Protein Function Oxygen Binding Proteins CH 339 K
Protein Function: Oxygen Binding Proteins CH 339 K

Myoglobin • Sperm Whale Myoglobin was the first protein to have its 3 dimensional structure determined – John Kendrew(1958) – Shared the 1962 Nobel in chemistry • Solving the structure wasn’t hard, but getting the samples was a real achievement… Kendrew, JC; Bodo, G; Dintzis, HM; Parrish, RG; Wyckoff, H; Phillips, DC (1958). "A threedimensional model of the myoglobin molecule obtained by x-ray analysis". Nature 181 (4610): 662– 666.
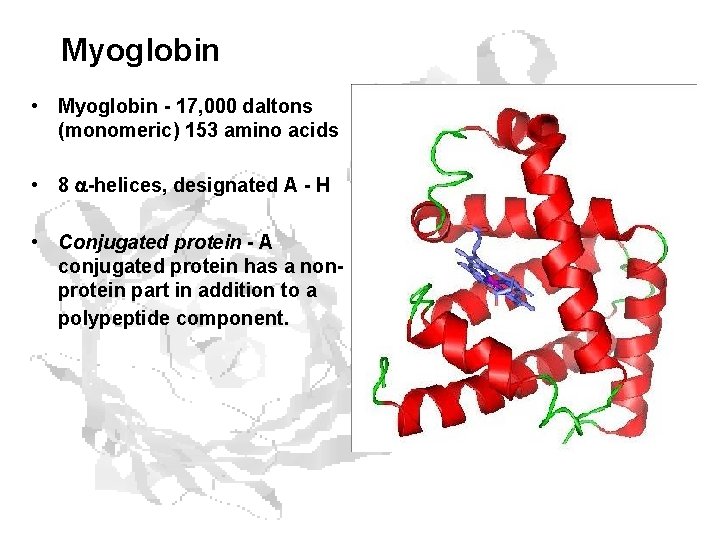
Myoglobin • Myoglobin - 17, 000 daltons (monomeric) 153 amino acids • 8 -helices, designated A - H • Conjugated protein - A conjugated protein has a nonprotein part in addition to a polypeptide component.
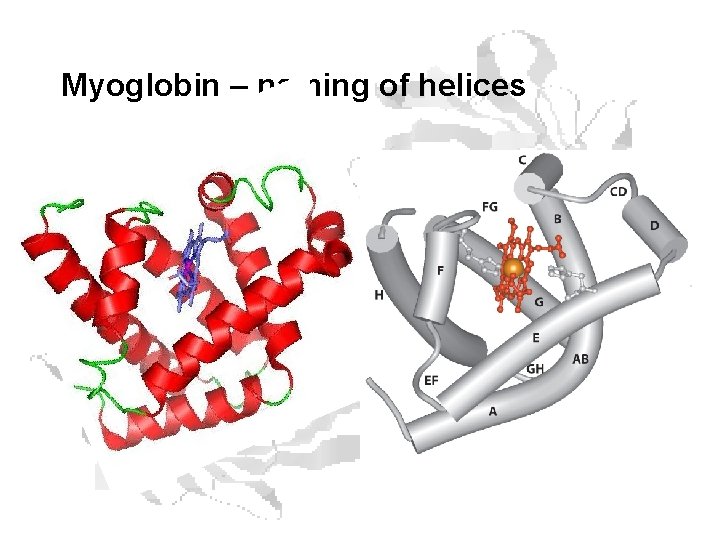
Myoglobin – naming of helices
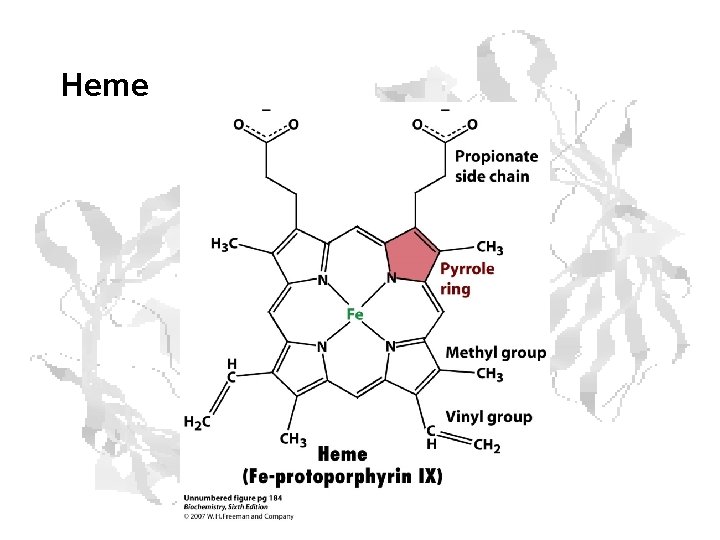
Heme
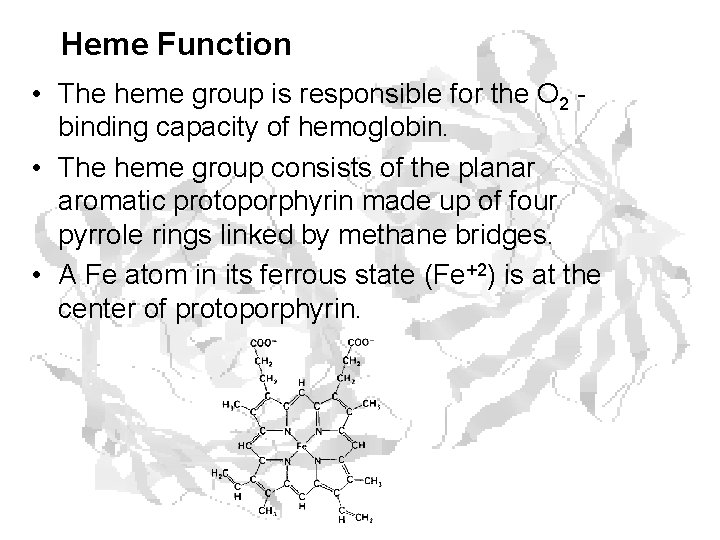
Heme Function • The heme group is responsible for the O 2 binding capacity of hemoglobin. • The heme group consists of the planar aromatic protoporphyrin made up of four pyrrole rings linked by methane bridges. • A Fe atom in its ferrous state (Fe+2) is at the center of protoporphyrin.
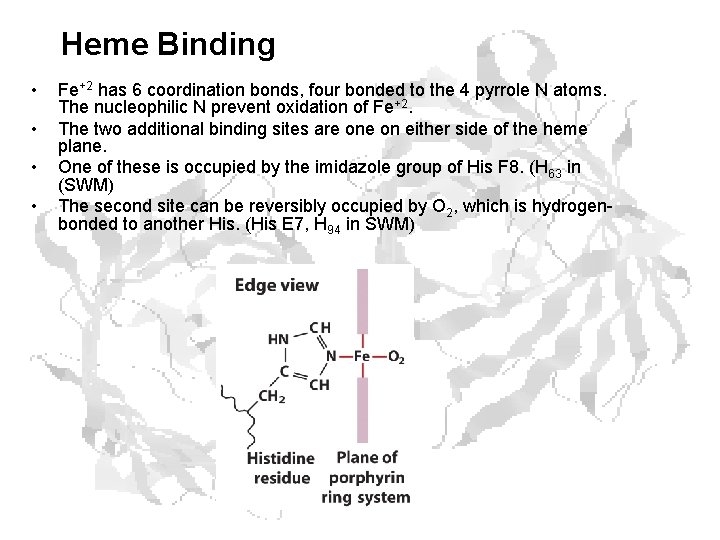
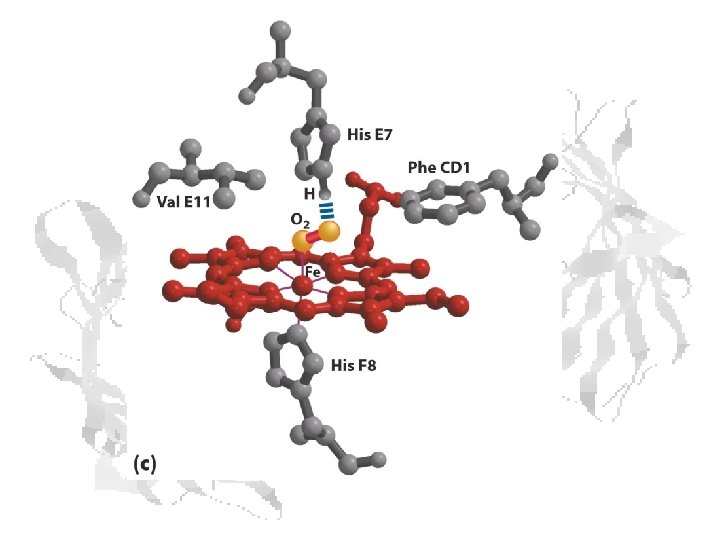
Heme Binding • • Fe+2 has 6 coordination bonds, four bonded to the 4 pyrrole N atoms. The nucleophilic N prevent oxidation of Fe+2. The two additional binding sites are on either side of the heme plane. One of these is occupied by the imidazole group of His F 8. (H 63 in (SWM) The second site can be reversibly occupied by O 2, which is hydrogenbonded to another His. (His E 7, H 94 in SWM)
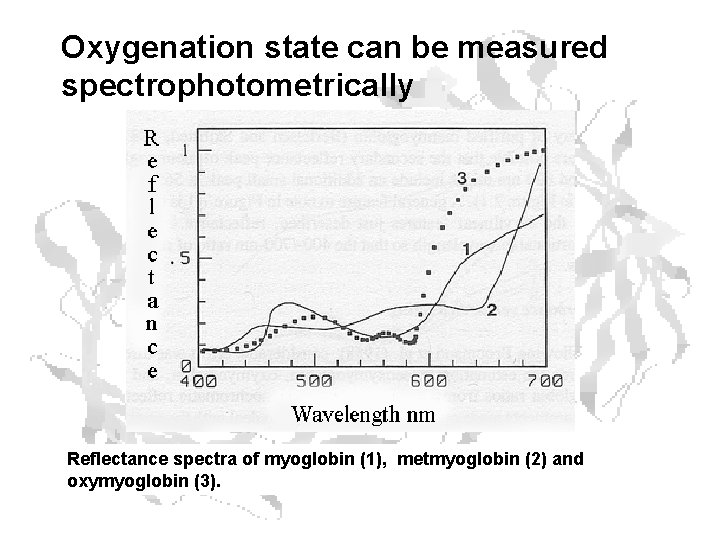
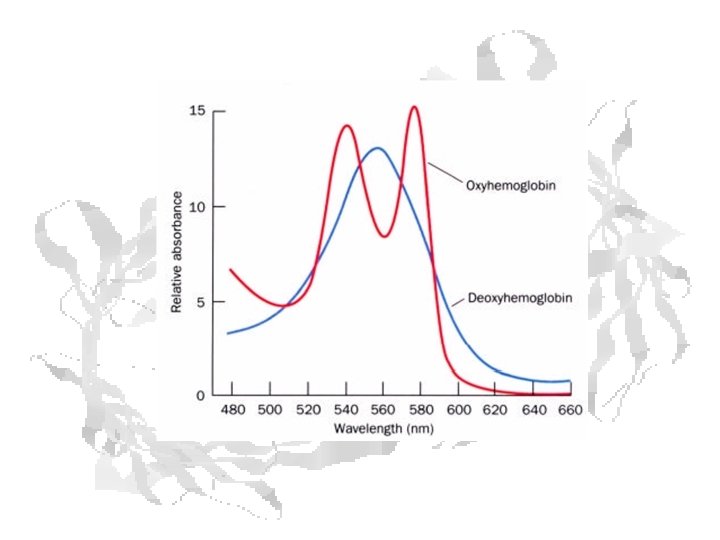
Oxygenation state can be measured spectrophotometrically Reflectance spectra of myoglobin (1), metmyoglobin (2) and oxymyoglobin (3).
