Protein Separation BME 273 Cathy Castellon Advisor Dr

Protein Separation BME 273 Cathy Castellon Advisor: Dr. Haselton Graduate Advisor: Greg Stone 9/16/2020
Overview of Proteins 1 z Master molecules of living things z Composition z Development (central dogma)
Master Molecules of Living Things 1 y. Enzymes y. Structural proteins y. Contractile proteins y. Membrane proteins y. Transport proteins y. Storage proteins y. Protective proteins y. Hormones
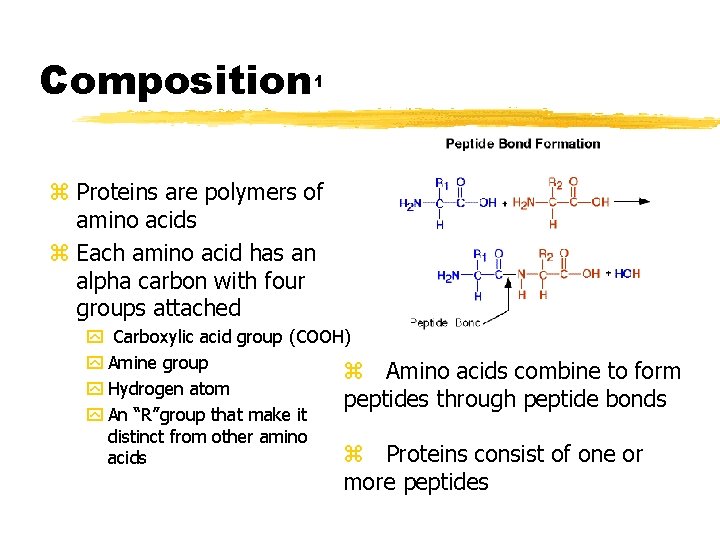
Composition 1 z Proteins are polymers of amino acids z Each amino acid has an alpha carbon with four groups attached y Carboxylic acid group (COOH) y Amine group z Amino acids combine to form y Hydrogen atom peptides through peptide bonds y An “R”group that make it distinct from other amino z Proteins consist of one or acids more peptides
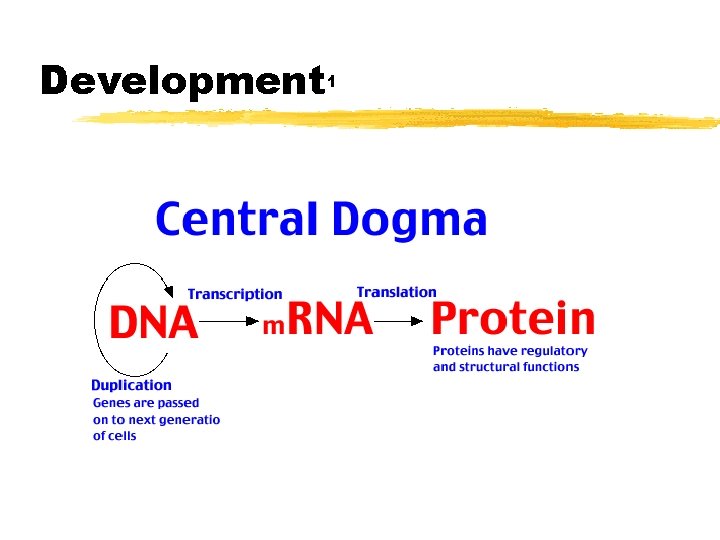
Development 1
Improving Protein Separation z. Current Technology y 2 D protein electrophoresis(SDS-PAGE) z. The Need y To compare the expression of protein profiles from an arbitrary reference state of a cell, tissue, or organism, to the profile of an non-standard condition z. Economics y. Faster more efficient technology

How does 2 DE work? z Separates proteins by isoelectric point (p. I) z And second by size (molecular weight) using sodium didecyl sulfate polyacrylamide gel electorphoresis (SDSPAGE) 1

SDS-PAGE

Why a New Design? 1 z Time consuming y Running the two gels can take up to 10 -15 hrs z Labor intensive y Gels are often hand cast y Samples must be loaded by hand y Tube gel must be properly combined with the slab gel z Resolution problems y Usually Less then 1000 proteins can be resolved

Our Thoughts z. Design Microfluid Technique y. Separation based on hydrophobicity y. Create flow channel (lithography) y. Create inlet and outlet points y. Load fluorescently labeled protein solution into one end y. Pump buffer solution through the channel y. Fluoremeter will detect separation
Game Plan ü Accomplished ü Slides ü gradient ü contact angle measurements ü Simple Channel Design ü Lithograph technique ü PDMS ü Detailed Channel Design ü Mix protein ü Protein Labeling ü In the Works ü Flurometer Setup ü Prototype

Slides z Gradient yhydro-phobic/phyllic z Contact Angle Measurements z PDMS Adherence

Simple Channel Design z Lithography Technique z Single 2 X 2 cm Lane z PDMS z Works with clean slide z Doesn’t work (yet) with hydrophobic slide z Claming the prototype

Detailed Channel Design z 2 X 2 cm lanes z. Hydro-phobic/phyllic on same slide (R, L) z. Posts used to aid mixing and accentuate the separation
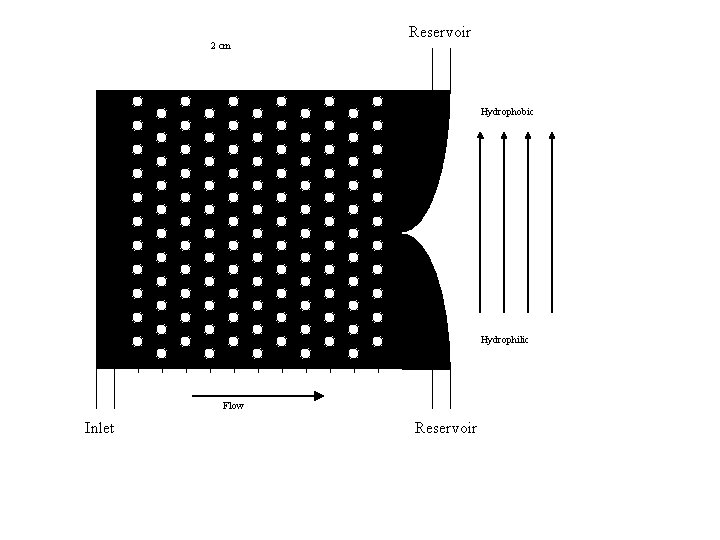
2 cm Reservoir Hydrophobic Hydrophilic Flow Inlet Reservoir

Protein Labeling z 2 different proteins y. Cytocrsome C and Lysosyme z 2 different labels y. Alexa. Fluro 430 (540 nm) and 350 (442 nm)

Furture Work: Setup Flow Chamber/ Fluorescence Spectrophotometer z. Flow Chamber-basic idea ymonitor pressure of flow ymonitor flow rate z. Spectrophotometer ytest each reservoir and measure labeled protein signal

References z. Protein Notes- Greg Stone (1) z. Lithograph Technique- Dave Schaffer z. Mass Spectrometry (SDS-PAGE)- Dr. David Hachey
- Slides: 18