Protein Purification BL 4010 10 05 The basic

Protein Purification BL 4010 10. 05

The basic techniques • Concentration (size) – – precipitation ultrafiltration dialysis centrifugation • Chromatography (size/charge/chemistry) – – ion exchange size exclusion affinity hydrophobic interaction • Electrophoresis (size/charge) – – "native" denaturing isoelectric focusing 2 -dimensional • Immunological – chromatography – in situ imaging – immunoblotting
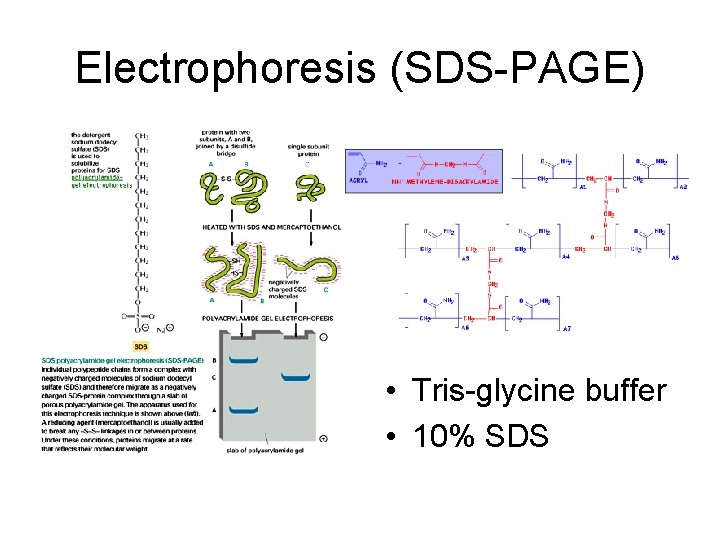
Electrophoresis (SDS-PAGE) • Tris-glycine buffer • 10% SDS
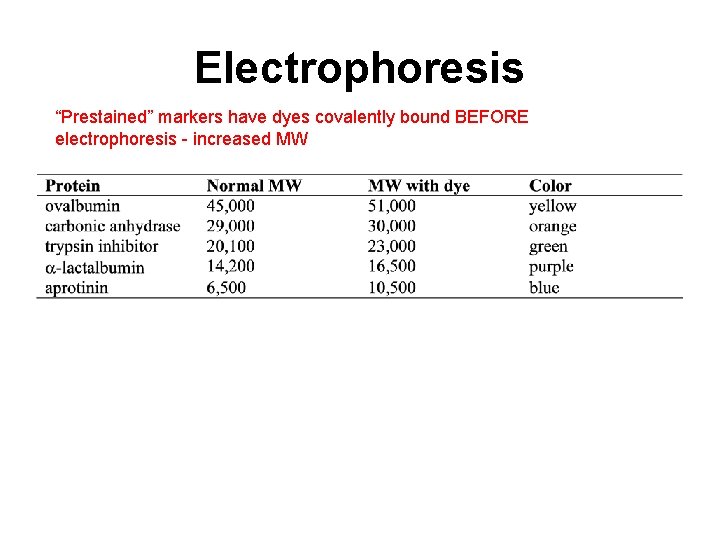
Electrophoresis “Prestained” markers have dyes covalently bound BEFORE electrophoresis - increased MW
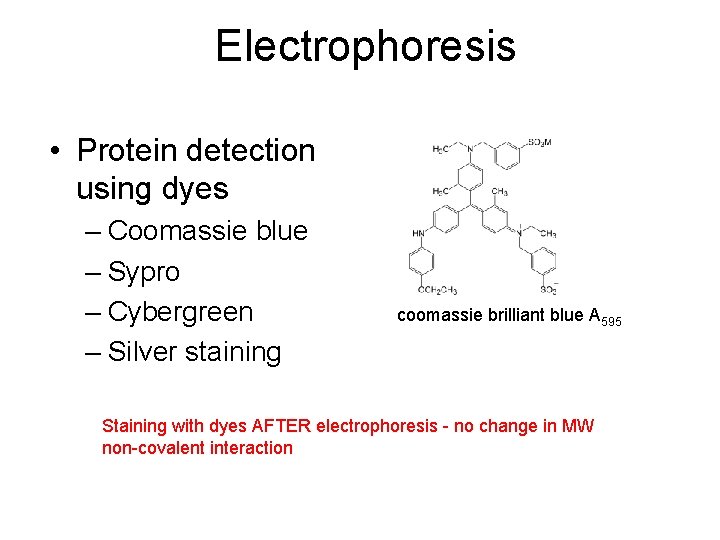
Electrophoresis • Protein detection using dyes – Coomassie blue – Sypro – Cybergreen – Silver staining coomassie brilliant blue A 595 Staining with dyes AFTER electrophoresis - no change in MW non-covalent interaction
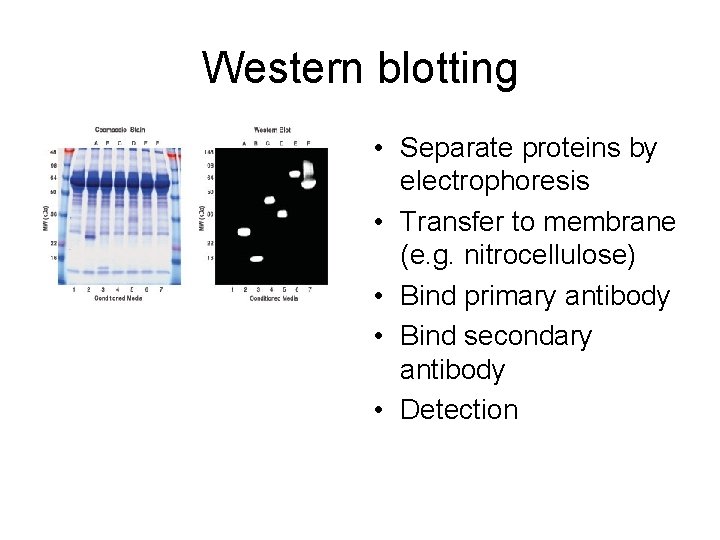
Western blotting • Separate proteins by electrophoresis • Transfer to membrane (e. g. nitrocellulose) • Bind primary antibody • Bind secondary antibody • Detection
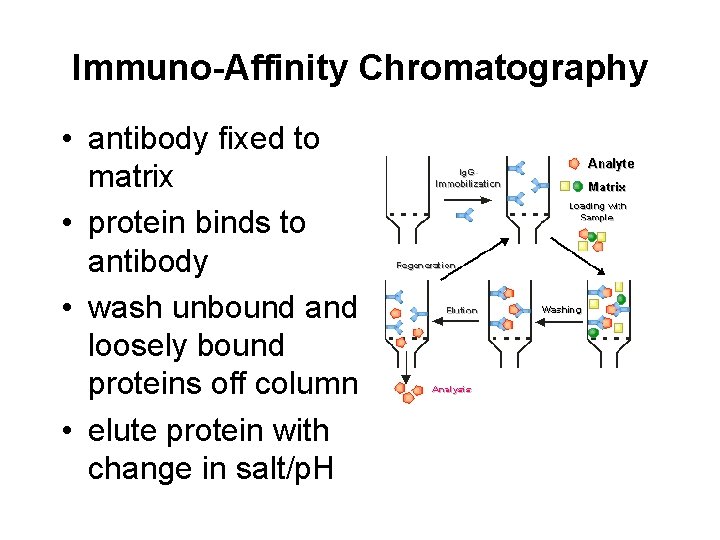
Immuno-Affinity Chromatography • antibody fixed to matrix • protein binds to antibody • wash unbound and loosely bound proteins off column • elute protein with change in salt/p. H

