Protein Structure FDSC 400 Protein Functions Biological Food

Protein Structure FDSC 400

Protein Functions • Biological? • Food?
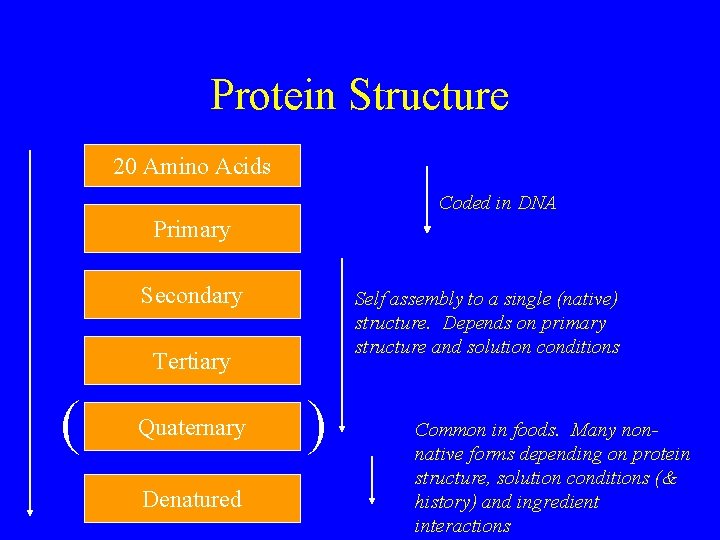
Protein Structure 20 Amino Acids Coded in DNA Primary Secondary Self assembly to a single (native) structure. Depends on primary structure and solution conditions Tertiary ( Quaternary Denatured ) Common in foods. Many nonnative forms depending on protein structure, solution conditions (& history) and ingredient interactions
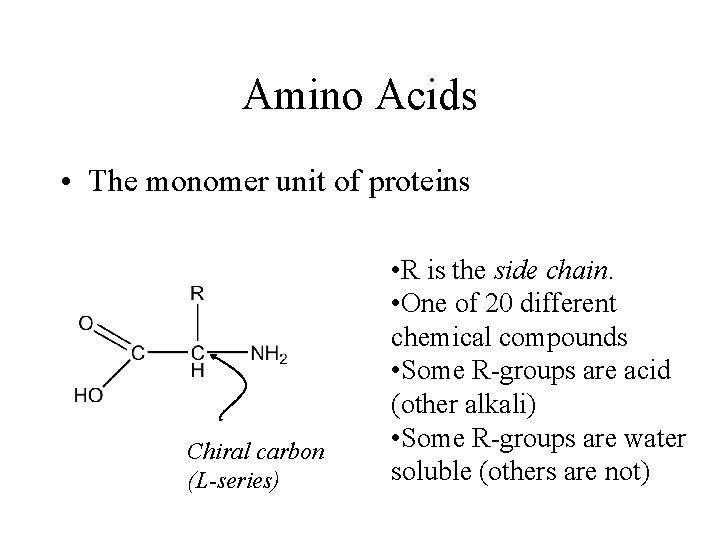
Amino Acids • The monomer unit of proteins Chiral carbon (L-series) • R is the side chain. • One of 20 different chemical compounds • Some R-groups are acid (other alkali) • Some R-groups are water soluble (others are not)

