PROTEIN METABOLISM PROTEINS Function of Proteins v Catalytic

PROTEIN METABOLISM

PROTEINS Function of Proteins v. Catalytic functions v. Protection v. Transport v. Structure v. Regulation v. Nerve İmpulse transmission v. Movement v. Receptor v. Storage http

• Humans require 20 aminoacids - 9 are essential - 11 are not essential • Cellular proteins are synthesized by ribosomes using the information encoded in m. RNA • Proteins are composed of aminoacids that are linked together by peptide bonds
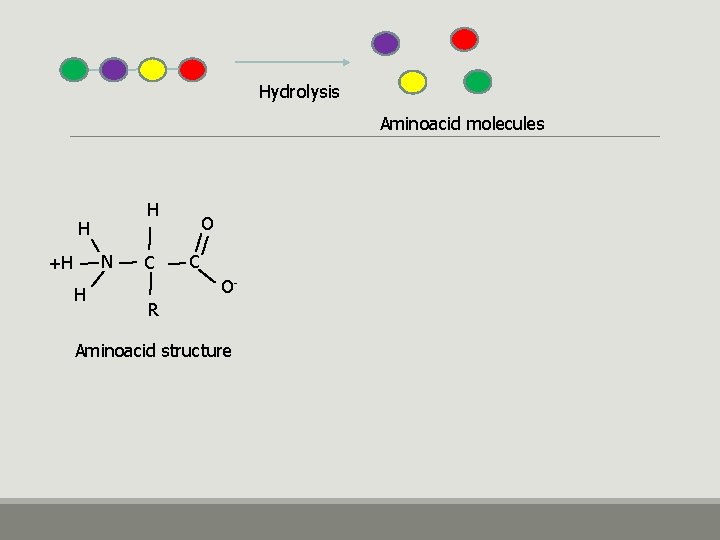
Hydrolysis Aminoacid molecules H H N +H H C O- R Aminoacid structure

PROTEIN METABOLISM v Protein metabolism is a chemical procedure v Break down dietary or endogenous proteins into amino acids v Ingested dietary proteins are metabolised to amino acids via peptide bond cleaving enzymes (proteinases) v Absorbed into the bloodstream

v No aminoacid stroge in the body v Aminoacids can be provided by diet, de novo synthesis of aminoacids and protein degredation

Aminoacid Pool Endogenous body proteins Exogenous diyet proteins Non essential aminoacid synthesis Aminoacid Pool Protein Synthesis of molecules containing nitrogen Conversion of amino acids to glucose, glycogen, fatty acids Aminoacid oxidation

Negative Nitrogen Balance v. Nitrogen excretion > nitrogen content of the diet v. Tissue destruction. Positive Nitrogen Balance v. Nitrogen excretion < nitrogen content of the diet v Protein formaiton

DIGESTION OF DIETARY PROTEINS v. A large amount of dietary nitrogen is originated from protein v. Proteins are hydrolyzed……Di, tripeptides, aminoacids are produced………. and they can be absorbed.

Proteolytic enzymes §Responsible for degrading proteins §They are produced by stomach, pancreas, small intestine

Digestion of Proteins (Stomach) v Pepsin is the digestive enzyme in the stomach v Pepsin, p. H 2 -3 active, p. H> 5 inactive v HCl; killing bacteria, protein denaturation, partial activation of pepsinogen v Pepsinojen, is the inactive form of pepsin, activated by HCl and a partially active pepsin enzyme.

DIGESTION BY PANCREATIC ENZYMES v The pancreas secretes pancreatic digestive enzymes. Trypsin, chymotrypsin, carboxypeptidase, elastase. v These proteolytic enzymes are responsible for breaking down polypeptides into smaller peptides. v Secretin and cholecystokinin are hormones secreted by the small intestine. v The secretin stimulates the release of bicarbonate by the pancreas v Cholecystokinin causes digestive enzymes to reach the small intestine by the pancreas. v It contains pancreatic fluid, water, bicarbonate and digestive enzymes.

Activation of Zymogens; v. Include extra amino acids v. Catalytically inactive form of an anzyme v. Removal of extra amino acid sequence → appropriate folding → catalytically active enzyme

PANCREATITIS Normally Pancreatic Zymogens Reach to the small intestine Then they are normally activated Pancreatitis Inappropriate activation of trypsin Dangeorus sitution occurs

Enterokinase Trypsinogen Trypsin Chymotrypsinogen Proelastase Procarboxipeptidase A Procarboxpeptidase B Chymotrypsin Elastase Carboxipeptidase A Carboxipeptidase B

