Protein Disulfide Isomerase PDI in Cell Signaling Bulent

Protein Disulfide Isomerase (PDI) in Cell Signaling Bulent Mutus Dept. Chemistry & Biochemistry University of Windsor, Canada Thiol Workshop- SFRBM '06 1
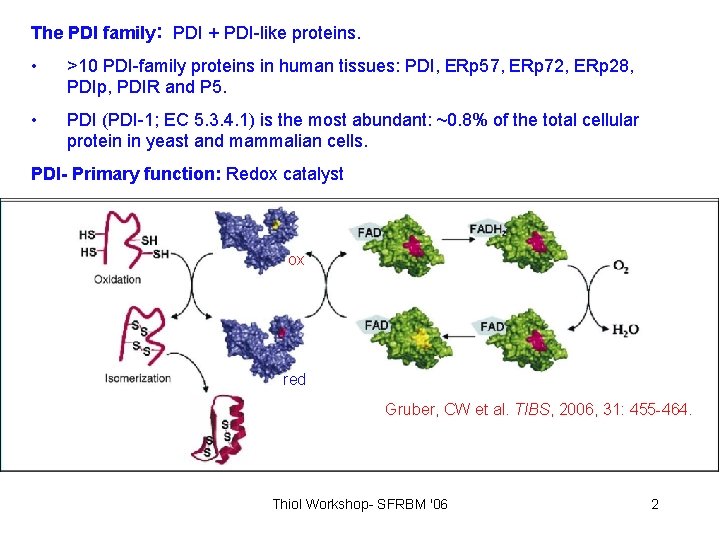
The PDI family: PDI + PDI-like proteins. • >10 PDI-family proteins in human tissues: PDI, ERp 57, ERp 72, ERp 28, PDIp, PDIR and P 5. • PDI (PDI-1; EC 5. 3. 4. 1) is the most abundant: ~0. 8% of the total cellular protein in yeast and mammalian cells. PDI- Primary function: Redox catalyst ox red Gruber, CW et al. TIBS, 2006, 31: 455 -464. Thiol Workshop- SFRBM '06 2

Tian, G. et al. (2006) Cell 124: 61– 73. Thiol Workshop- SFRBM '06 3
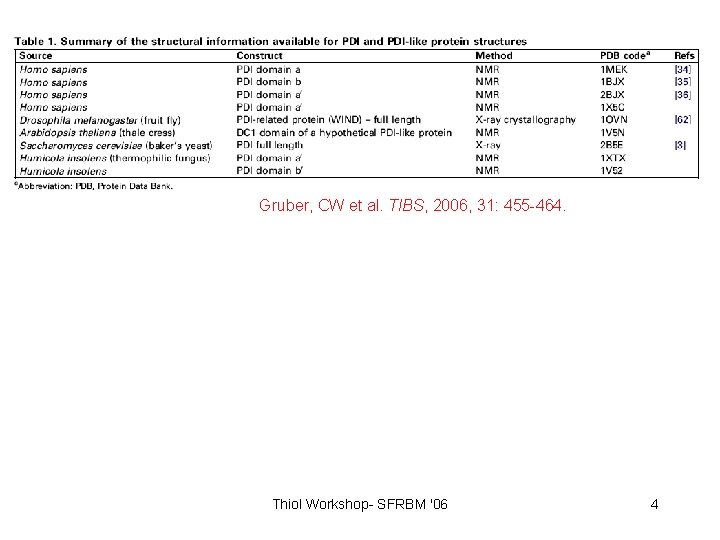
Gruber, CW et al. TIBS, 2006, 31: 455 -464. Thiol Workshop- SFRBM '06 4