Hydrophobic interaction chromatography • Hydrophobic group bound to solid phase • Binding – high salt (increases water surface tension, decreases available water molecules, increases hydrophobic interactions) • Elution – decrease salt – add detergent – decrease polarity of mobile phase
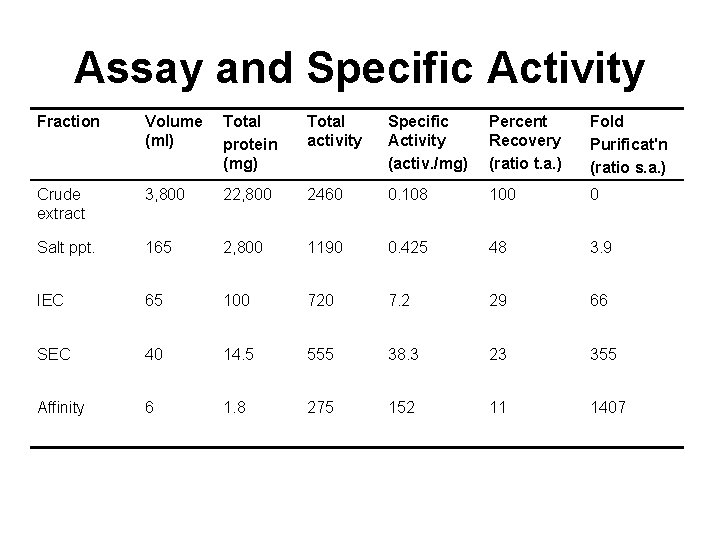
Assay and Specific Activity Fraction Volume (ml) Total protein (mg) Total activity Specific Activity (activ. /mg) Percent Recovery (ratio t. a. ) Fold Purificat'n (ratio s. a. ) Crude extract 3, 800 22, 800 2460 0. 108 100 0 Salt ppt. 165 2, 800 1190 0. 425 48 3. 9 IEC 65 100 720 7. 2 29 66 SEC 40 14. 5 555 38. 3 23 355 Affinity 6 1. 8 275 152 11 1407

Criteria for purity When is protein pure or pure enough? • homogeneity – protein complexes? • constant specific activity • Practical: further attempts at purification are futile since the only material left in the fraction is the material that actually is responsible for the activity being assayed.

