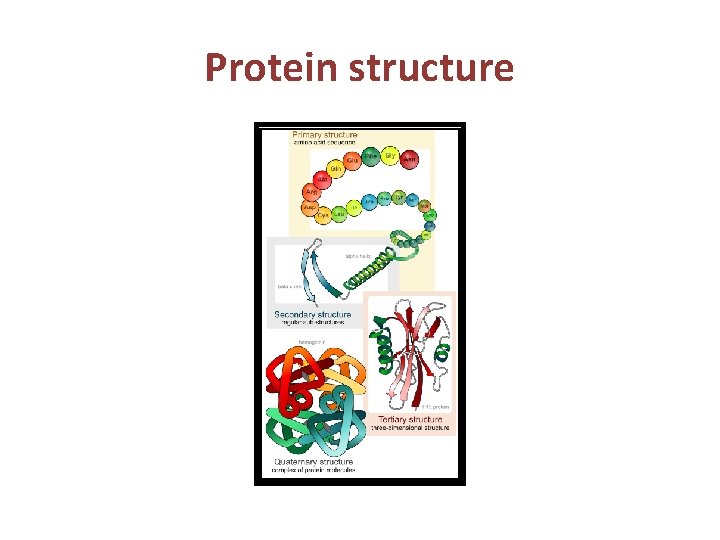
Protein Structure • Peptide bond formation • Primary structure of protein • Secondry structure of protein • Tertiary structure of protein • Quaternary structure of protein
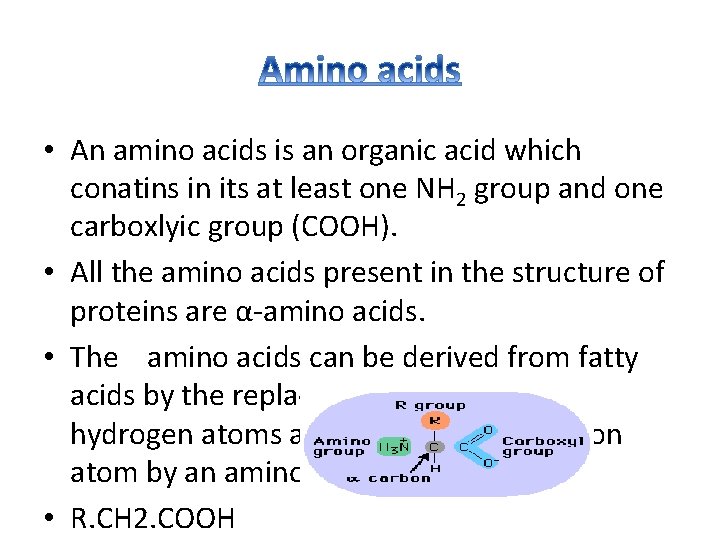
• An amino acids is an organic acid which conatins in its at least one NH 2 group and one carboxlyic group (COOH). • All the amino acids present in the structure of proteins are α-amino acids. • The amino acids can be derived from fatty acids by the replacement of one of the hydrogen atoms attached to the α- carbon atom by an amino group. • R. CH 2. COOH

Peptide bond formation • The alph amino acids polymerize, through the elimination of a water molecule. • The resulting CO____NH linkage is known as a peptide bond. • Polymers composed of two, three, (3 -10) called peptide units, It is known as dipeptides, tripeptides, oligopeptides and polypeptides.
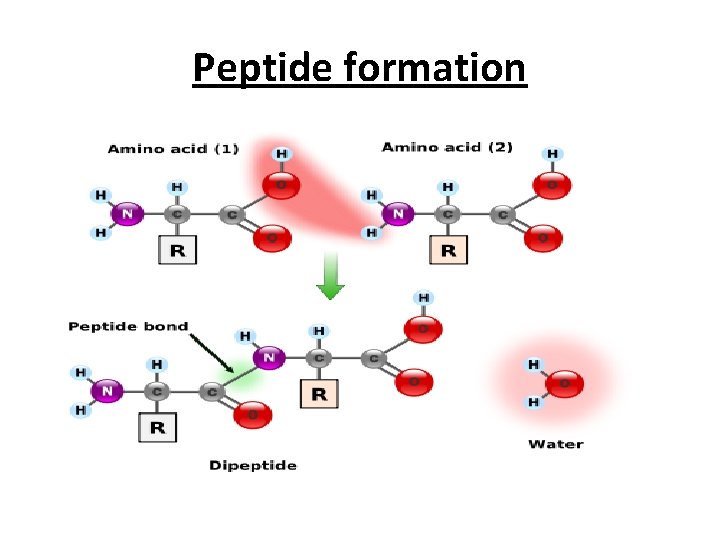
Peptide formation

Primary Structure of Proteins • The primary structure of peptides and proteins refers to the linear number and order of the amino acids present. • The primary structure is held together by covalent or peptide bonds, which are made during the process of protein biosynthesis or translation. • The two ends of the polypeptide chain are referred to as the carboxyl terminus (C-terminus) and the amino terminus (N-terminus) based on the nature of the free group on each extremity. The primary structure of a protein is determined by the gene corresponding to the protein
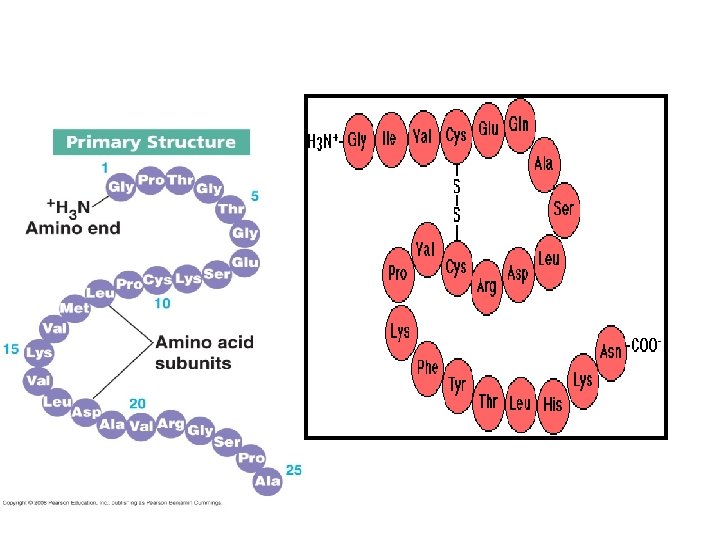