PDI- other functions (postulated from in vitro studies): Chaperone role: prevents protein aggregation and retention in ER PDI leaks out and is attached via hydrophobic interactions to cell surfaces (cs. PDI) Mediation of platelet function by ps. PDI: • • Essex DW, Chen K, Swiatkowska M. Localization of protein disulfide isomerase to the external surface of the platelet plasma membrane. Blood. 1995, 86: 2168 -73. Essex DW, et al. Protein disulfide isomerase catalyzes the formation of disulfidelinked complexes of vitronectin with thrombin-antithrombin. Biochemistry. 1999, 38: 10398 -405. Essex DW, Li M. Protein disulphide isomerase mediates platelet aggregation and secretion. Br J Haematol. 1999, 104: 448 -54. Milev Y, Essex DW. Links Protein disulfide isomerase catalyzes the formation of disulfide-linked complexes of thrombospondin-1 with thrombin-antithrombin III. Arch Biochem Biophys. 1999, 361: 120 -6. Lahav J, et al. Protein disulfide isomerase mediates integrin-dependent adhesion. FEBS Lett. 2000 475: 89 -92. Lahav J, et al. Sustained integrin ligation involves extracellular free sulfhydryls and enzymatically catalyzed disulfide exchange. Blood. 2002, 100: 2472 -8. Burgess JK, et al. Physical proximity and functional association of glycoprotein 1 balpha and protein-disulfide isomerase on the platelet plasma membrane. J Biol Chem. 2000, 27: 9758 -66. Thiol Workshop- SFRBM '06 5

PDI- other functions (postulated from in vitro studies): Mediation of platelet function by ps. PDI: (continued) • Lahav J, et al. Enzymatically catalyzed disulfide exchange is required for platelet adhesion to collagen via integrin alpha 2 beta 1. Blood. 2003, 102: 2085 -92. Mediation of the transport of NO-equivalents (RSNO) across membranes: • • • A. Zai, M. A. Rudd, A. W. Scribner, J. Loscalzo, Cell-surface protein disulfide isomerase catalyzes transnitrosation and regulates intracellular transfer of nitric oxide, J. Clin. Invest. 1999, 103: 393– 399. N. Ramachandran, P. Root, X. M. Jiang, P. J. Hogg, B. Mutus, Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface protein disulfide isomerase, Proc. Natl. Acad. Sci. USA 2001, 98: 9539– 9544. Root, P et al. Platelet cell-surface protein disulphide-isomerase mediated Snitrosoglutathione consumption. Biochem J. 2004, 382: 575 -80. Sliskovic I, Raturi A, Mutus B. Characterization of the S-denitrosation activity of protein disulfide isomerase. J Biol Chem. 2005, 280: 8733 -41. Shah CM, Bell SE, Locke IC, Chowdrey HS, Gordge MP. Interactions between cell surface protein disulphide isomerase and S-nitrosoglutathione during nitric oxide delivery. Nitric Oxide. 2006 Aug 11; [Epub ahead of print] Thiol Workshop- SFRBM '06 6

Presentation Focus: Methodology utilized to demonstrate cell signaling roles of cs. PDI. 1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface protein disulfide isomerase. 2) Characterization of the S-denitrosation activity of protein disulfide isomerase. 3) Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D) (new work). Thiol Workshop- SFRBM '06 7
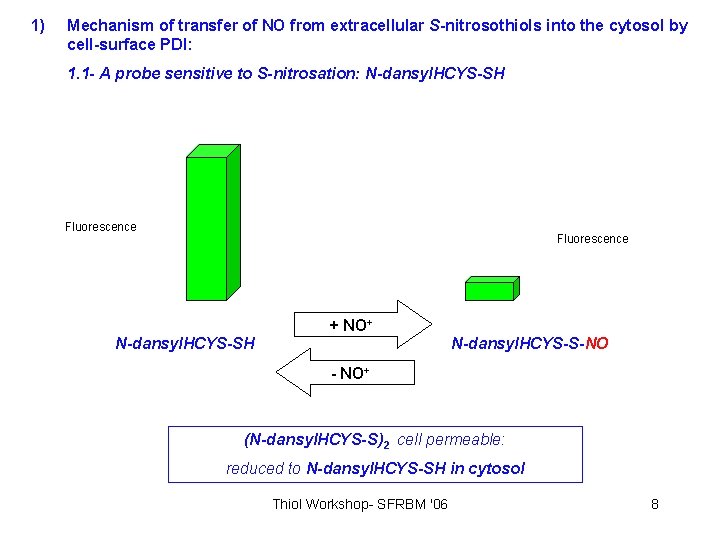
1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface PDI: 1. 1 - A probe sensitive to S-nitrosation: N-dansyl. HCYS-SH Fluorescence + NO+ N-dansyl. HCYS-SH N-dansyl. HCYS-S-NO - NO+ (N-dansyl. HCYS-S)2 cell permeable: reduced to N-dansyl. HCYS-SH in cytosol Thiol Workshop- SFRBM '06 8