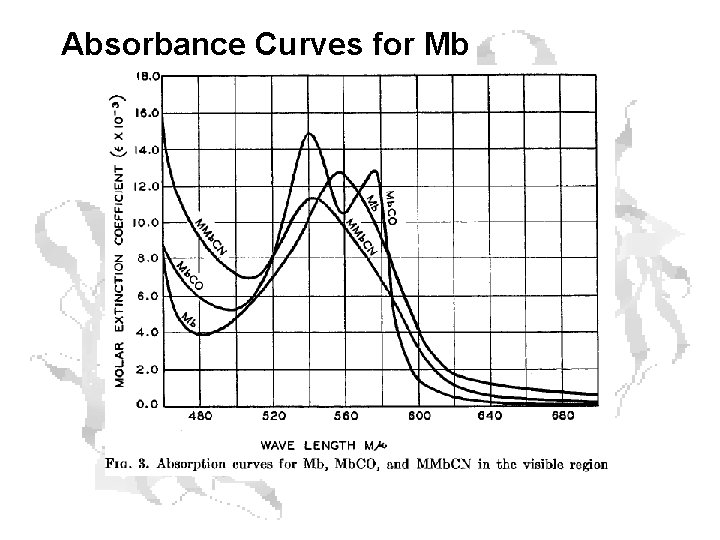
Absorbance Curves for Mb
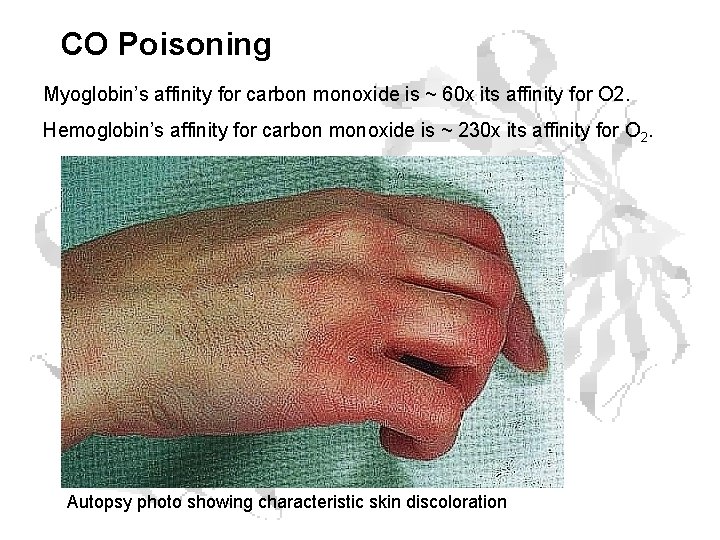
CO Poisoning Myoglobin’s affinity for carbon monoxide is ~ 60 x its affinity for O 2. Hemoglobin’s affinity for carbon monoxide is ~ 230 x its affinity for O 2. Autopsy photo showing characteristic skin discoloration
O 2 Binding Kinetics
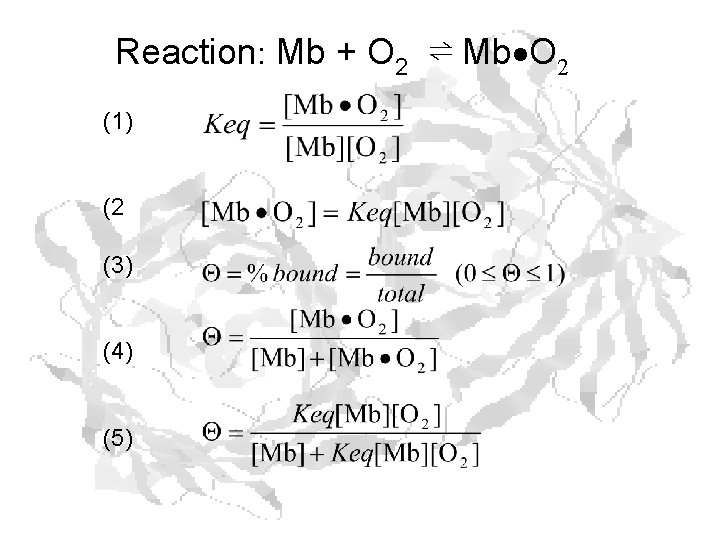
Reaction: Mb + O 2 ⇌ Mb O 2 (1) (2 (3) (4) (5)
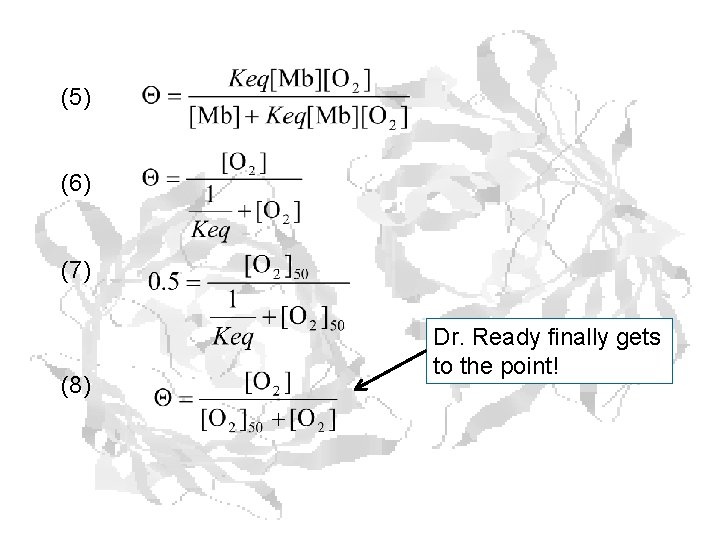
(5) (6) (7) (8) Dr. Ready finally gets to the point!
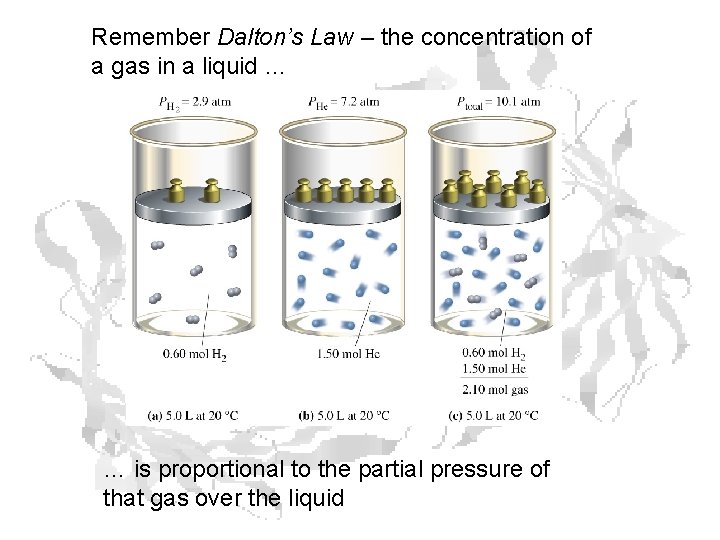
Remember Dalton’s Law – the concentration of a gas in a liquid … … is proportional to the partial pressure of that gas over the liquid
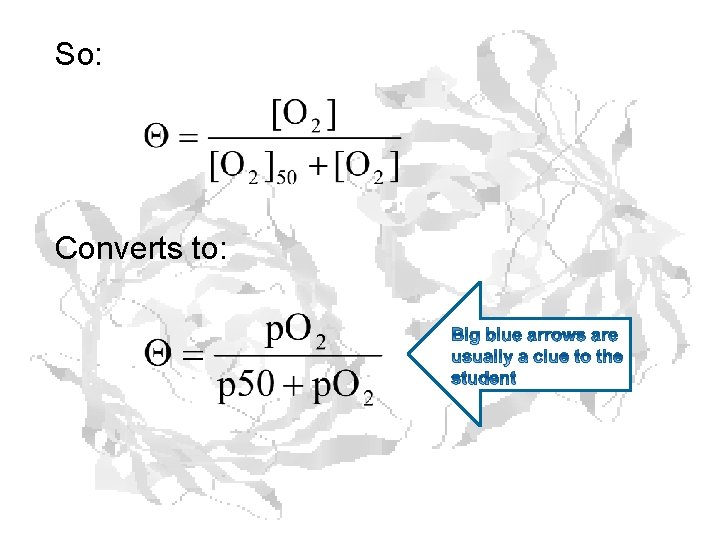
So: Converts to:
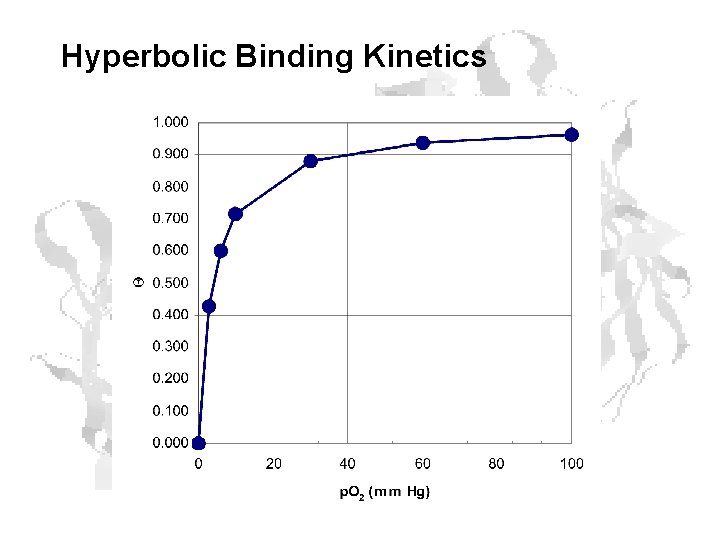
Hyperbolic Binding Kinetics
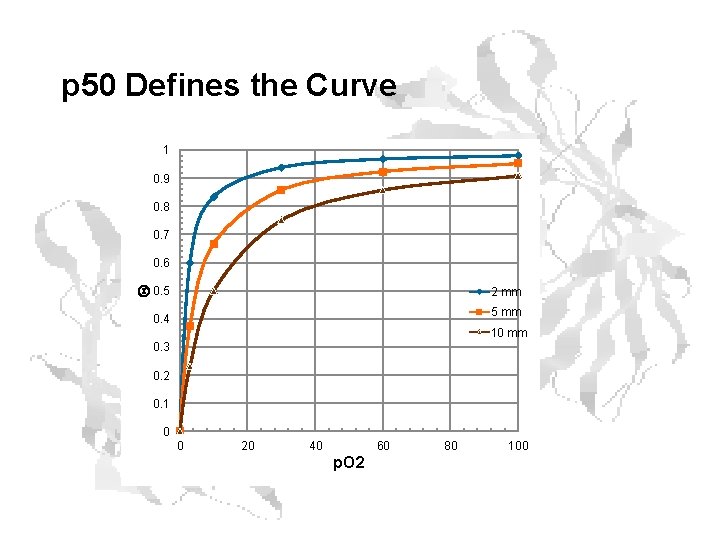
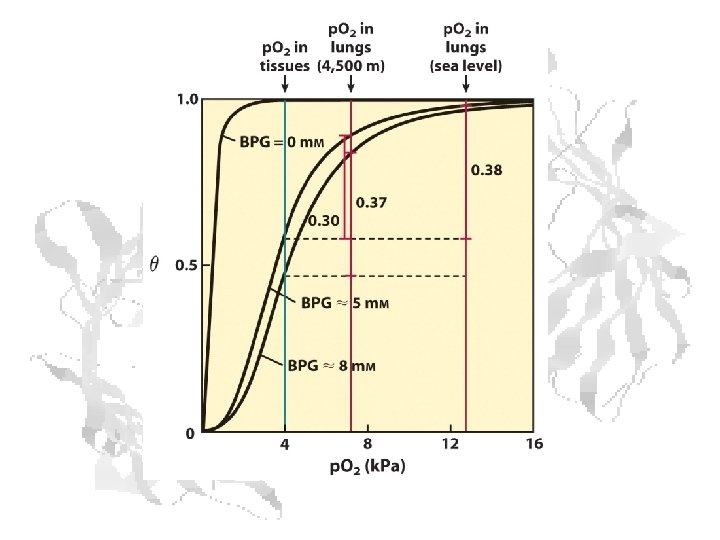
p 50 Defines the Curve 1 0. 9 0. 8 0. 7 Q 0. 6 0. 5 2 mm 5 mm 0. 4 10 mm 0. 3 0. 2 0. 1 0 0 20 40 60 p. O 2 80 100
