Protein metabolism Protein structure All amino acid contain
Protein metabolism
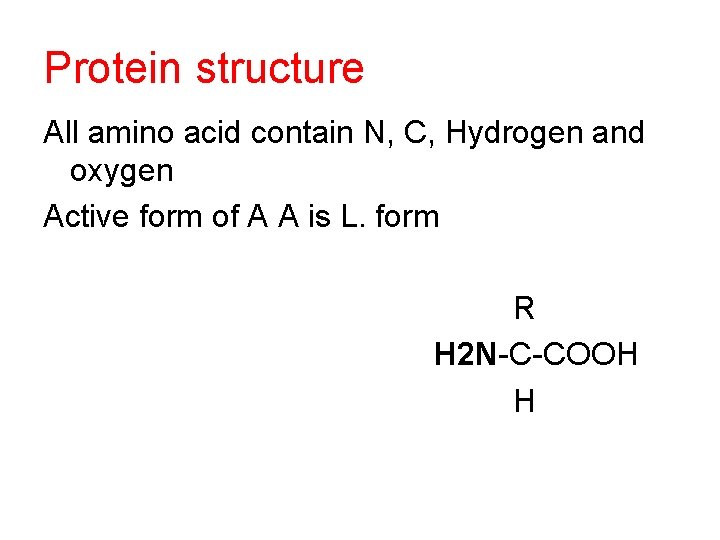
Protein structure All amino acid contain N, C, Hydrogen and oxygen Active form of A A is L. form R H 2 N-C-COOH H

Protein digestion In stomach • Pepsin Its endopeptidase act on AA on middle of polypeptide chain Digestion by pepsin proteosis, peptone, polypeptide • Renin Act on casin which is a milk protein Casin + rennin paracasin + Ca ( insoluble Ca paracasinate (milk clot )

In the intestine • By pancreatic enzyme • Trypsin -Its endopeptidase hydrolyse the peptide bond of basic AA -Release small peptide and free AA - Chemotypsin -Its endopeptidase hydrolyse aromatic AA • Carboxypeptidase - Its exodopeptidase hydrolyse peptide at periphery •

• By intestinal enzyme • Aminopeptidase - Hydrolyse peptide bond at amino end. • Dipeptidase : - Hydrolyse dipeptide give 2 AA

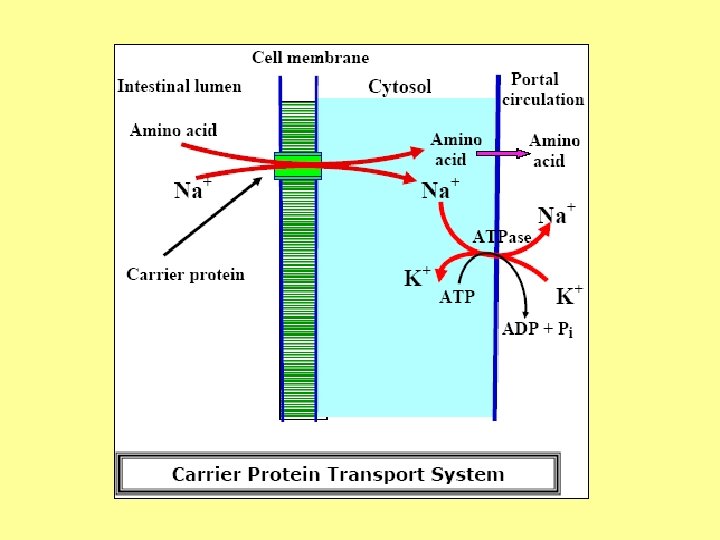
Absorption • AA absorbed by small intestine • Mechanism of absorption 1 - Active transport • Carrier protein transport • Glutathione transport system

- Deamination • It is the removal of amino group from amino acids in the form of ammonia (NH 3). • Types of deamination 1 - Oxidative deamination 2 - Non-oxidative deamination