Tripsin Chymotrypsin Elastase Carboxypeptidase A Carboxypeptidase B Arg, lys Trp, Tyr, Phe, Met, Leu Ala, Gly, Ser A. . Ala, Ile, Leu, Val B. . Arg, Lys Lippincott’s Illustrated Reviews Biochemistry Denise R. Ferrier Series Editor: Richard A. Harvey, 6 th Edition

Absorption of Aminoacids In enterocytes; v. Amino acids, di and tripeptides are absorbed. v. Absorption mechanisms of di, tripeptides and aminoacids are different
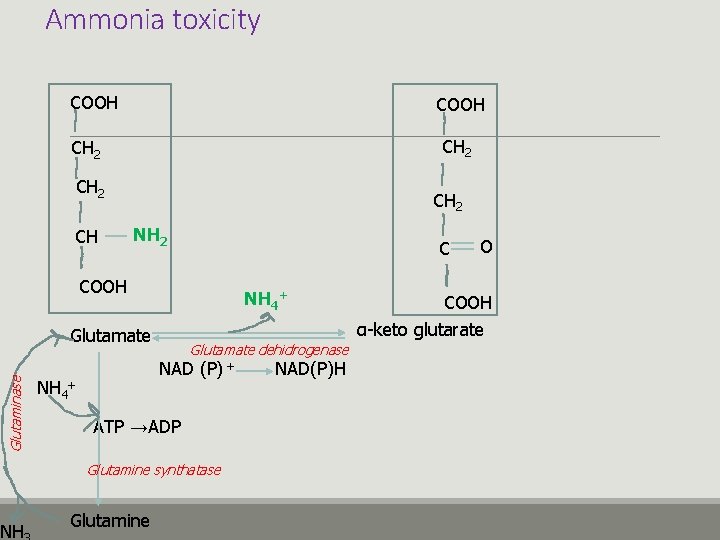
Ammonia toxicity COOH CH 2 CH CH 2 NH 2 C COOH NH 4+ Glutaminase Glutamate NH Glutamate dehidrogenase NAD (P) + NH 4+ ATP →ADP Glutamine synthatase Glutamine NAD(P)H O COOH α-keto glutarate

Ammonia toxicity v Glutamate is formed as a result of ammonia and alpha keto glutarate reaction v Glutamate is essential for glutamine production v Ammonia concentration↑ Glutamine ↑ v Alpha ketoglutarate↓ v Glutamate ↓ v The functions of the TCA cycle are disrupted in neurons. v Glutamate is necessary for GABA synthesis v Glutamine is osmotically active in brain astrocytes v Energy production↓ v GABA ↓ v Brain Edema

Ammonia Sources: 1. 2. 3. 4. 5. Aminoacids Glutamine Bacterial action (Intestine) Amines Pürins and pirimidins

Ammonia Transport v. Blood ammonia is quickly cleared by the liver v. The release of ammonia from the muscles to the bloodstream occurs in the form of glutamine or alanine. v. The main role of the glucose-alanine cycle; is to allow the skeletal muscle to eliminate nitrogen. v. The amino acid of alanine plays a role in the non-toxic transport of ammonia to the liver.
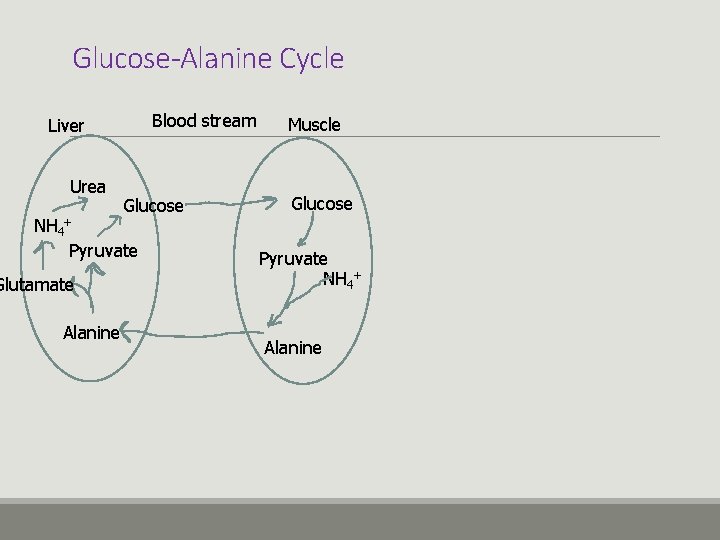
Glucose-Alanine Cycle Blood stream Liver Urea Glucose NH 4+ Pyruvate Glutamate Alanine Muscle Glucose Pyruvate NH 4+ Alanine

REFERENCES Lippincott’s Illustrated Reviews Biochemistry Denise R. Ferrier Series Editor: Richard A. Harvey, 6 th Edition https: //www. ncbi. nlm. nih. gov/pubmed/22139560
- Slides: 23