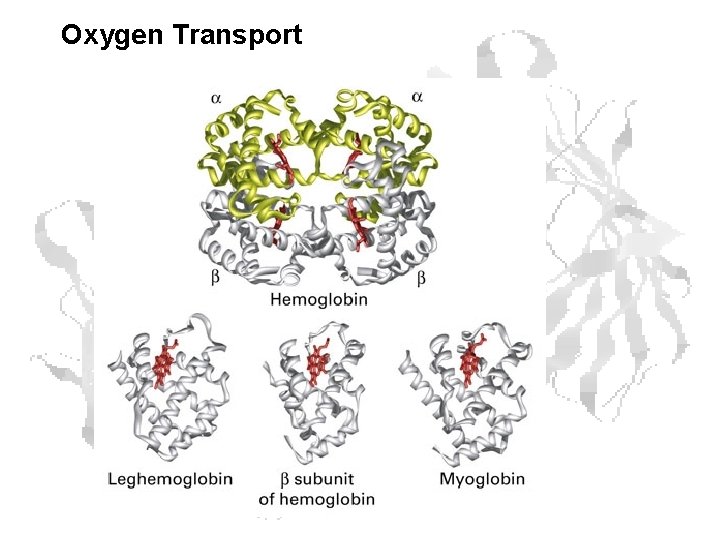
Oxygen Transport
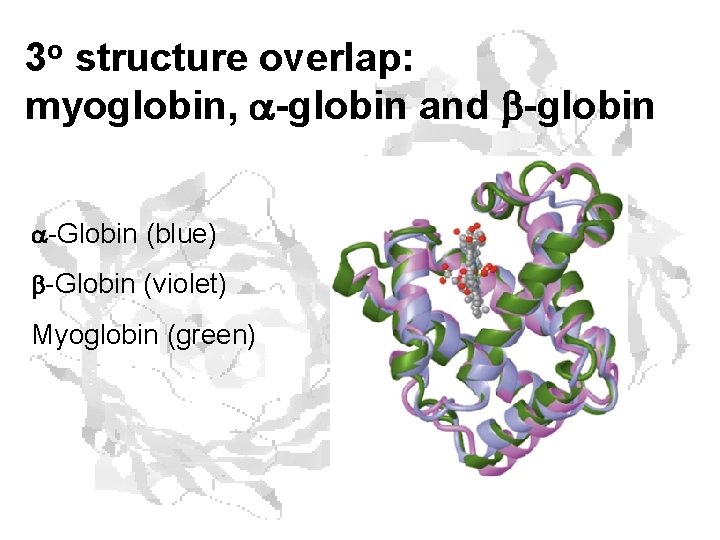
3 o structure overlap: myoglobin, -globin and b-globin -Globin (blue) b-Globin (violet) Myoglobin (green)
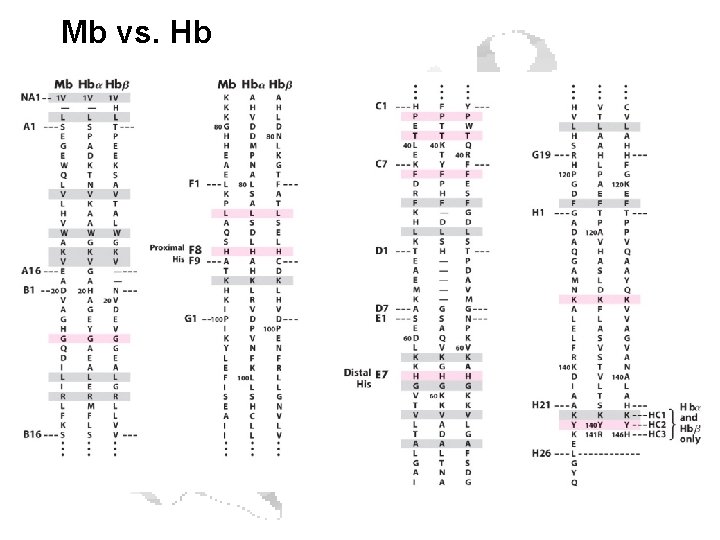
Mb vs. Hb

Hemoglobin O 2 carrying capability • • Erythrocytes/ml blood: 5 billion ( 5 x 109 ) Hemoglobin/red cell: 280 million ( 2. 8 x 108 ) O 2 molecules/hemoglobin: 4 O 2 ml blood: (5 x 109)(2. 8 x 108)(4) = (5. 6 x 1018) or (5. 6 x 1020) molecules of O 2/100 ml blood or ~ 0. 3 g/l or ~ 9 m. M By comparison: • Solubility of O 2 in saline: ~ 0. 007 g/l • or ~ 0. 2 m. M
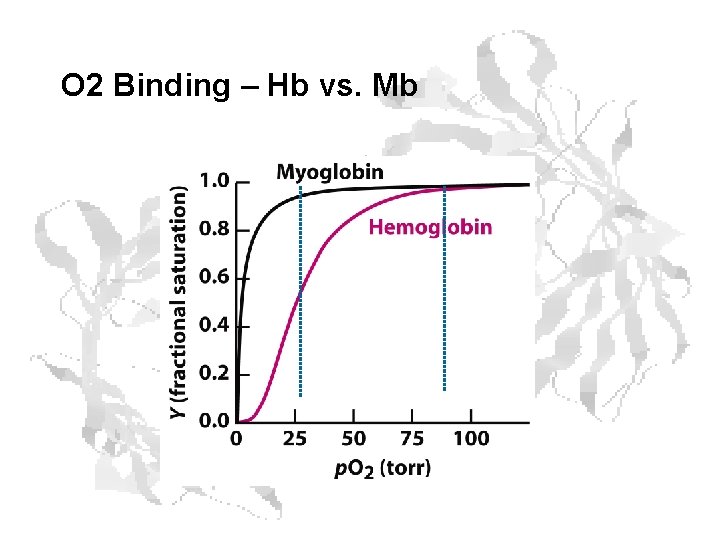
O 2 Binding – Hb vs. Mb
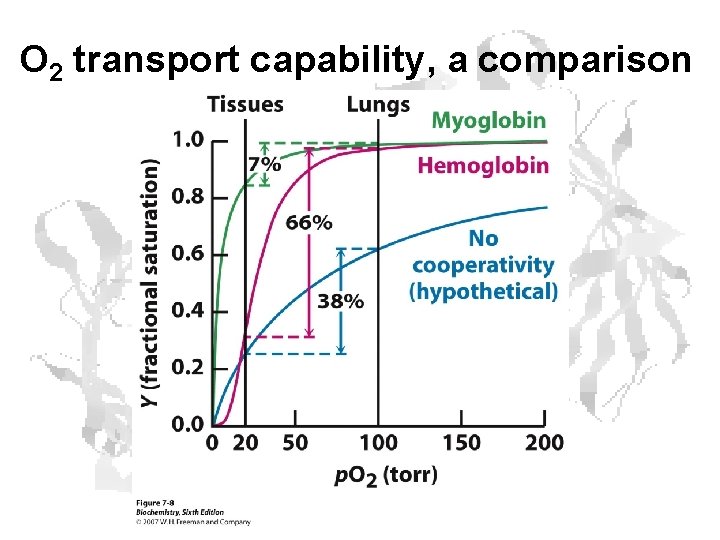
O 2 transport capability, a comparison

Cooperativity Substrate affinity changes with substrate concentration or (rephrased) Substrate affinity changes with substrate binding Characteristic of (many) proteins with multiple binding sites.