Oxidative deamination 1 - L-glutamate dehydrogenase 2 - L-amino acid oxidase. 3 - D-amino acid oxidase.
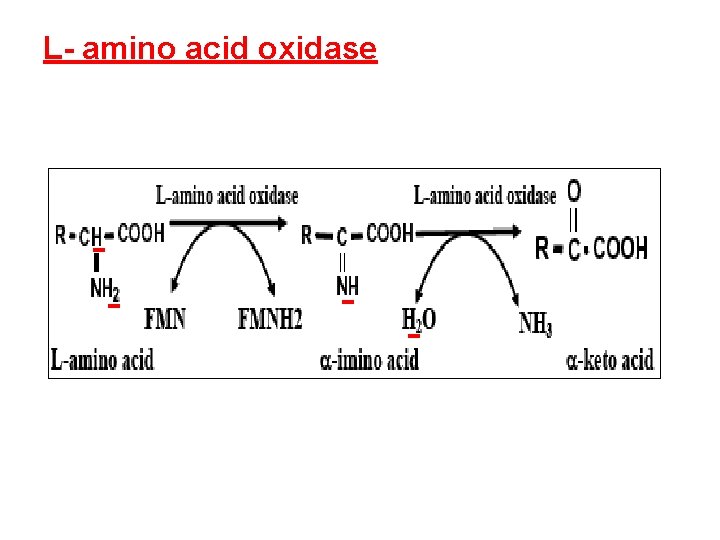
L- amino acid oxidase
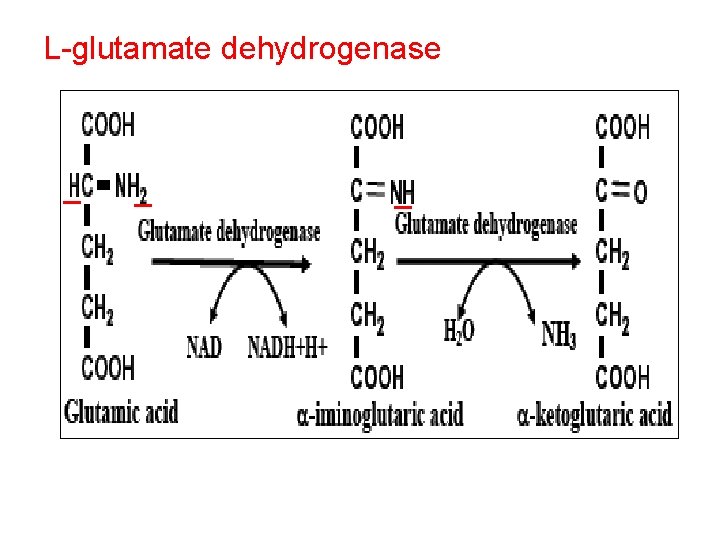
L-glutamate dehydrogenase

• Function of L. Glutamate dehydrogenase 1 - Removal of NH 2 as NH 3 2 - Formation of non essential AA