1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface PDI: 1. 1 - A probe sensitive to S-nitrosation: N-dansyl. HCYS-SH cont. Thiol Workshop- SFRBM '06 9

1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface PDI: 1. 2 - HT 1080 fibroblastomas underexressing/overexpressing PDI Thiol Workshop- SFRBM '06 10

1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface PDI: 1. 3 - a vicinal thiol-specific agent: GSAO +GSAO -GSAO Thiol Workshop- SFRBM '06 11

1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface PDI: 1. 3 - a N 2 O 3 quencher: -tocopherol + -tocopherol - -tocopherol Thiol Workshop- SFRBM '06 12

1) Mechanism of transfer of NO from extracellular S-nitrosothiols into the cytosol by cell-surface PDI: Thiol Workshop- SFRBM '06 13
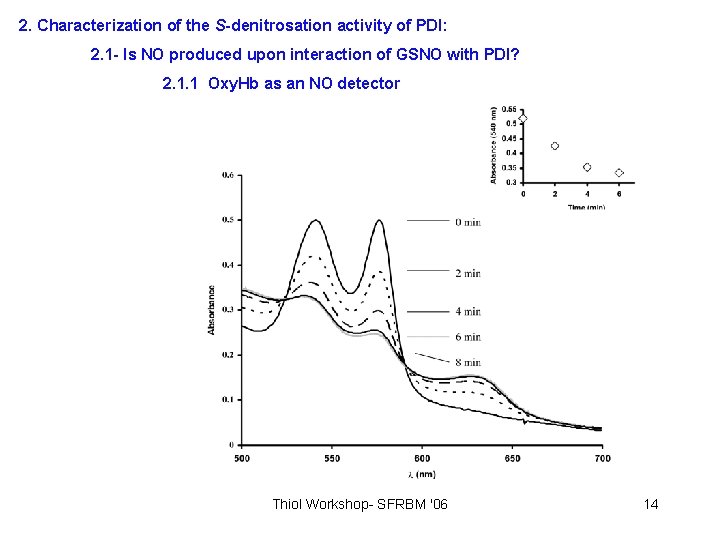
2. Characterization of the S-denitrosation activity of PDI: 2. 1 - Is NO produced upon interaction of GSNO with PDI? 2. 1. 1 Oxy. Hb as an NO detector Thiol Workshop- SFRBM '06 14

2. Characterization of the S-denitrosation activity of PDI: 2. 1 - Is NO and thiyl/dithiyl radicals produced upon interaction of GSNO with PDI? 2. 1. 1 Oxy. Hb as an NO detector 2. 1. 2 Ac-Tempo fluorogenic thiyl/dithiyl radical probe Thiol Workshop- SFRBM '06 15

2. Characterization of the S-denitrosation activity of PDI: 2. 1 - Is NO produced upon interaction of GSNO with PDI? 2. 1. 3 NO electrode Thiol Workshop- SFRBM '06 16