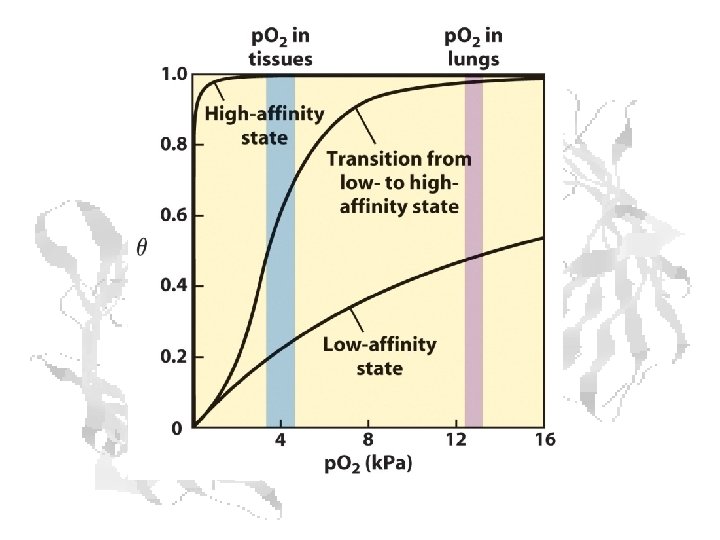
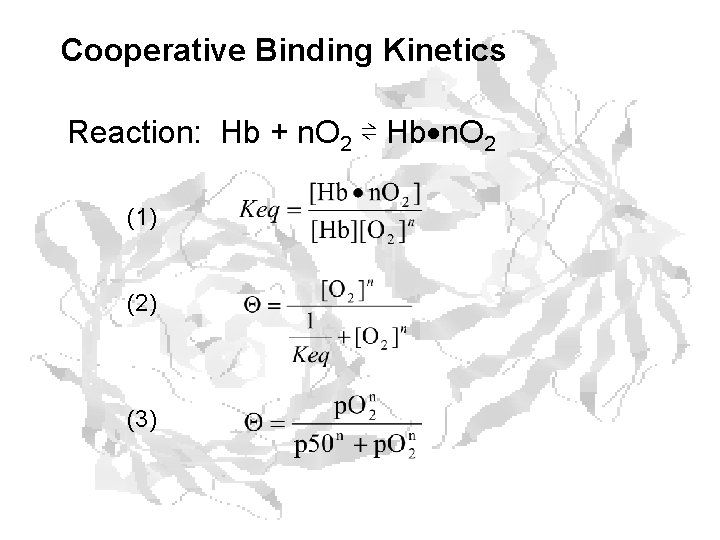
Cooperative Binding Kinetics Reaction: Hb + n. O 2 ⇌ Hb n. O 2 (1) (2) (3)
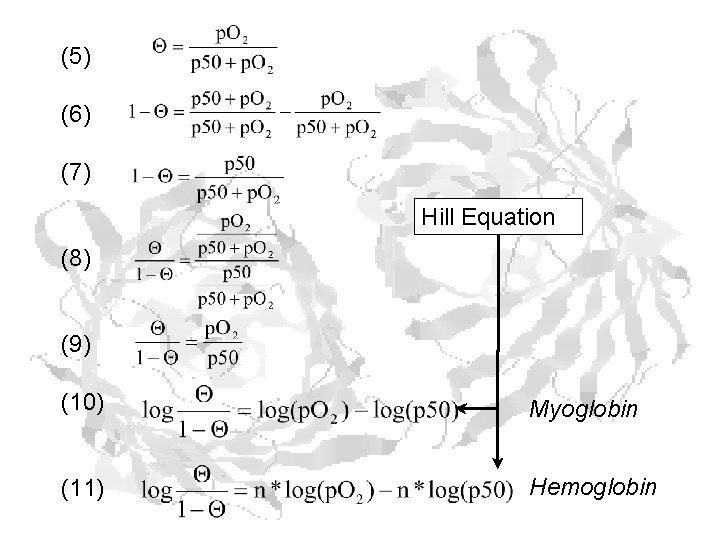
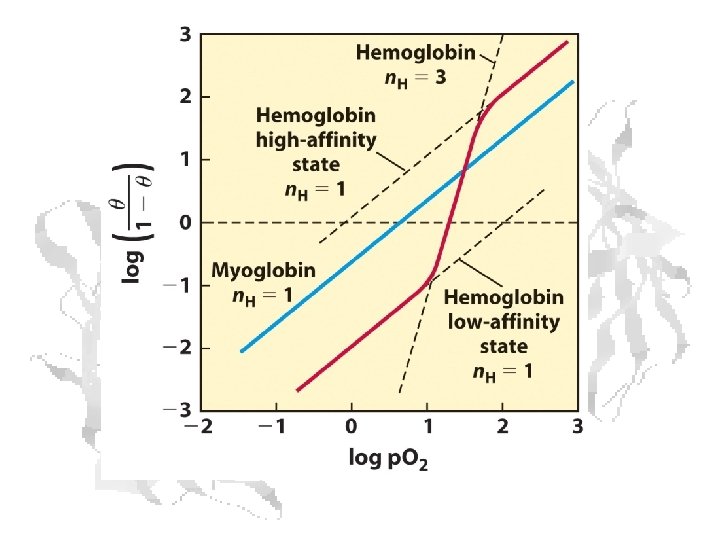
(5) (6) (7) Hill Equation (8) (9) (10) Myoglobin (11) Hemoglobin
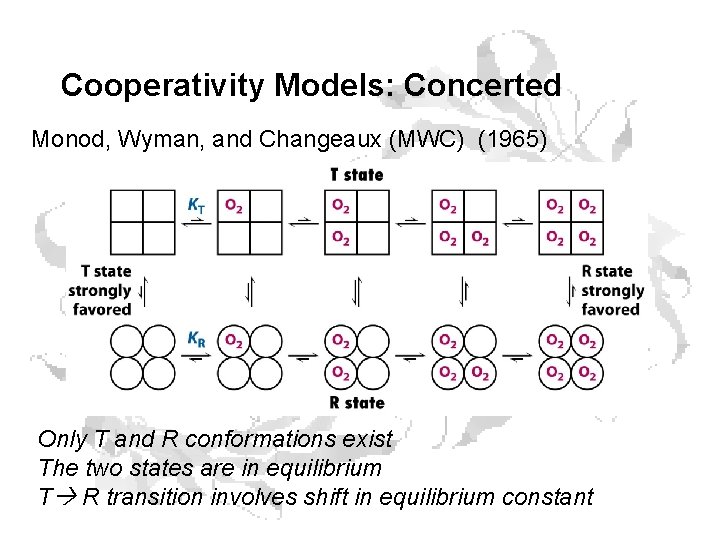
Cooperativity Models: Concerted Monod, Wyman, and Changeaux (MWC) (1965) Only T and R conformations exist The two states are in equilibrium T R transition involves shift in equilibrium constant
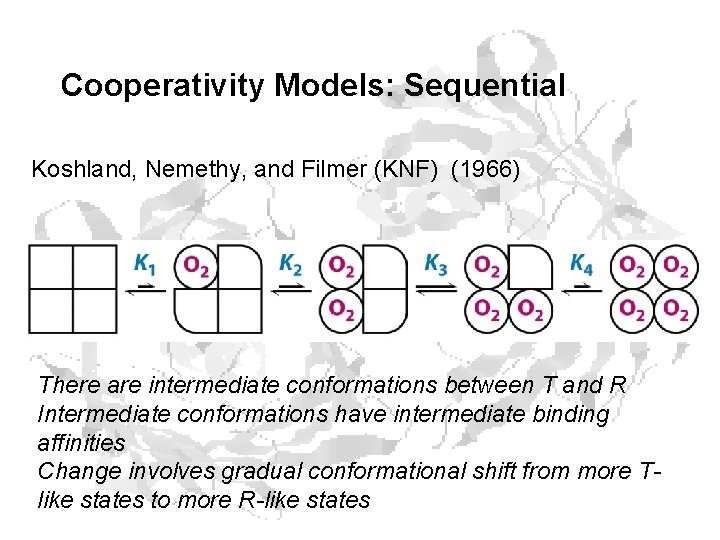
Cooperativity Models: Sequential Koshland, Nemethy, and Filmer (KNF) (1966) There are intermediate conformations between T and R Intermediate conformations have intermediate binding affinities Change involves gradual conformational shift from more Tlike states to more R-like states
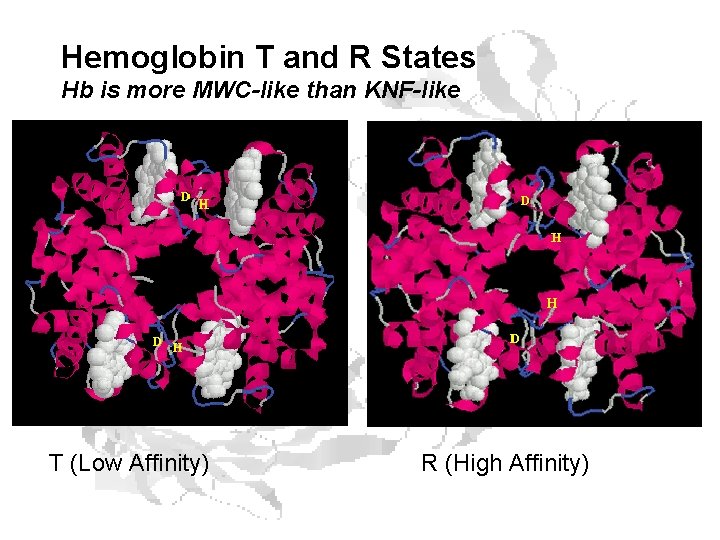
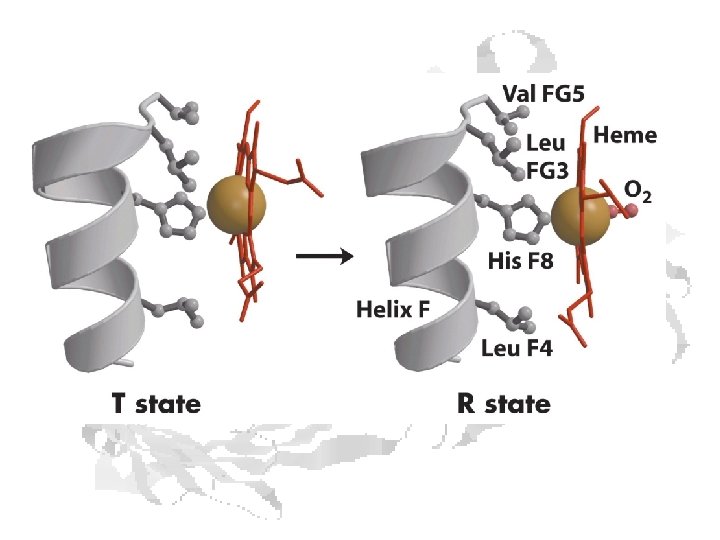
Hemoglobin T and R States Hb is more MWC-like than KNF-like T (Low Affinity) R (High Affinity)
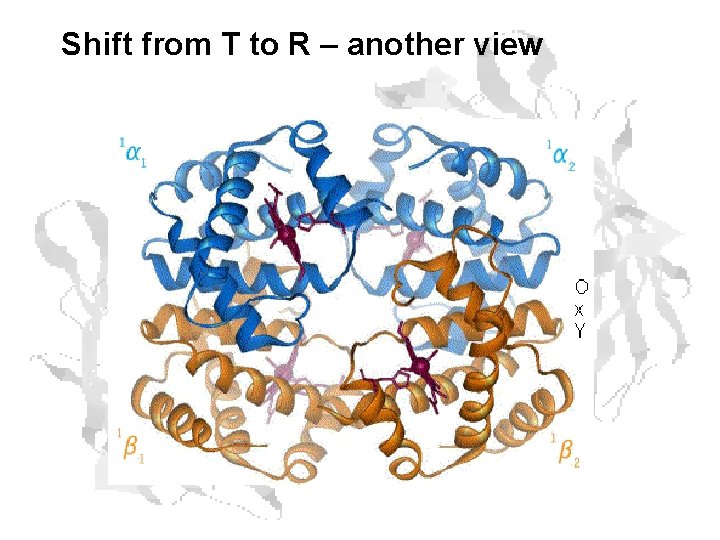
Shift from T to R – another view
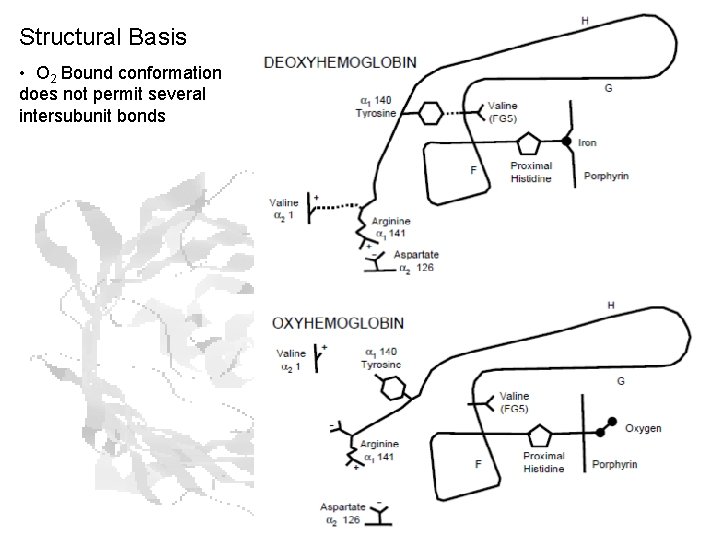