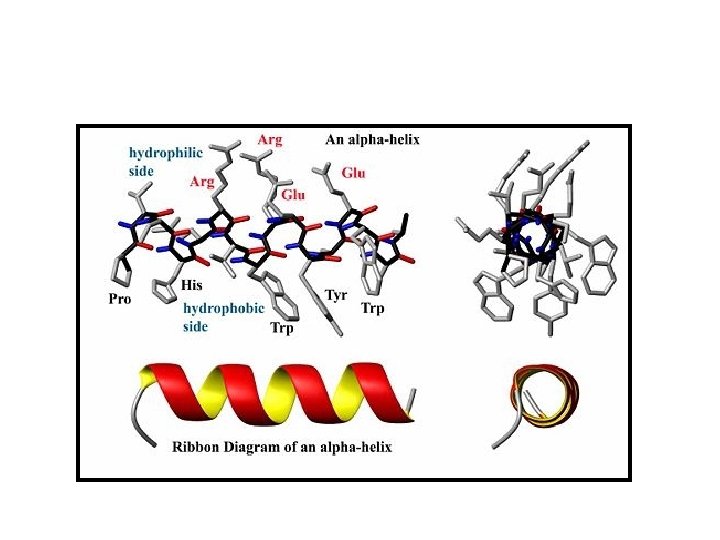
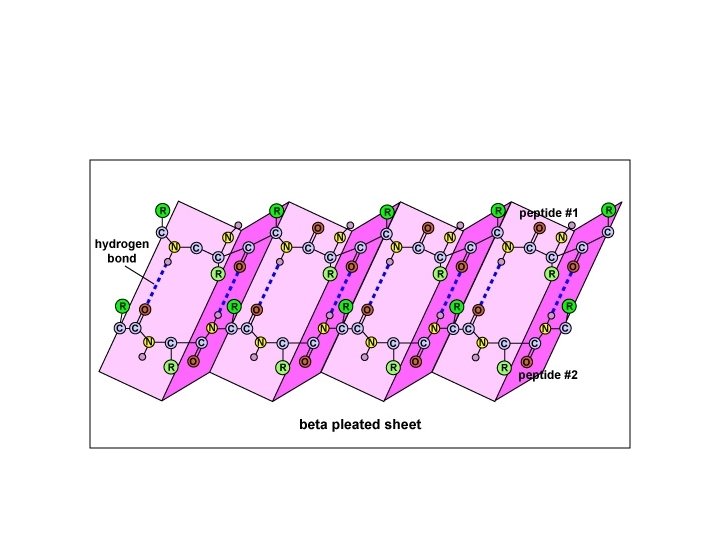
SECONDARY STRUCTURE • Secondary structure refers to highly regular local sub-structures. Two main types of secondary structure, the alpha helix and the beta strand, • These secondary structures are defined by patterns of hydrogen bonds between the mainchain peptide groups. • Both the alpha helix and the beta-sheet represent a way of saturating all the hydrogen bond donors and acceptors in the peptide backbone. Some parts of the protein are ordered but do not form any regular structures. • In general proteins fold into two broad classes of structure termed, globular proteins or fibrous


Tertiary Structure of Proteins • Tertiary structure refers to the complete three -dimensional structure of the polypeptide units of a given protein. Included in this description is the spatial relationship of different secondary structures to one another within a polypeptide chain and how these secondary structures themselves fold into the three-dimensional form of the protein. • The interactions of different domains is governed by several forces: These include
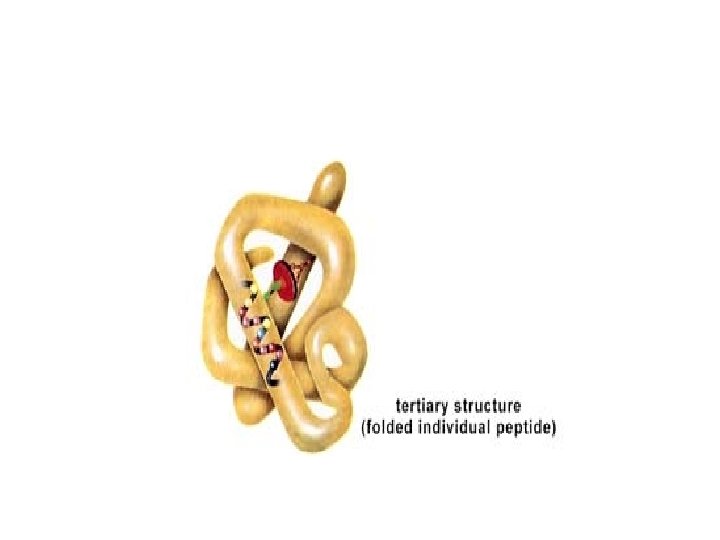
Protein structure
- Slides: 25