2. Characterization of the S-denitrosation activity of PDI: 2. 2 - PDI Thiol reactivity and PDI-SNO formation 2. 2. 1 - DTNB 2. 2. 2 - UV/vis Thiol Workshop- SFRBM '06 17
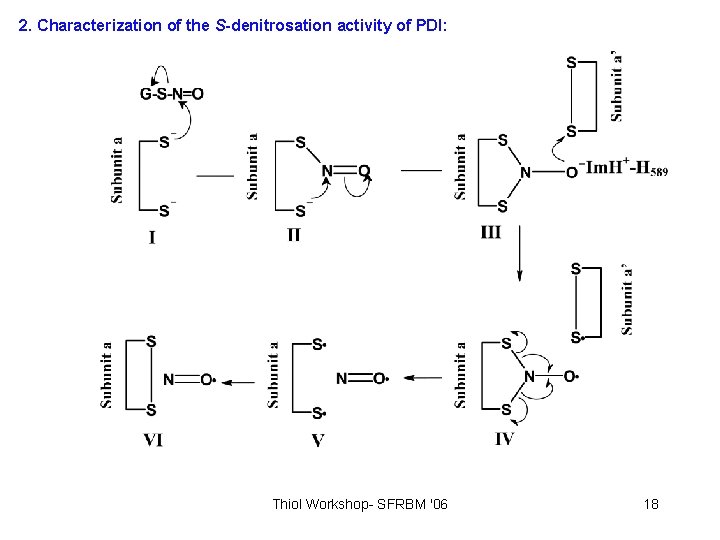
2. Characterization of the S-denitrosation activity of PDI: Thiol Workshop- SFRBM '06 18