Structural Basis • O 2 Bound conformation does not permit several intersubunit bonds
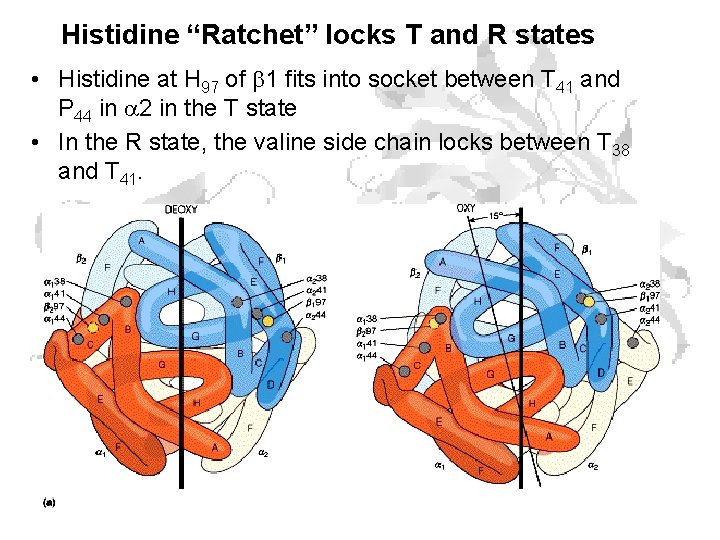
Histidine “Ratchet” locks T and R states • Histidine at H 97 of 1 fits into socket between T 41 and P 44 in a 2 in the T state • In the R state, the valine side chain locks between T 38 and T 41.
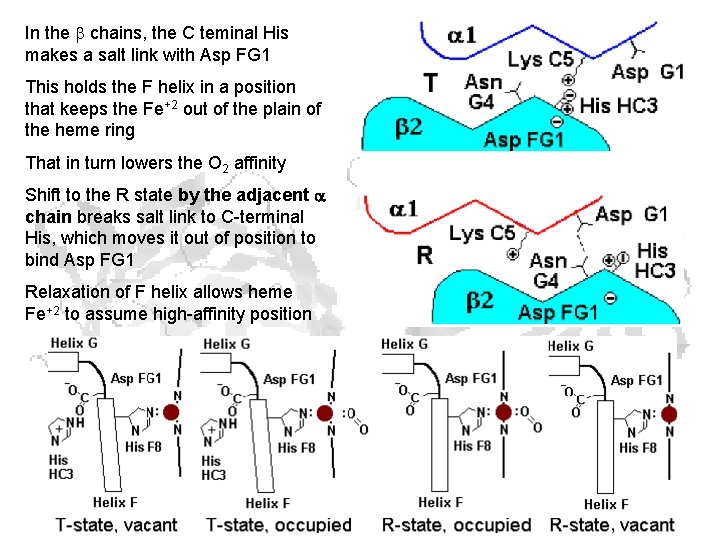
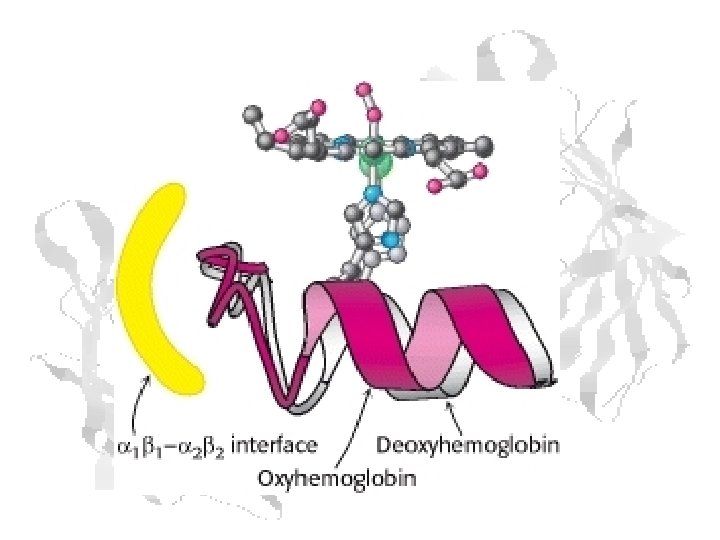
In the chains, the C teminal His makes a salt link with Asp FG 1 This holds the F helix in a position that keeps the Fe+2 out of the plain of the heme ring That in turn lowers the O 2 affinity Shift to the R state by the adjacent chain breaks salt link to C-terminal His, which moves it out of position to bind Asp FG 1 Relaxation of F helix allows heme Fe+2 to assume high-affinity position
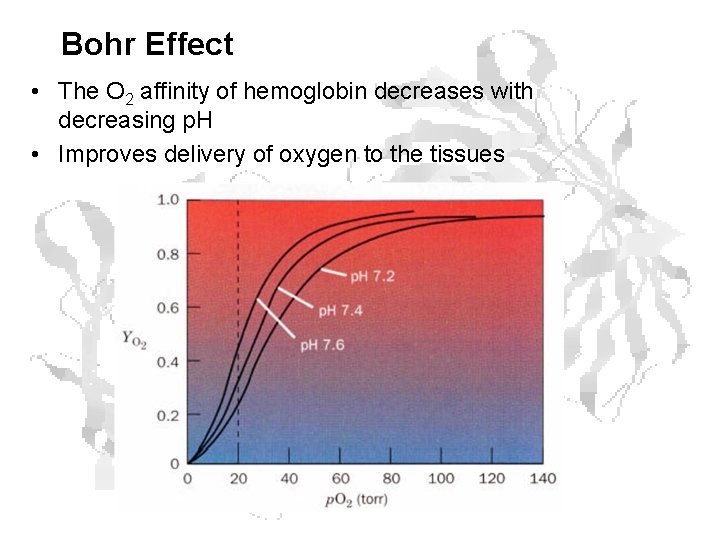
Bohr Effect • The O 2 affinity of hemoglobin decreases with decreasing p. H • Improves delivery of oxygen to the tissues
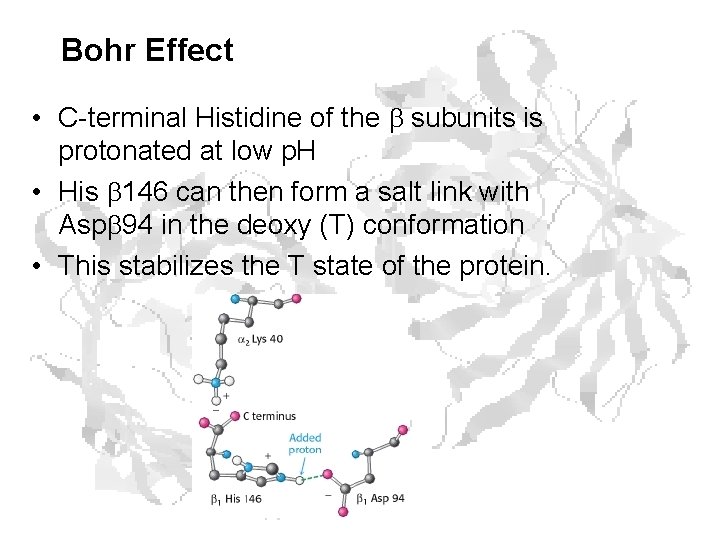
Bohr Effect • C-terminal Histidine of the subunits is protonated at low p. H • His 146 can then form a salt link with Asp 94 in the deoxy (T) conformation • This stabilizes the T state of the protein.