D- amino acid oxidase • Not present in human
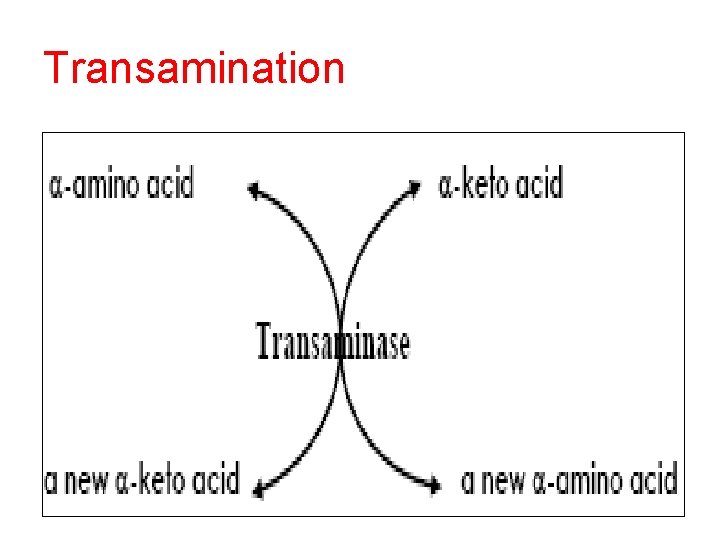
Transamination
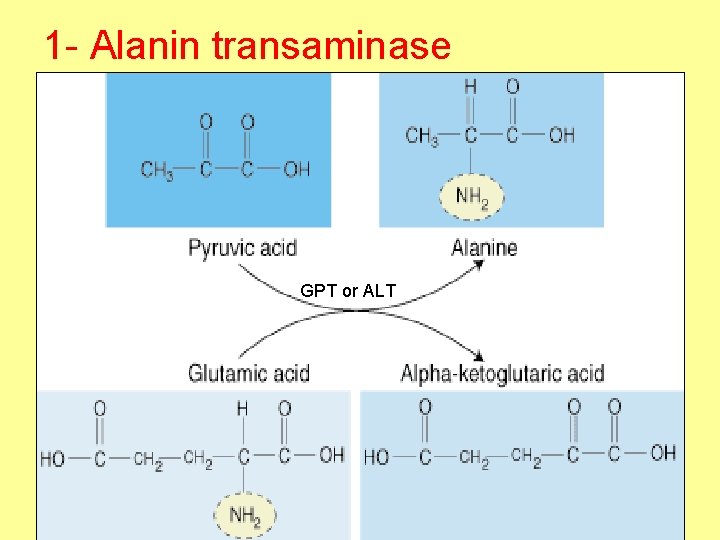
1 - Alanin transaminase GPT or ALT
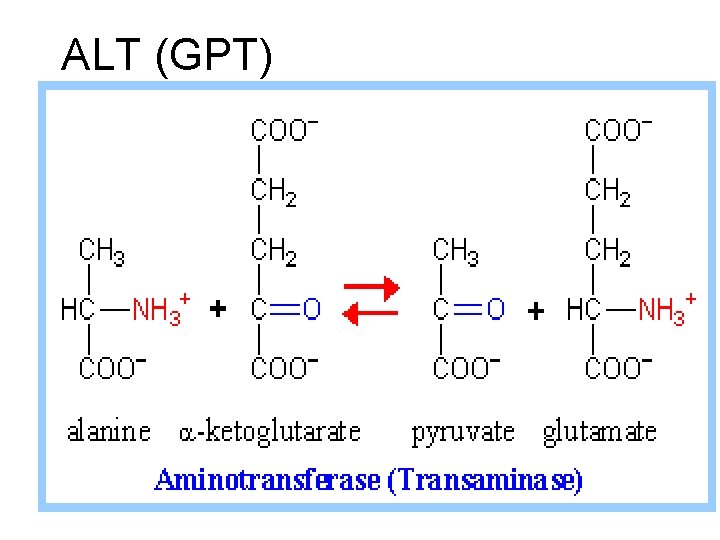
ALT (GPT)

• Clinical importance : GPT (ALT) increase in liver disease as hepatitis (damage of liver cells).
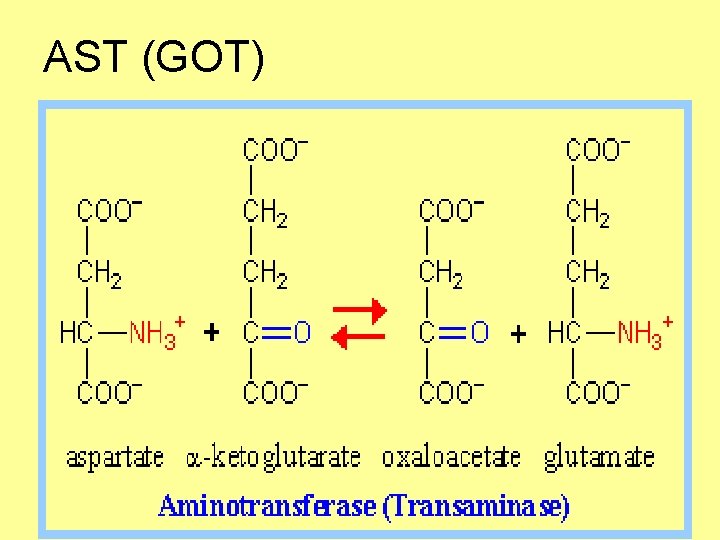
AST (GOT)

• Elevated level of AST suggests damage to heart muscle (myocardial infarction), skeletal muscle or kidney
Ammonia