3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Background: PDI essential for platelet aggregation . Essex et al. Biochemistry. 2001, 40: 6070 -5 Thiol Workshop- SFRBM '06 19
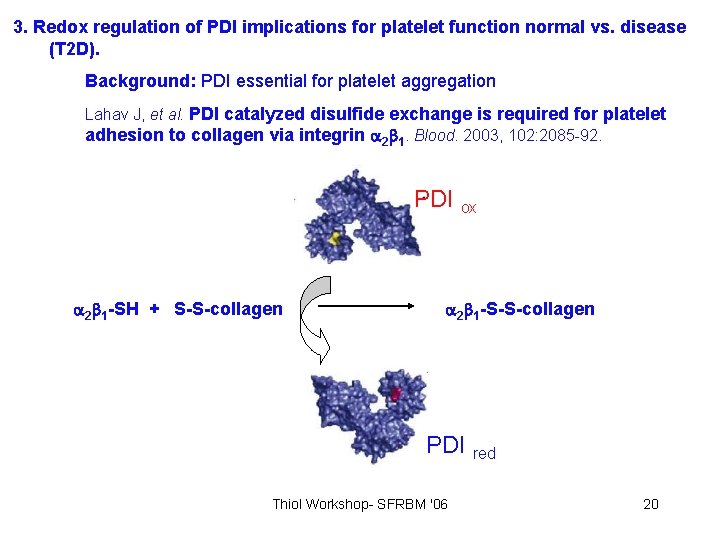
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Background: PDI essential for platelet aggregation Lahav J, et al. PDI catalyzed disulfide exchange is required for platelet adhesion to collagen via integrin 2 1. Blood. 2003, 102: 2085 -92. PDI ox 2 1 -SH + S-S-collagen 2 1 -S-S-collagen PDI red Thiol Workshop- SFRBM '06 20
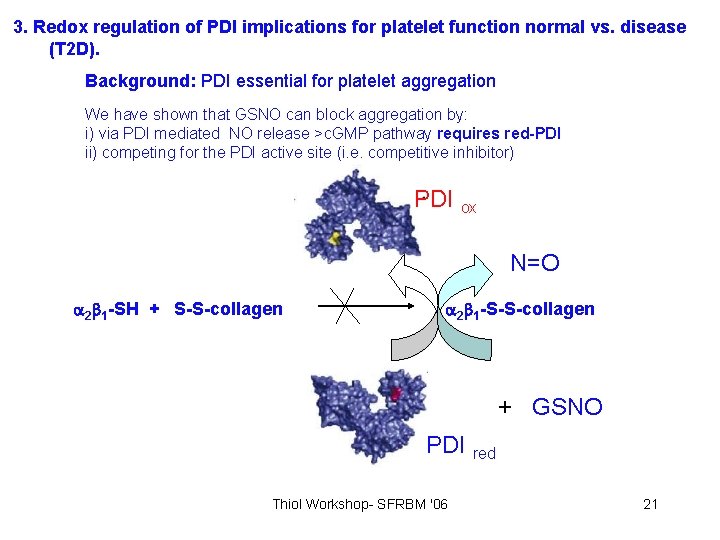
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Background: PDI essential for platelet aggregation We have shown that GSNO can block aggregation by: i) via PDI mediated NO release >c. GMP pathway requires red-PDI ii) competing for the PDI active site (i. e. competitive inhibitor) PDI ox N=O 2 1 -SH + S-S-collagen 2 1 -S-S-collagen + GSNO PDI red Thiol Workshop- SFRBM '06 21
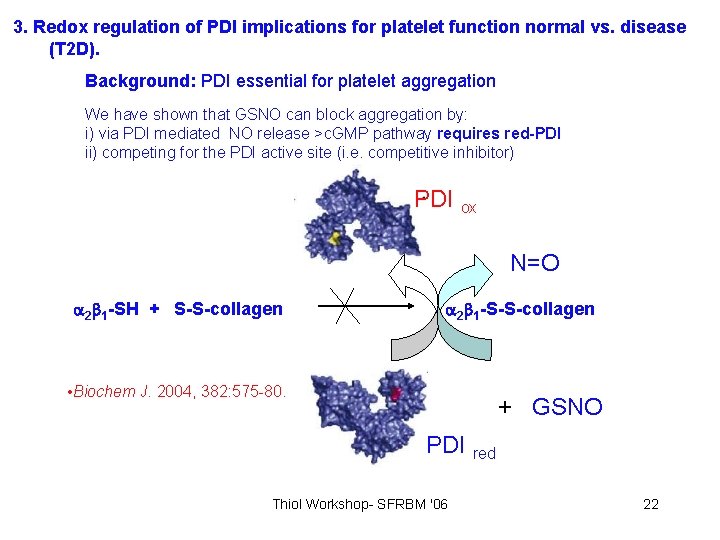
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Background: PDI essential for platelet aggregation We have shown that GSNO can block aggregation by: i) via PDI mediated NO release >c. GMP pathway requires red-PDI ii) competing for the PDI active site (i. e. competitive inhibitor) PDI ox N=O 2 1 -SH + S-S-collagen 2 1 -S-S-collagen • Biochem J. 2004, 382: 575 -80. + GSNO PDI red Thiol Workshop- SFRBM '06 22
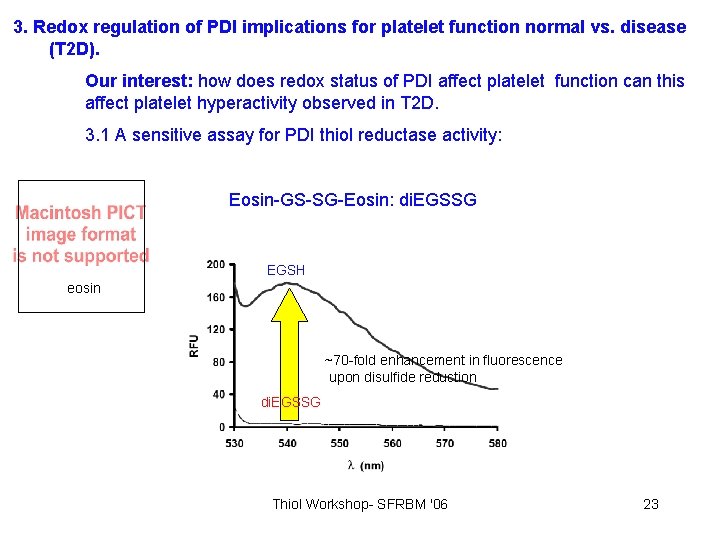
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Our interest: how does redox status of PDI affect platelet function can this affect platelet hyperactivity observed in T 2 D. 3. 1 A sensitive assay for PDI thiol reductase activity: Eosin-GS-SG-Eosin: di. EGSSG EGSH eosin ~70 -fold enhancement in fluorescence upon disulfide reduction di. EGSSG Thiol Workshop- SFRBM '06 23
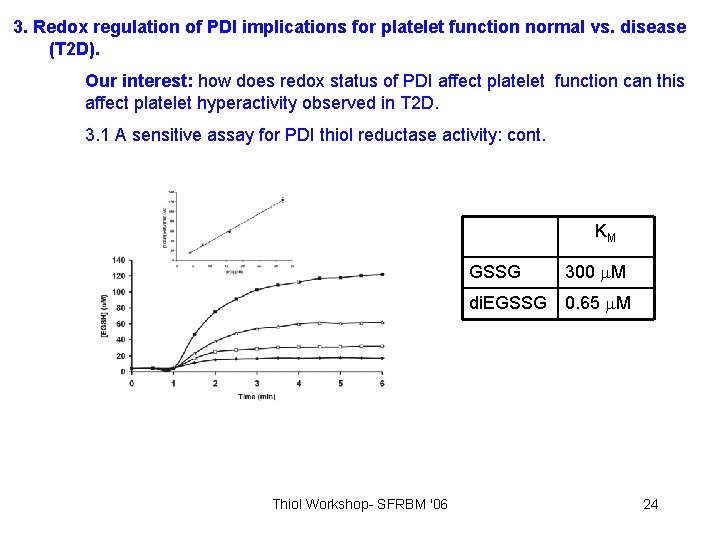
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Our interest: how does redox status of PDI affect platelet function can this affect platelet hyperactivity observed in T 2 D. 3. 1 A sensitive assay for PDI thiol reductase activity: cont. KM Thiol Workshop- SFRBM '06 GSSG 300 M di. EGSSG 0. 65 M 24
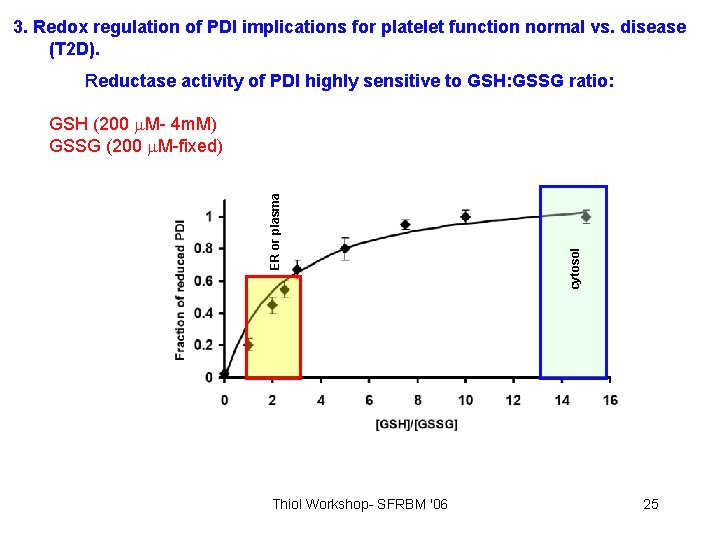
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). Reductase activity of PDI highly sensitive to GSH: GSSG ratio: Thiol Workshop- SFRBM '06 cytosol ER or plasma GSH (200 M- 4 m. M) GSSG (200 M-fixed) 25