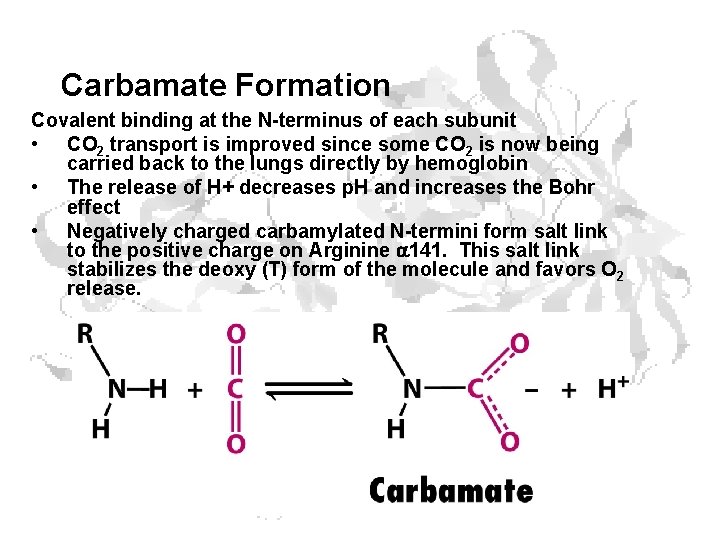
Carbamate Formation Covalent binding at the N-terminus of each subunit • CO 2 transport is improved since some CO 2 is now being carried back to the lungs directly by hemoglobin • The release of H+ decreases p. H and increases the Bohr effect • Negatively charged carbamylated N-termini form salt link to the positive charge on Arginine 141. This salt link stabilizes the deoxy (T) form of the molecule and favors O 2 release.
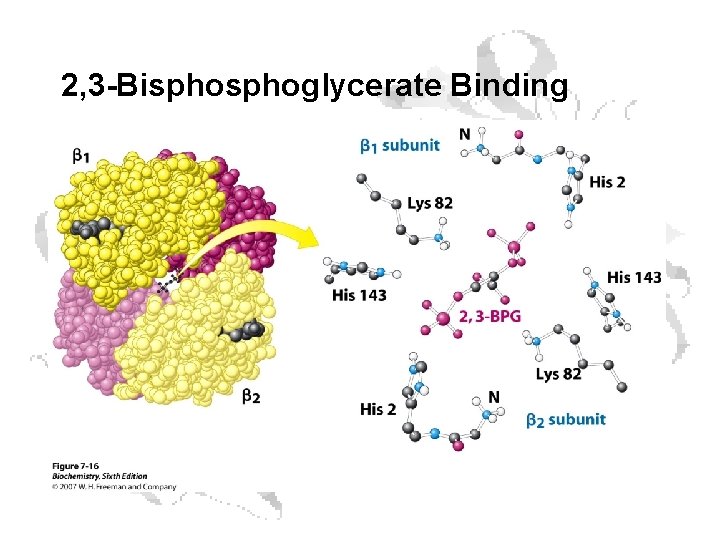
2, 3 -Bisphoglycerate Binding
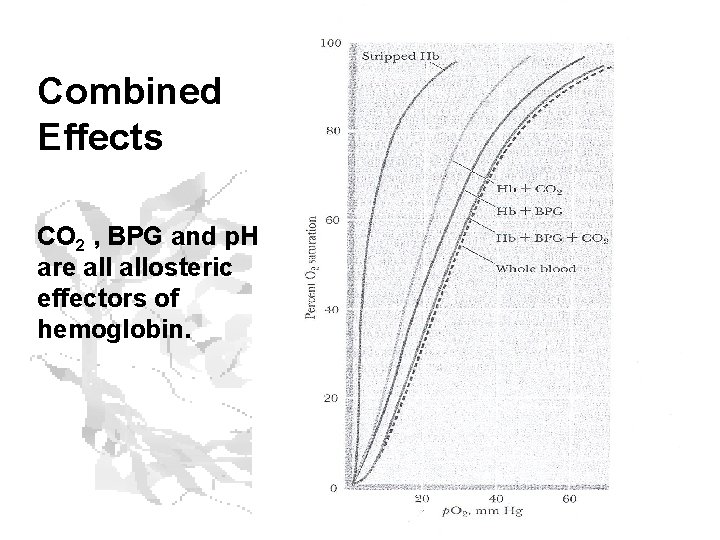
Combined Effects CO 2 , BPG and p. H are allosteric effectors of hemoglobin.

Fetal Hb • • Fetal hemoglobin has 2 α and 2 g chains The g chain is 72% identical to the chain. A His involved in binding to 2, 3 -BPG is replaced with Ser. Thus, fetal Hb has two less + charges than adult Hb. The binding affinity of fetal hemoglobin for 2, 3 -BPG is significantly lower than that of adult hemoglobin Thus, the O 2 saturation capacity of fetal hemoglobin is greater than that of adult hemoglobin This allows for the transfer of maternal O 2 to the developing fetus
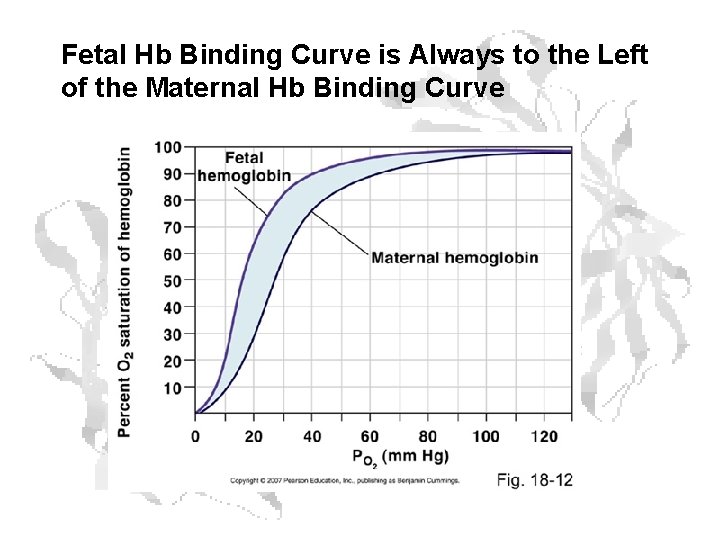
Fetal Hb Binding Curve is Always to the Left of the Maternal Hb Binding Curve
Disease From a Hemoglobin Mutation
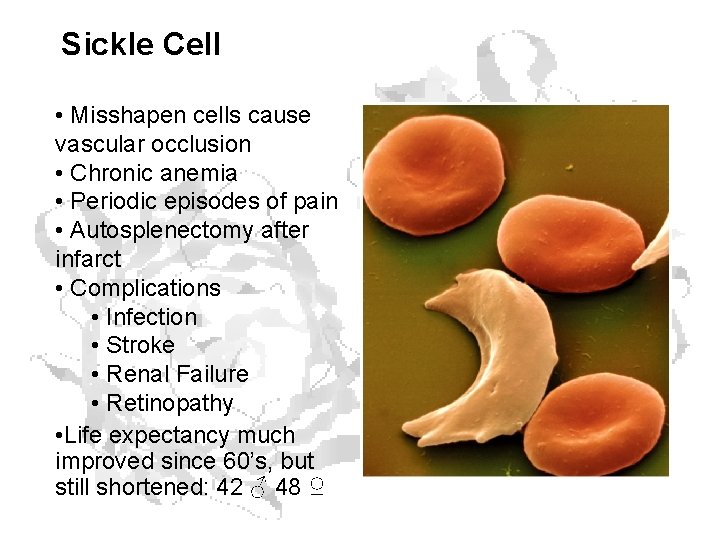
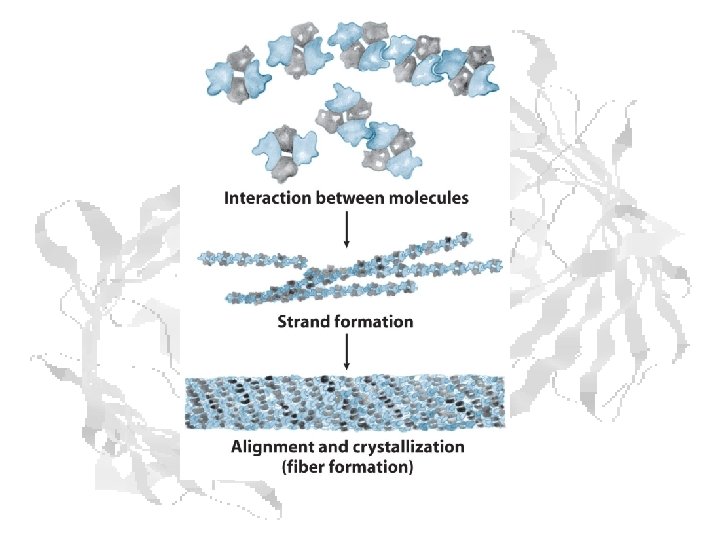
Sickle Cell • Misshapen cells cause vascular occlusion • Chronic anemia • Periodic episodes of pain • Autosplenectomy after infarct • Complications • Infection • Stroke • Renal Failure • Retinopathy • Life expectancy much improved since 60’s, but still shortened: 42 ♂ 48 ♀
