Protein Production Jackpot GFP column chromatography Post p










- Slides: 16

Protein Production Jackpot! GFP column chromatography

Post p. GLO: Now you have a protein but…. How do you use it? How would you be able to test just the GFP protein? How do you verify your results?

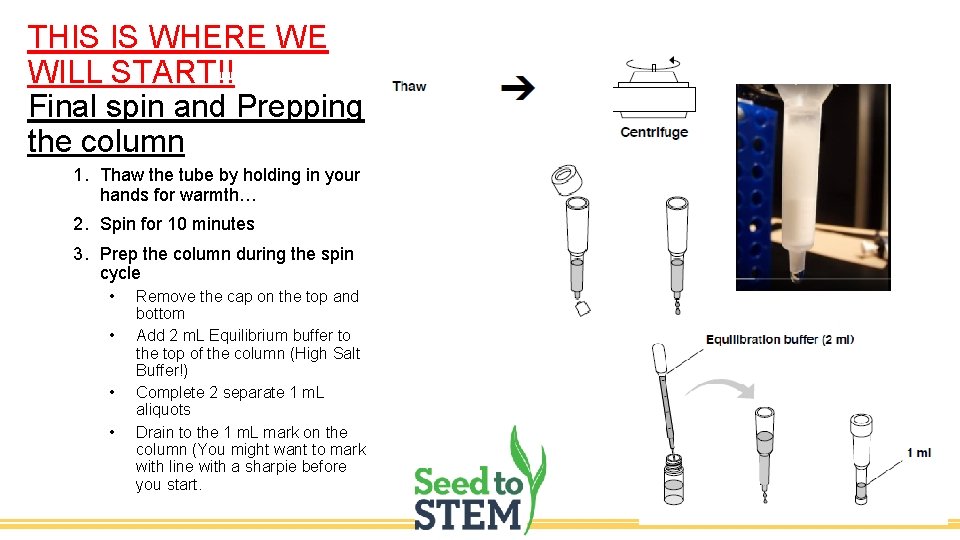
THIS IS WHERE WE WILL START!! Final spin and Prepping the column 1. Thaw the tube by holding in your hands for warmth… 2. Spin for 10 minutes 3. Prep the column during the spin cycle • • Remove the cap on the top and bottom Add 2 m. L Equilibrium buffer to the top of the column (High Salt Buffer!) Complete 2 separate 1 m. L aliquots Drain to the 1 m. L mark on the column (You might want to mark with line with a sharpie before you start.

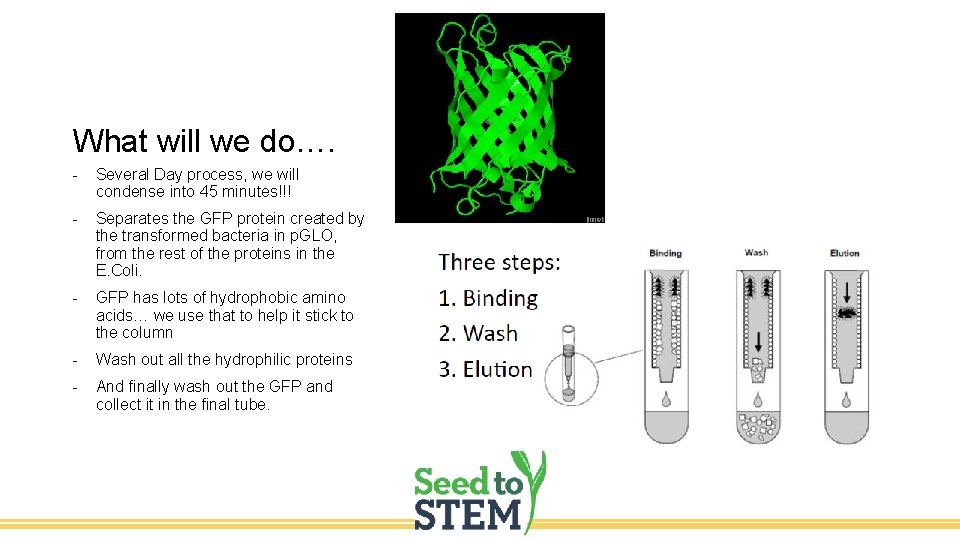
What will we do…. - Several Day process, we will condense into 45 minutes!!! - Separates the GFP protein created by the transformed bacteria in p. GLO, from the rest of the proteins in the E. Coli. - GFP has lots of hydrophobic amino acids… we use that to help it stick to the column - Wash out all the hydrophilic proteins - And finally wash out the GFP and collect it in the final tube.