3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). compare platelets from control and T 2 D human subjects (n=22) for: platelet- initial rates of aggregation: PDI- denitrosation activity PDI- disulfide reductase activity Thiol Workshop- SFRBM '06 26
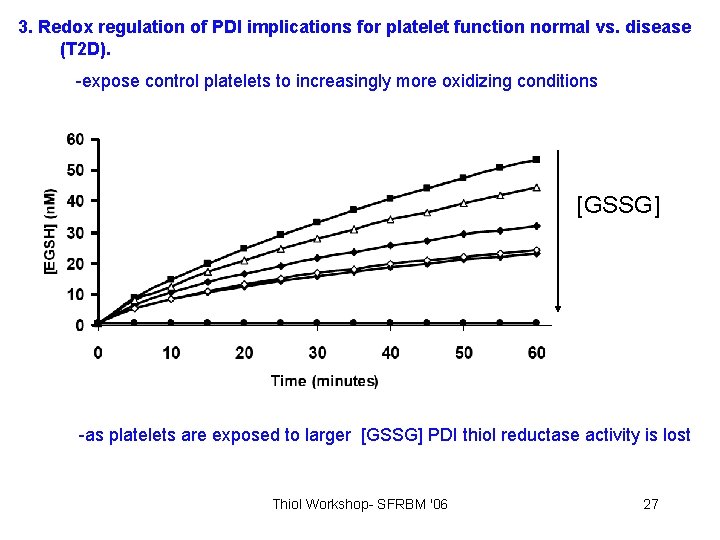
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). -expose control platelets to increasingly more oxidizing conditions [GSSG] -as platelets are exposed to larger [GSSG] PDI thiol reductase activity is lost Thiol Workshop- SFRBM '06 27