Fate of ammonia (normal level of NH 3 10 – 110 μg/dl) 1 - Glutamine synthesis • Is the major mechanism for removal of from NH 3 by brain 2 - Urea synthesis • Is the major mechanism for removal of from NH 3 by liver 3 - Direct excretion in urine 4 - Synthesis of new AA • NH 3 + α ketoglutaric glutamic 5 - Synthesis of purine and pyrimidine
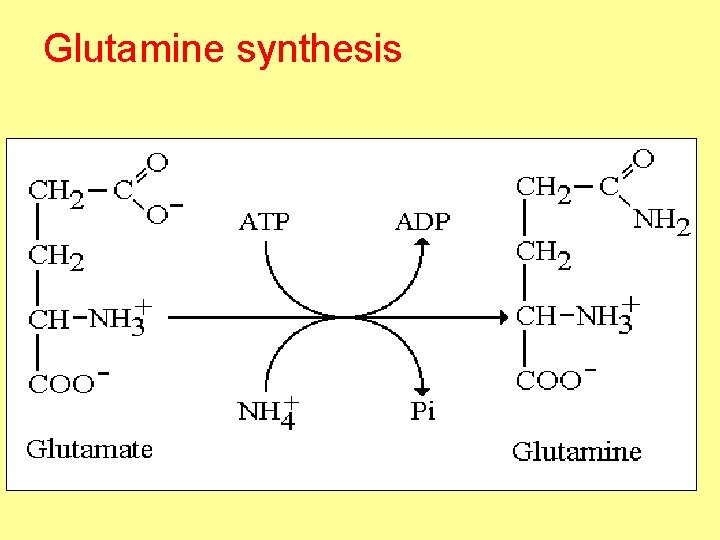
Glutamine synthesis

• Function of glutamine 1. Regulation of acid base balance 2. Remove toxic effect of NH 3 in brain 3. Glutamic is source of N 3, N 9 of purine

Urea cycle Atom of urea derived from N 1 derived from NH 3 N 2 derived from Aspartic C=O derived from CO 2 First 2 reaction occur in mitochondria , the remaining occur in cytoplasm 3 ATP required in cycle Urea synthesized in liver