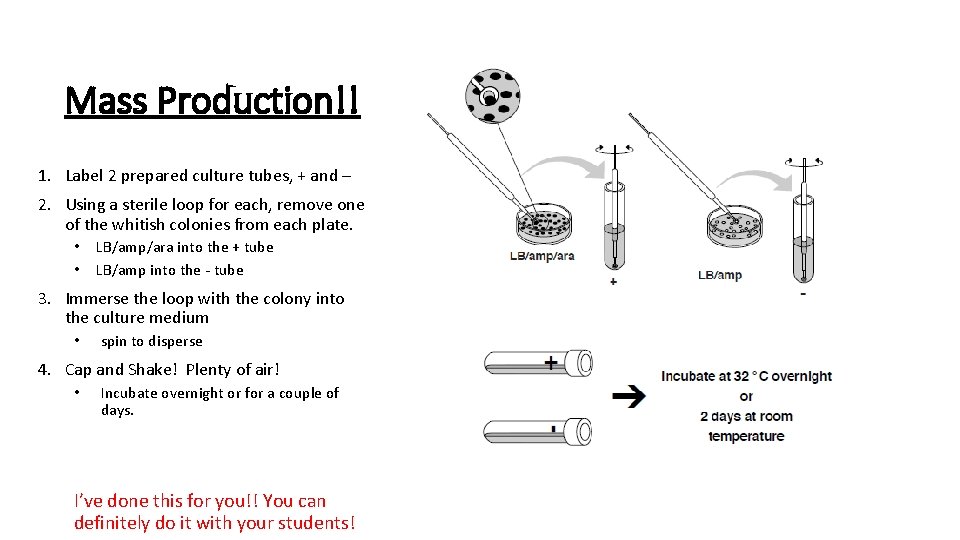
Mass Production!! 1. Label 2 prepared culture tubes, + and – 2. Using a sterile loop for each, remove one of the whitish colonies from each plate. • • LB/amp/ara into the + tube LB/amp into the - tube 3. Immerse the loop with the colony into the culture medium • spin to disperse 4. Cap and Shake! Plenty of air! • Incubate overnight or for a couple of days. I’ve done this for you!! You can definitely do it with your students!

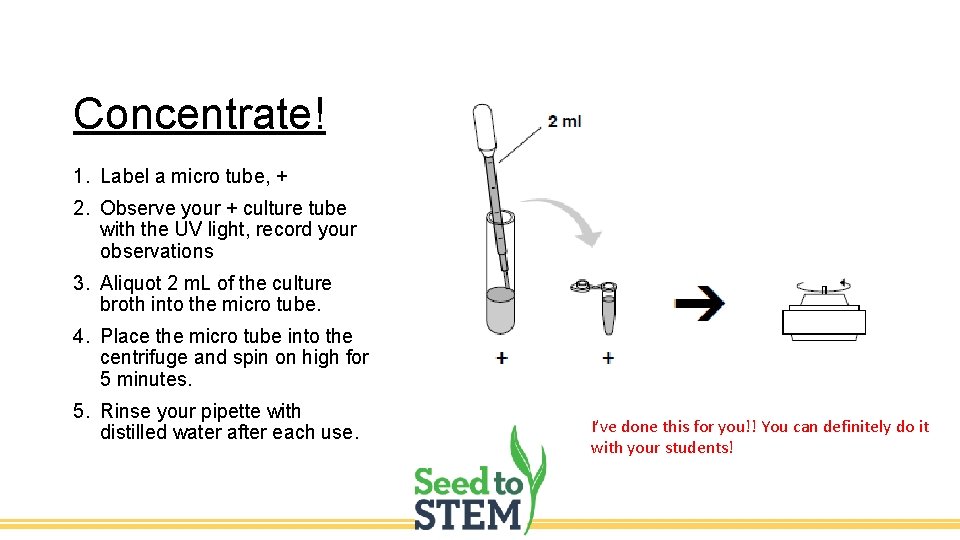
Concentrate! 1. Label a micro tube, + 2. Observe your + culture tube with the UV light, record your observations 3. Aliquot 2 m. L of the culture broth into the micro tube. 4. Place the micro tube into the centrifuge and spin on high for 5 minutes. 5. Rinse your pipette with distilled water after each use. I’ve done this for you!! You can definitely do it with your students!