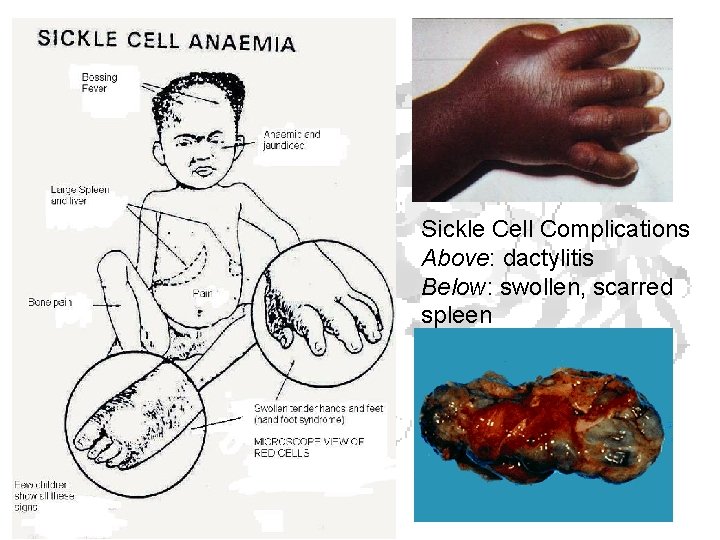
Sickle Cell Complications Above: dactylitis Below: swollen, scarred spleen
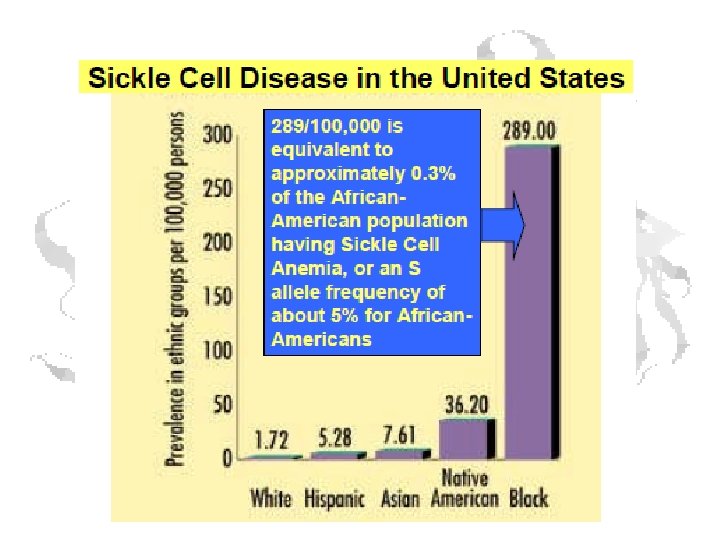
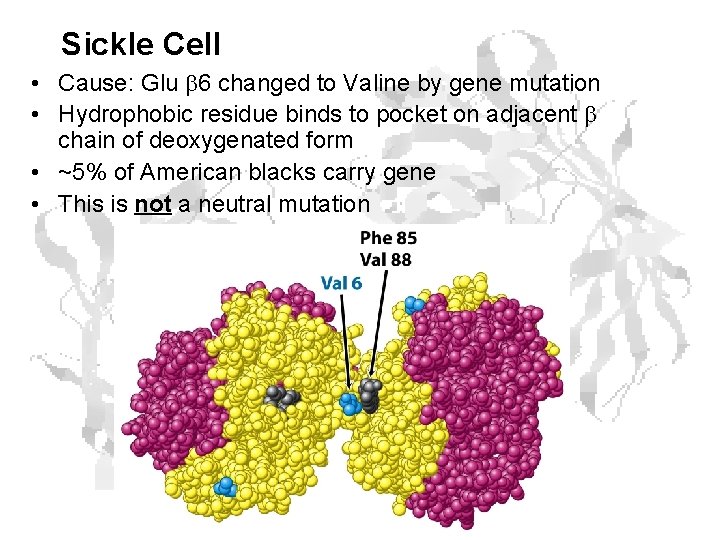
Sickle Cell • Cause: Glu 6 changed to Valine by gene mutation • Hydrophobic residue binds to pocket on adjacent chain of deoxygenated form • ~5% of American blacks carry gene • This is not a neutral mutation
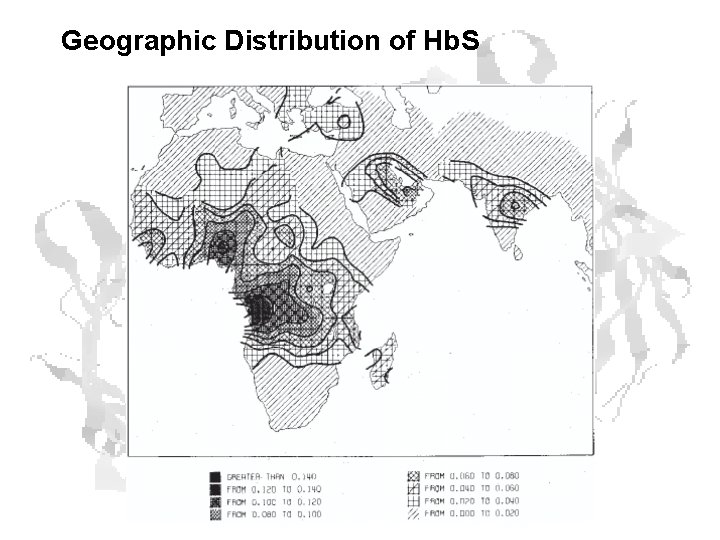
Geographic Distribution of Hb. S
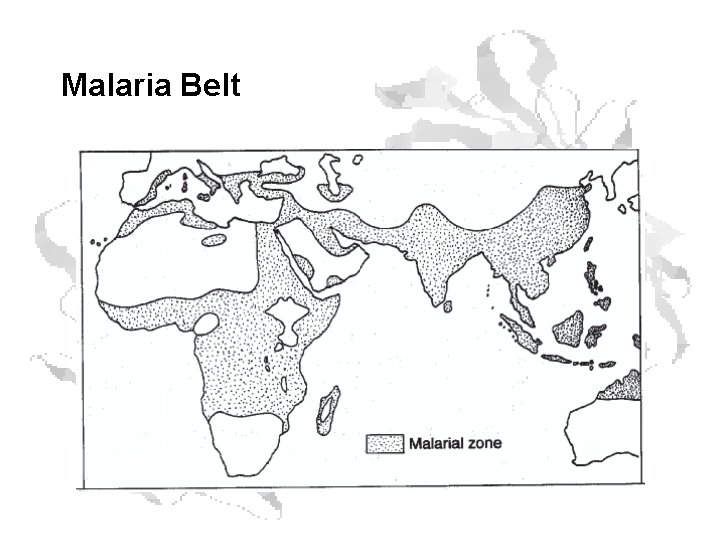
Malaria Belt

Heterozygote Advantage • Heterozygous individuals in Nigeria had a 29% higher likelihood of surviving to adulthood than homozygous normals. • The gene is maintained in the population by selection against both homozygotes.
Other O 2 Binding Proteins (w/o Heme)
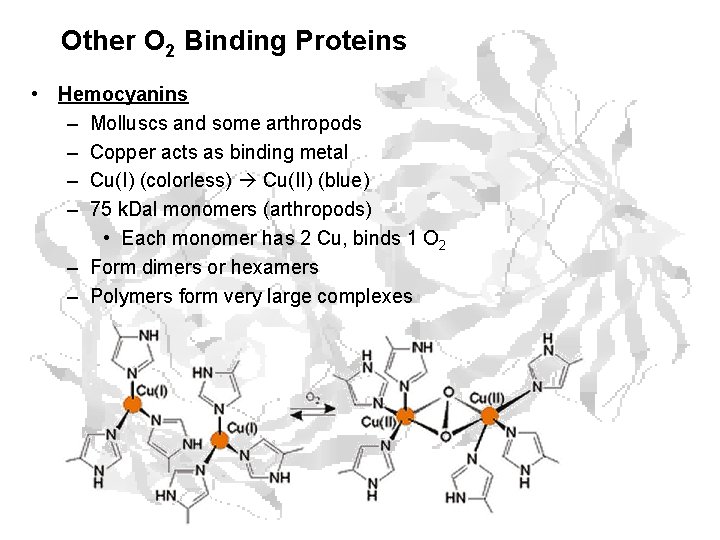
Other O 2 Binding Proteins • Hemocyanins – Molluscs and some arthropods – Copper acts as binding metal – Cu(I) (colorless) Cu(II) (blue) – 75 k. Dal monomers (arthropods) • Each monomer has 2 Cu, binds 1 O 2 – Form dimers or hexamers – Polymers form very large complexes
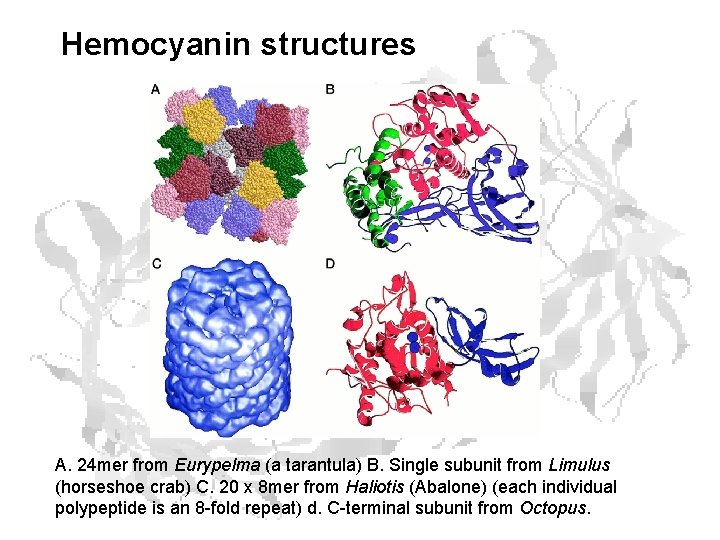
Hemocyanin structures A. 24 mer from Eurypelma (a tarantula) B. Single subunit from Limulus (horseshoe crab) C. 20 x 8 mer from Haliotis (Abalone) (each individual polypeptide is an 8 -fold repeat) d. C-terminal subunit from Octopus.