Amino Acids Polar • Uncharged. Ser, Thr, Asn, Gln, Cys Non-Polar • Aliphatic. Ala, Ile, Leu, Met, Pro, Val • Positive (basic). Arg, • Aromatic. Phe, Trp, Lys, His Tyr • Negative (acidic). Asp, Glu,
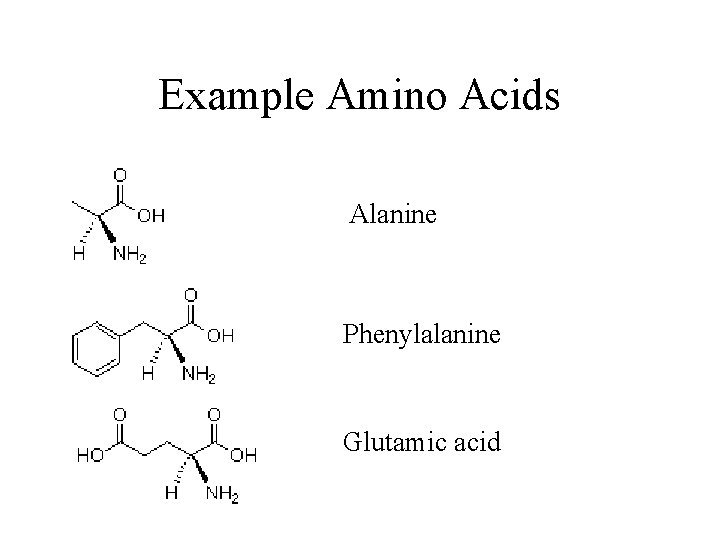
Example Amino Acids Alanine Phenylalanine Glutamic acid
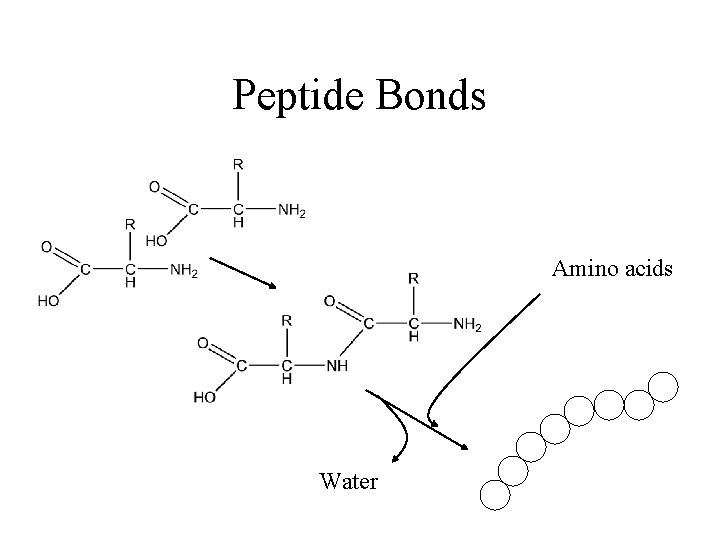
Peptide Bonds Amino acids Water
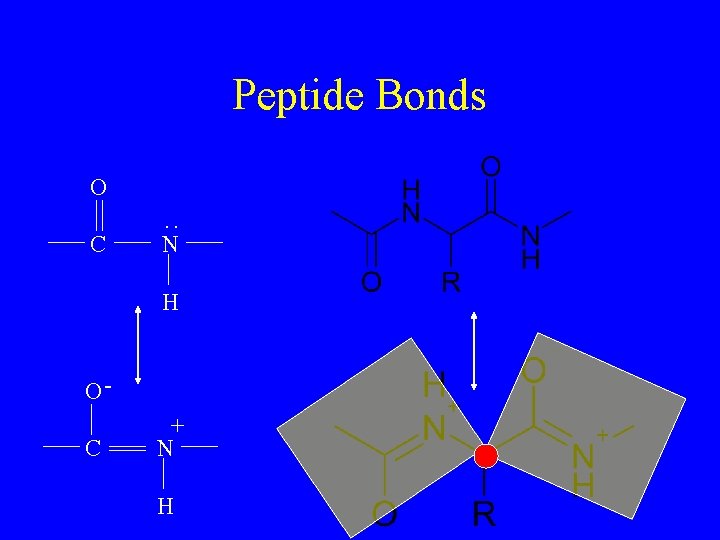
Peptide Bonds : O C N H O+ C N H

Disulfide Bonds • Two cysteine molecules under oxidizing conditions • Intermolecular or intramolecular crosslink
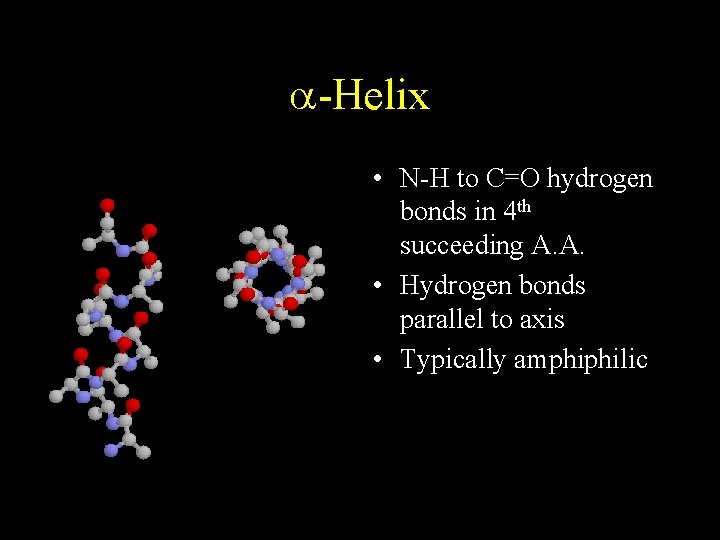
a-Helix • N-H to C=O hydrogen bonds in 4 th succeeding A. A. • Hydrogen bonds parallel to axis • Typically amphiphilic
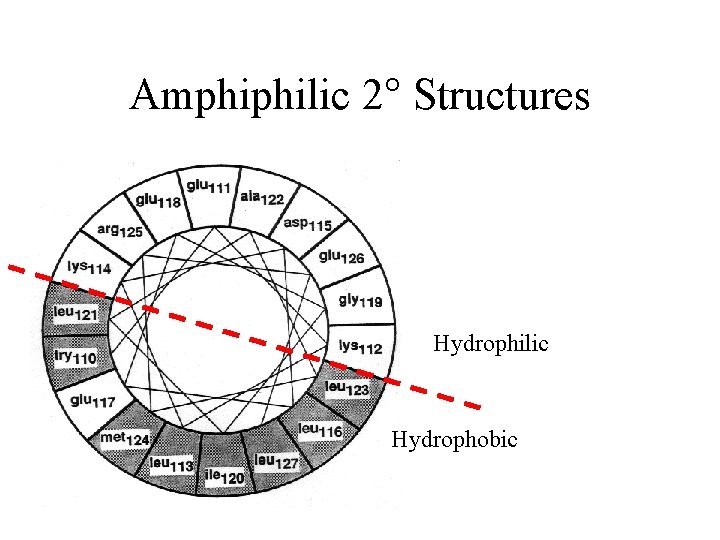
Amphiphilic 2° Structures Hydrophilic Hydrophobic