Urea cycle
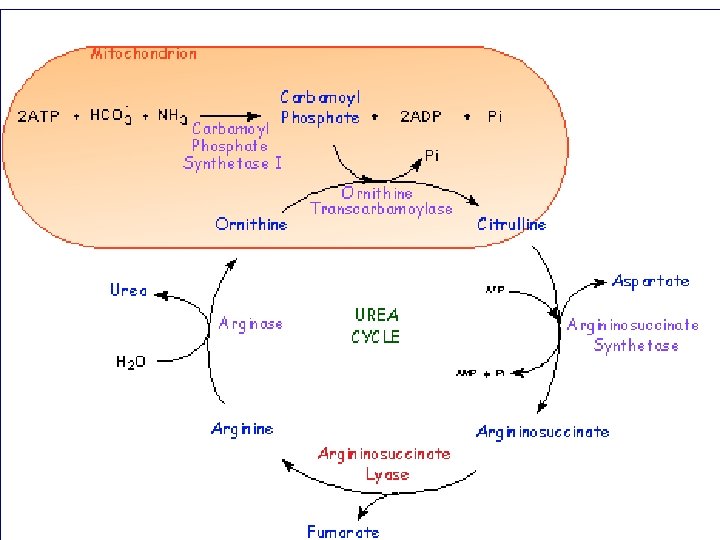
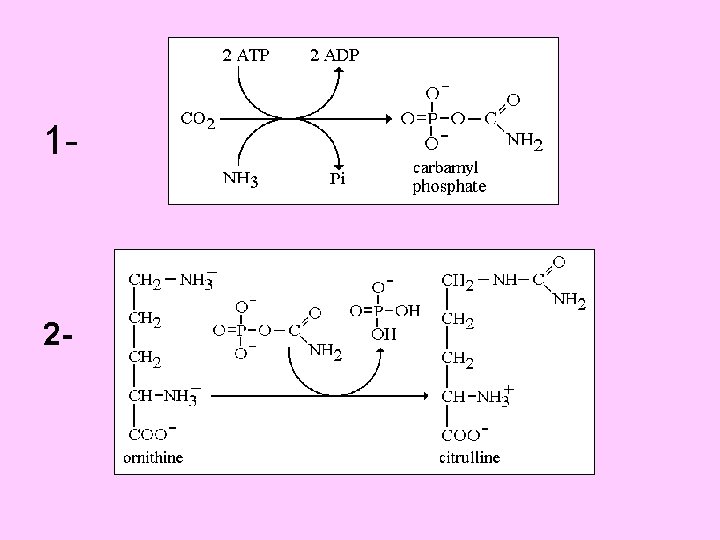
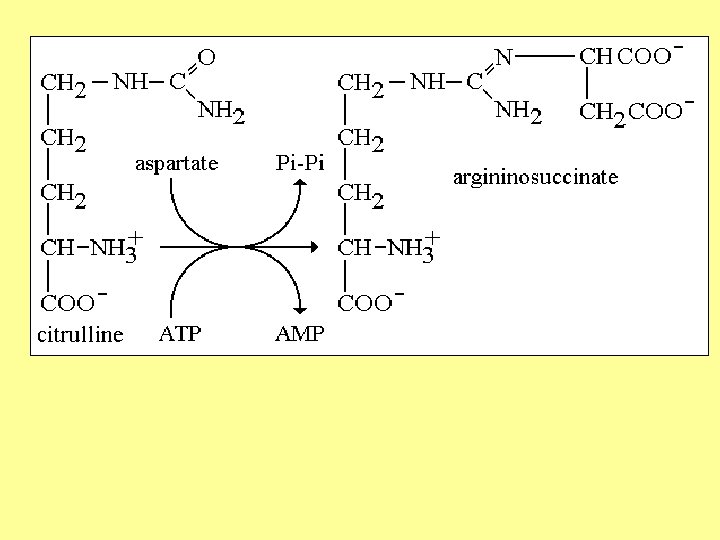
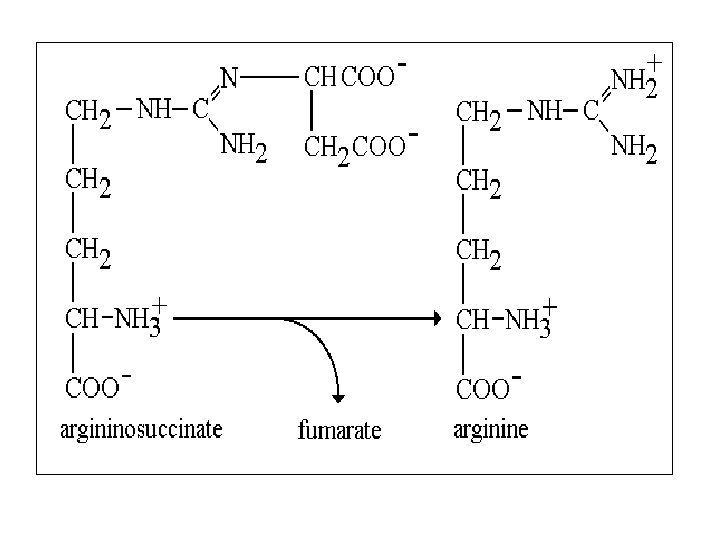
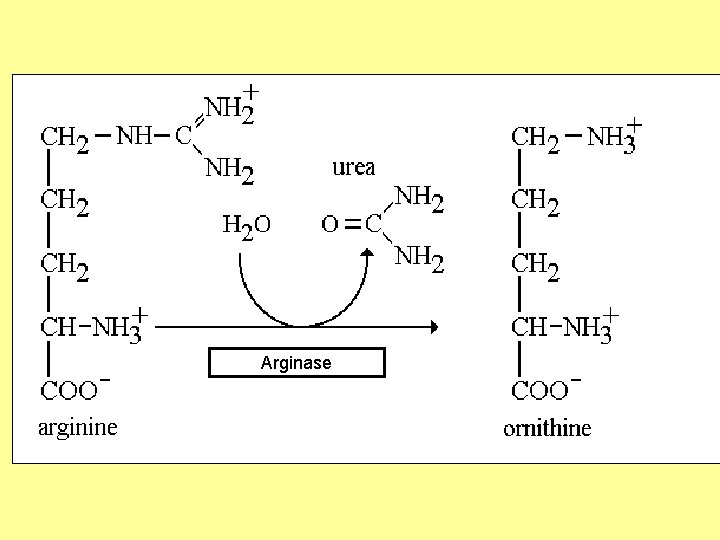
5 - Arginase

Regulation of urea cycle - Long term regulation Increase protein in diet lead to increase all enzyme of urea cycle