Protein purification simuation • http: //www. tlsu. leeds. ac. uk/courses/bioc 20 60/proteinlab 102/proteinlab. html

Enzymes BL 4010 10. 12. 05

Objectives • What is an enzyme? • How do enzymes work? – energetics – underlying general mechanism – components (prosthetic groups, coenzymes) – specific mechanisms Ch. 13. 1, 13. 2, 14. 1, 14. 2, 14. 3, 14. 4, 14. 5

What is an enzyme? Macromolecular biological catalyst Can be protein or RNA

What is an enzyme? Macromolecular biological catalyst What is a catalyst? – is not altered by reaction • participates but emerges unchanged – increases the rate at which substrates and products reach equilibrium – does not alter equilibrium

Why enzymes? • Why invest energy and resources into creating a large catalyst? – Enzymes endow cells with the remarkable capacity to exert kinetic control over thermodynamic potentiality • Fine tune selectivity (substrate binding specificity) • Fine tune catalytic rate • Additional regulatory control (e. g. allostery, signalling networks)

Enzymes are good catalysts • Enzymes can accelerate reactions as much as 1016 over uncatalyzed rates! • Urease is a good example: – Catalyzed rate: 3 x 104/sec – Uncatalyzed rate: 3 x 10 -10/sec – Ratio is 1 x 1014 !

Enzymes are selective catalysts • Enzymes selectively recognize proper substrates over other molecules • Enzymes produce products in very high yields - often much greater than 95% • Specificity is controlled by structure - the unique fit of substrate with enzyme controls the selectivity for substrate and the product yield
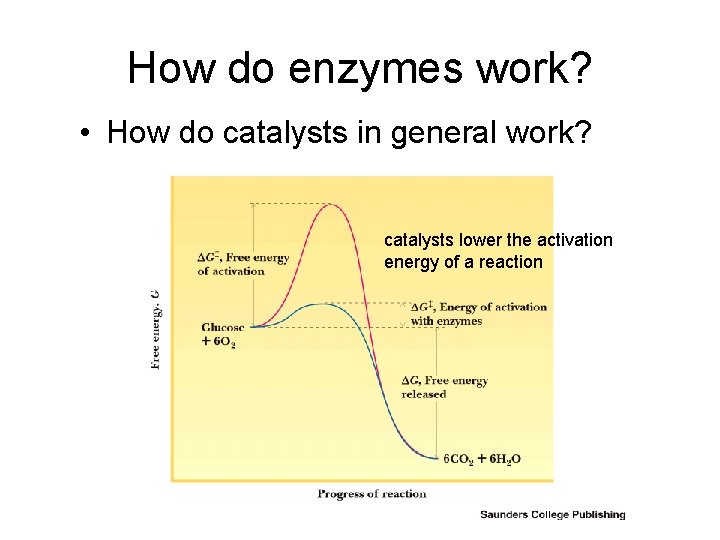
How do enzymes work? • How do catalysts in general work? catalysts lower the activation energy of a reaction

The transition state Understand the difference between G and G‡ • The overall free energy change for a reaction is related to the equilibrium constant • The free energy of activation for a reaction is related to the rate constant • It is extremely important to appreciate this distinction!
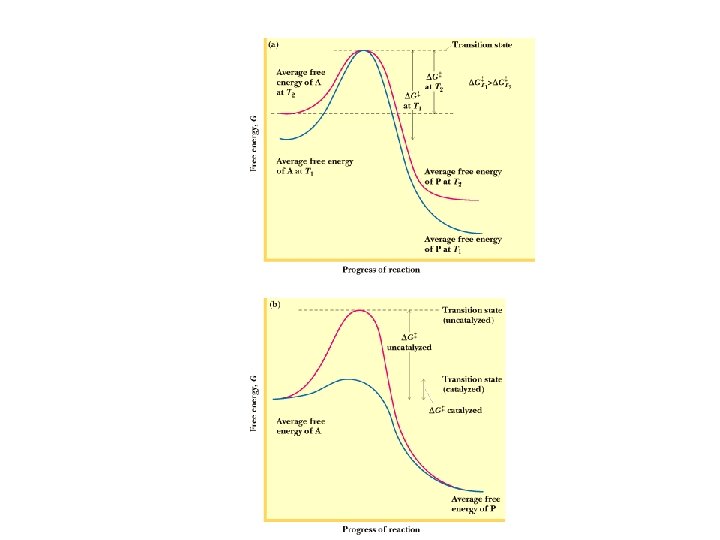

How do enzymes work? • Enzymes accelerate reactions by lowering the free energy of activation HOW?

Four contributing factors to enzyme catalysis NO ONE MECHANISM ACCOUNTS FOR CATALYSIS ALONE! • Specific substrate binding – local concentration of reactants – productive orientation of reactants – binding energy used to offset loss of entropy • Control over solvent interactions – desolvation (binding energy offsets) – ordered solvent in binding pocket • Induction of strain on reactants • Alternate reactive pathway – transient involvement of enzyme functional groups

How do enzymes work? • Enzymes accelerate reactions by lowering the free energy of activation • Enzymes do this by binding the transition state of the reaction better than the substrate
- Slides: 24