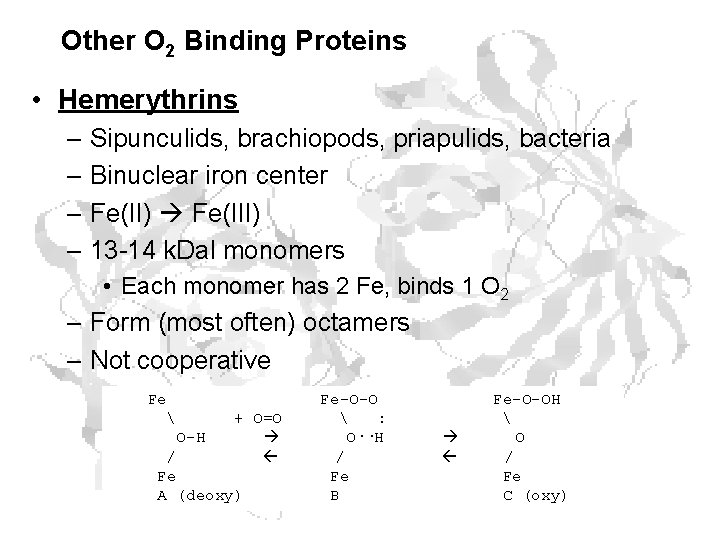
Other O 2 Binding Proteins • Hemerythrins – Sipunculids, brachiopods, priapulids, bacteria – Binuclear iron center – Fe(II) Fe(III) – 13 -14 k. Dal monomers • Each monomer has 2 Fe, binds 1 O 2 – Form (most often) octamers – Not cooperative Fe O-H + O=O / Fe A (deoxy) Fe-O-O : O··H / Fe B Fe-O-OH O / Fe C (oxy)
Sipunculid Priapulid Brachiopod
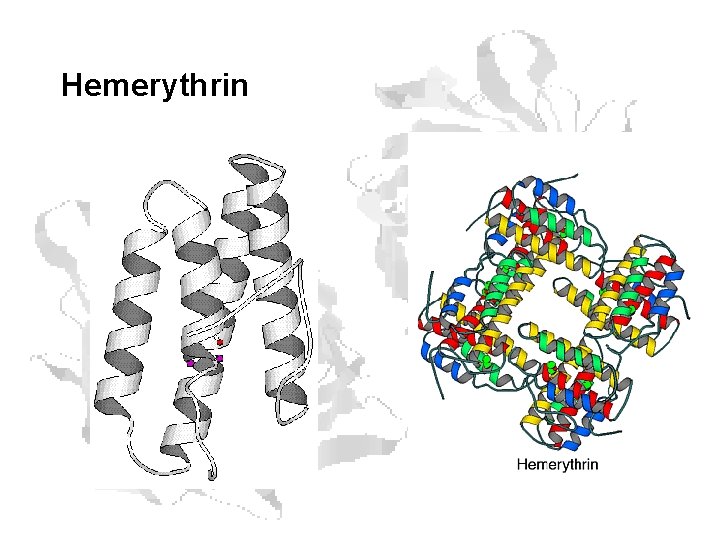
Hemerythrin
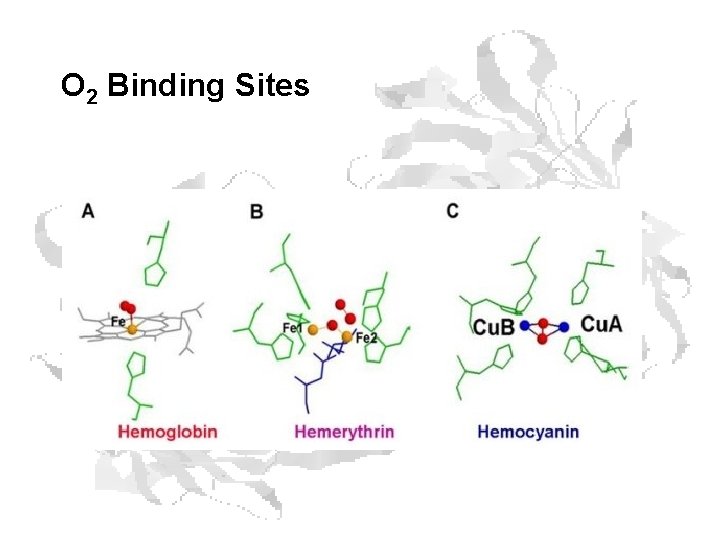
O 2 Binding Sites
Another Heme Protein That Doesn’t Bind O 2
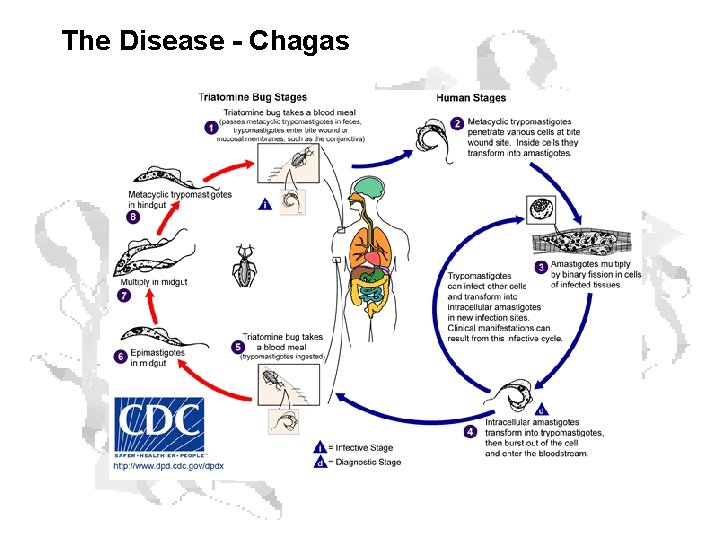
The Disease - Chagas

Symptoms • Acute Phase (weeks to months) • Chronic Phase (10 -20 years post-infection) • • • Swelling at the infection site Fever Fatigue Rash Body aches Headache Loss of appetite Nausea, diarrhea or vomiting Swollen glands Enlargement of your liver or spleen • • Irregular heartbeat Inflamed, enlarged heart (cardiomyopathy) Congestive heart failure Sudden cardiac arrest Difficulty swallowing due to enlarged esophagus Abdominal pain or constipation due to enlarged colon
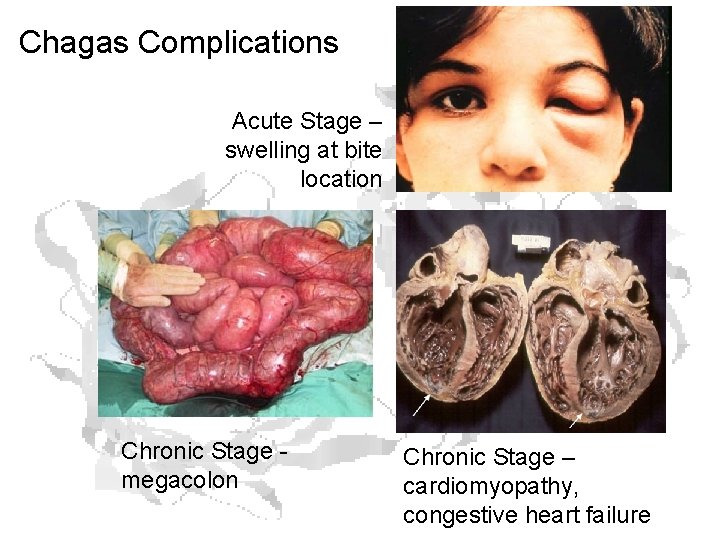
Chagas Complications Acute Stage – swelling at bite location Chronic Stage megacolon Chronic Stage – cardiomyopathy, congestive heart failure
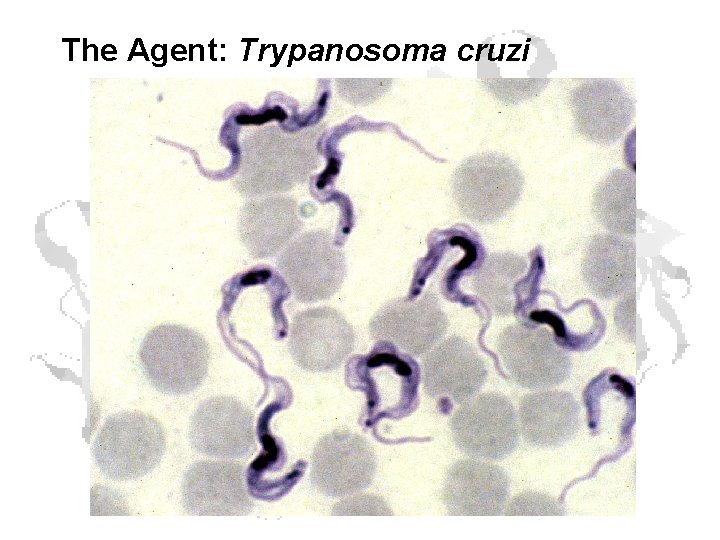
The Agent: Trypanosoma cruzi

The Vector – Cone Nosed Bugs Rhodnius prolixus (Tropical) Triatoma gerstaeckeri (Local)
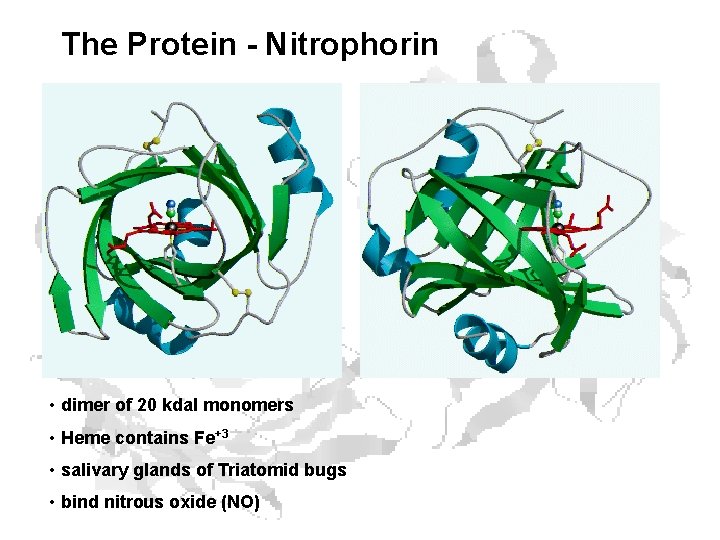
The Protein - Nitrophorin • dimer of 20 kdal monomers • Heme contains Fe+3 • salivary glands of Triatomid bugs • bind nitrous oxide (NO)

Nitrophorin Action – slightly dramatized • Ravenous insect climbs onto face of peacefully sleeping human victim • Inserts hideous proboscis into helpless victim’s flesh • Nitrophorin, with NO bound, is injected into the bite wound • In the alkaline environment of the ghastly wound, NO is released • NO acts as vasodilator, prevents platelet accumulation • Empty binding site on nitrophorin binds histamine • Antihistamine effect prevents irritation to wake hapless blood donor.

Lest you think this is all theoretical… Potential future distribution in Texas From Emerging Infectious Diseases (2003) 9(1): 103 -105
- Slides: 73