Protease Activated Receptors PARs Molecular Cell Biology 2013
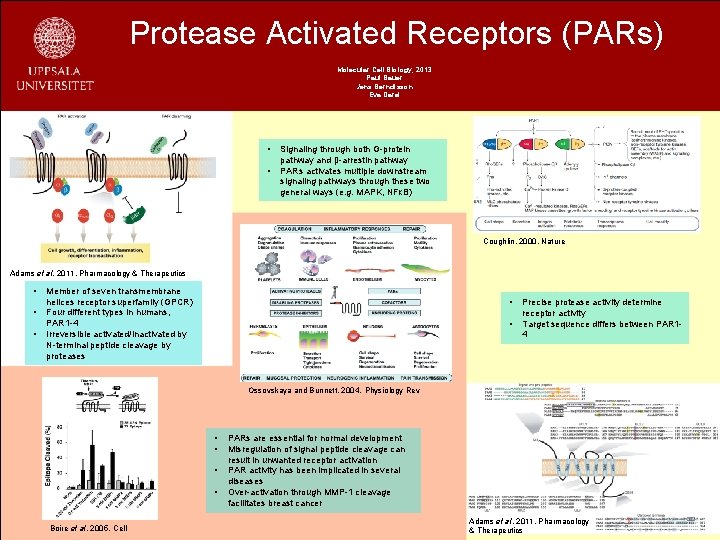
Protease Activated Receptors (PARs) Molecular Cell Biology, 2013 Paul Bauer Jens Berndtsson Eva Darai • • Signaling through both G-protein pathway and β-arrestin pathway PARs activates multiple downstream signaling pathways through these two general ways (e. g. MAPK, NFκB) Coughlin. 2000, Nature Adams et al. 2011, Pharmacology & Therapeutics • • • Member of seven transmembrane helices receptor superfamily (GPCR) Four different types in humans, PAR 1 -4 Irreversible activated/inactivated by N-terminal peptide cleavage by proteases • • Precise protease activity determine receptor activity Target sequence differs between PAR 14 Ossovskaya and Bunnett. 2004, Physiology Rev • • Boire et al. 2005, Cell PARs are essential for normal development Misregulation of signal peptide cleavage can result in unwanted receptor activation PAR activity has been implicated in several diseases Over-activation through MMP-1 cleavage facilitates breast cancer Adams et al. 2011, Pharmacology & Therapeutics

GPCR and PAR 101 Wide range of proteases cleave PARs Bockaert and Pin. 1999, EMBO GPRCRs of the largest families of proteins in the mammalian genome Mediate extracellular messages by interacting with G proteins Large variety of ligands Adams et al. 2011, Pharmacology & Therapeutics • PAR 1: Potential cleavage site for thrombin • PAR 2: 30% amino acid identity to PAR 1. Putative trypsin cleavage site • PAR 3: 28% sequence homology to human PAR 1 and PAR 2. Thrombin cleavage site. • PAR 4: . 33% homologous to the other human PARs, Cleavage site for thrombin and trypsin PARs - N-terminus cleaved by soluble proteases - New N-terminus exposed - Tethered ligand domain, binds intramolecularly to induce intracellular signal transduction - activates the cleaved receptor OR - generate a disabled receptor

Receptor structure and downstream signaling Adams et al. 2011, Pharmacology & Therapeutics N-terminus • Signal- and pro-peptideregions • Tethered ligand • Hirudin-like domains (PAR 1/3) • N-glycosylation sites Transmembrane region • Three extracellular loops (ECL) • Three intracellular loops (ICL) • Cystein for disulfide-link (ECL 2 TM 3) • PTM sites • N-glycosylation sites β-arrestin pathway • PAR 2 most completely characterized. • Phosphorylated cterminus of PAR binds to the scaffold protein β-arrestin. • Mediate downstream signaling by also binding other peptide, permitting complex formations. G-protein pathway • PAR 1/2: Gαq, Gαi, Gα 12/13, Gβγ, NFκB (inflammatory response, mostly PAR 2) • PAR 3: unclear, serves as co-factor for PAR 4 but also seems to have own signaling pathways activating ERK among others. • PAR 4: Gαq, Gαi/o C-terminus • Putative palmitylation sites • Tyrosin based motif for regulation of receptor trafficking • PTM sites Coughlin. 2000, Nature

Implications for cancer Ossovskaya and Bunnett. 2004, Physiology Rev • • PAR receptors have been implicateted in a number of diseases Function of PAR is important for normal development Mis-regulation has been linked to several forms of cancer Activation of PAR 1 and PAR 2 most studied Boire et al. 2005, Cell • • Study on the different activation by Thrombin and MMP 1 Epitope cleavage is specific for both proteases Other proteases don't cleave specific Clevage causes downstream activation through Ca²⁺ signaling

Questions and feedback
- Slides: 5