b-Sheet • C=O and N-H perpendicular to chain form inter-segment H-bonds • Parallel or antiparallel • b-strands typically 5 -15 A. A. • More stable than a-helix b-sheet
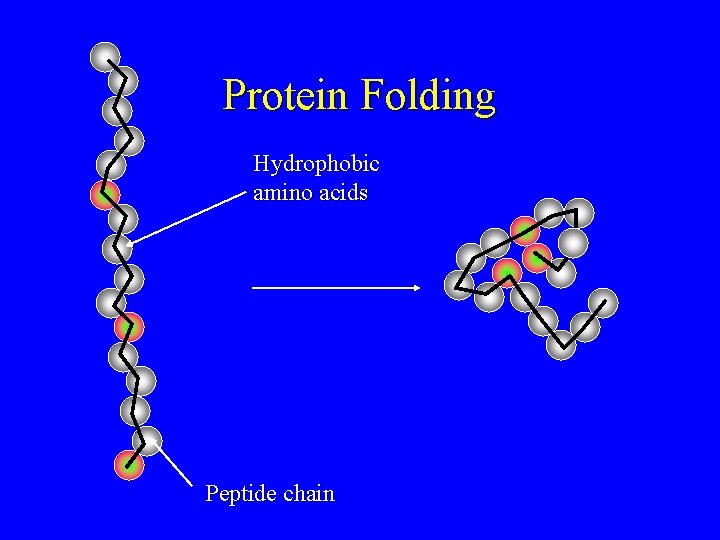
Protein Folding Hydrophobic amino acids Peptide chain

Tertiary Structure
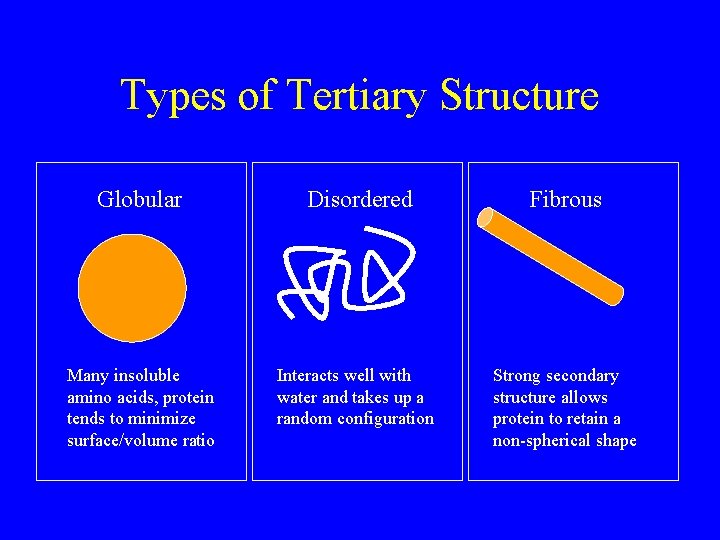
Types of Tertiary Structure Globular Disordered Fibrous Many insoluble amino acids, protein tends to minimize surface/volume ratio Interacts well with water and takes up a random configuration Strong secondary structure allows protein to retain a non-spherical shape
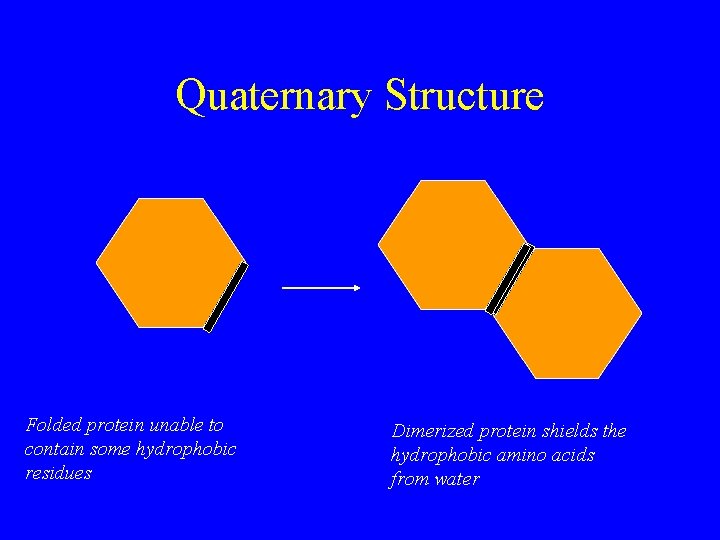
Quaternary Structure Folded protein unable to contain some hydrophobic residues Dimerized protein shields the hydrophobic amino acids from water
- Slides: 16