Draining… 1. Pour all the liquid out of the top of the centrifuge tube. 2. Observe the pellet at the bottom of the tube with the UV light. 3. Using the rinsed pipette, add 250 ul of TE buffer. 4. Move the liquid up and down in the tube using the pipette to resuspend the bacteria. I’ve done this for you!! You can definitely do it with your students!

Digestion 1. Add 1 Drop of Lysozyme to your tube. 2. Cap and gently mix by flicking a few times. 3. Place your sample in the freezer overnight. I’ve done this for you!! You can definitely do it with your students!

Recap! Let the separation Begin! What How Why Status 1. Mass produced bacteria that create GFP protein Grew transformed bacteria in culture tubes Increase amount of the desired protein DONE 2. Broken Down the cells walls Enzymatically- Lysozyme Physically- Freezing overnight Releases all the proteins from inside the bacterial cell walls. DONE 3. Collect the protein Column chromatography, Trap the GFP in the column, wash out everything else, then wash out the GFP Separates GFP (the desired protein) from all the other proteins created by the cells. NOT YET!

Binding the Protein 1. Using a NEW pipette add 250 ul of the supernatant (Liquid) into a NEW micro tube. 2. Add 250 ul of Binding Buffer (BB) to the micro tube with the sample. 3. Cap and gently flick with your finger to mix.

Let the Drain begin. 1. Label 3 collection tubes 1, 2, 3 2. Place in the foam rack 3. Remove the cap from the column and place into #1 tube 4. Let the buffer drain to the top of the matrix. 5. Add a little pressure to speed up this process: • • • Draw up the plunger on the syringe Place the stopper in the top of the column Gently push some air through the syringe until you reach right up the matrix.

Loading the sample 1. Using a NEW pipette aliquot 250 u. L sample from the micro tube into the column 2. SLOWLY add by touching the tip to the side of the column and dripping it down into the column 3. Let it continue to drain into tube 1 until it stops dripping. • Add pressure to speed up the process. Stop when the liquid reaches the top of the matrix.

Sorting the trash… 1. Switch to tube #2. 2. Using a new pipette, Add 250 u. L of Wash Buffer to the column 3. Let the entire amount flow through the column • Add pressure as needed. 4. Wait for it to stop dripping!

The Final Countdown 1. Switch to tube #3 2. Using a new Pipette, Add 750 u. L of TE buffer 3. Let the entire volume flow through the column 4. Observe using UV light, watch closely as the GFP moves down the column. 5. Cap all your collection tubes

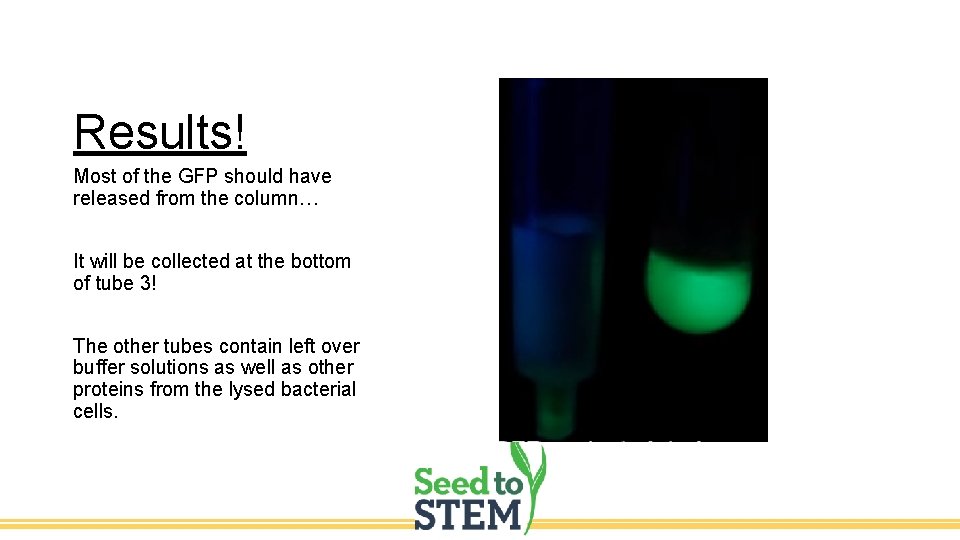
Results! Most of the GFP should have released from the column… It will be collected at the bottom of tube 3! The other tubes contain left over buffer solutions as well as other proteins from the lysed bacterial cells.

Brought to you by: Connect with us: #kansascorn. STEM kscorn. com