3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). vo aggregation (/min) -expose control platelets to increasingly more oxidizing conditions -as platelets are exposed to larger [GSSG] they aggregate faster Thiol Workshop- SFRBM '06 28
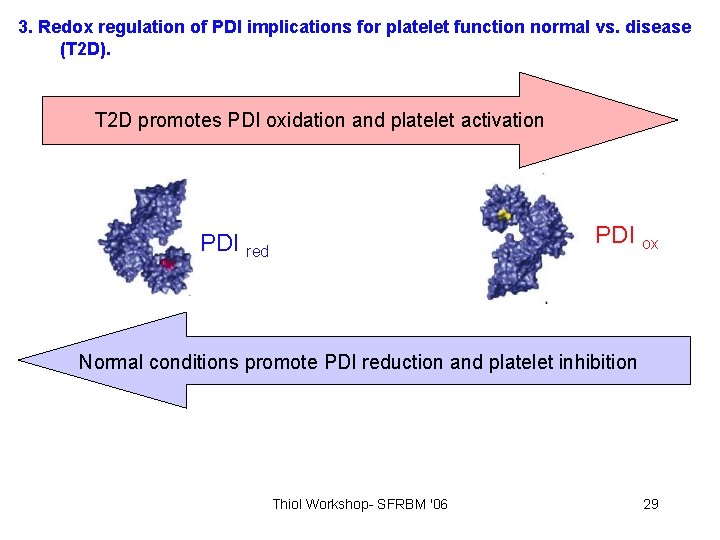
3. Redox regulation of PDI implications for platelet function normal vs. disease (T 2 D). T 2 D promotes PDI oxidation and platelet activation PDI ox PDI red Normal conditions promote PDI reduction and platelet inhibition Thiol Workshop- SFRBM '06 29

Thiol Workshop- SFRBM '06 30

Acknowledgements: Dr. Niro Ramachandran Arun Raturi Inga Sliskovic Shane Miersch Ruchi Chaube Harman Kaur Khaled Elmosrati Funding: Canadian Institutes of Health Research Canadian Diabetes Association Natural Sciences and Engineering Research Council U. Windsor Research Chair Funds Thiol Workshop- SFRBM '06 31
- Slides: 31