Relation between urea and Krebs cycle 1. CO 2 needed for urea derived from Krebs 2. ATP needed for urea derived from Krebs 3. Aspartic give oxaloacetate and vice versa 4. Fumaric produced by urea can oxidized by Krebs

Ammonia intoxication Symptoms 1 - Flapping tremor 2 - Slurring of speech 3 - Plurring of vision 4 - Vomiting in infant 5 - Coma and death

Types 1 - Acquired hyperammoniemia Causes : Liver cirrhosis 2 - Inherited hyperammoniemia 1 - Hyperammonemia type I : ↓ CAP I synthetase 2 - Hyperammonemia type II: ↓ ornithine transcarbamylase
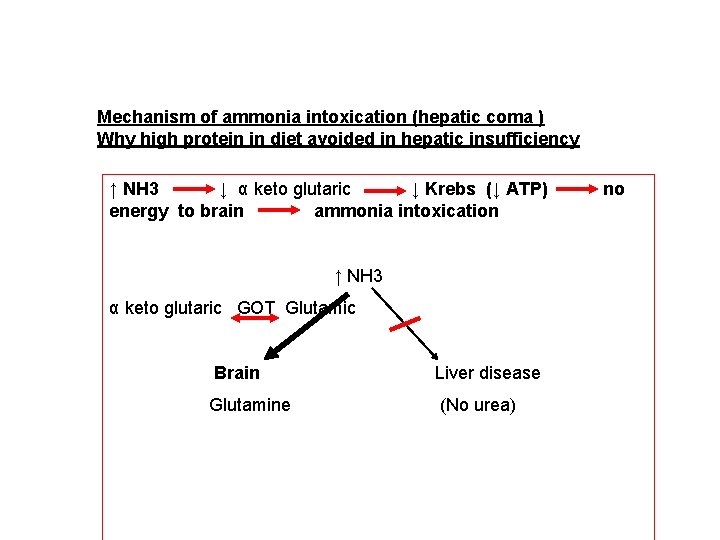
Mechanism of ammonia intoxication (hepatic coma ) Why high protein in diet avoided in hepatic insufficiency ↑ NH 3 ↓ α keto glutaric ↓ Krebs (↓ ATP) energy to brain ammonia intoxication ↑ NH 3 α keto glutaric GOT Glutamic Brain Glutamine Liver disease (No urea) no
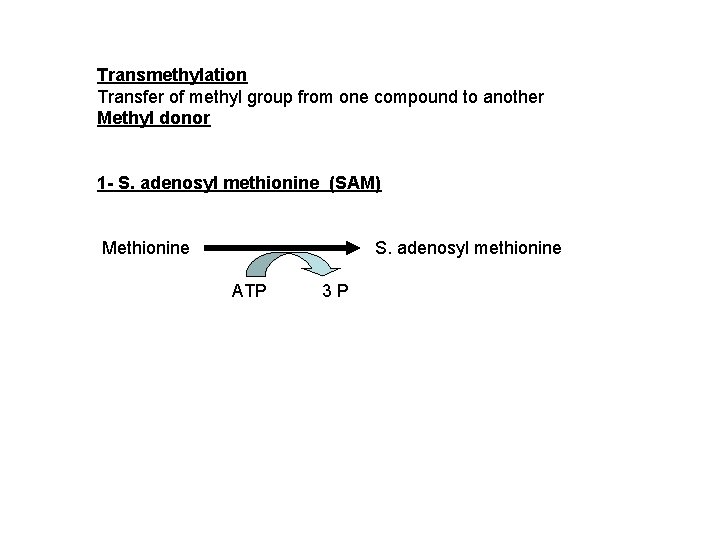
Transmethylation Transfer of methyl group from one compound to another Methyl donor 1 - S. adenosyl methionine (SAM) Methionine S. adenosyl methionine ATP 3 P

Important transmethylation reaction 1 - Norepinephrine SAM epinephrine 2 - Ethanol amine SAM choline 3 - Uracil SAM thymine 4 - Cytosine SAM methylcytosine

Fate of carbon skeleton Glugogenic & Ketogenic AA Glugogenic Isoleucine Lysine Phenyl alanine Tyrosine Typtophan Leucine The remaining AA
- Slides: 38