Prostate Cancer Epidemiology Estimated 2010 Total Active Disease












- Slides: 29

Prostate Cancer Epidemiology: Estimated 2010 Total Active Disease: 773, 000 US Census, SEER 2007, NODBIMPAC Stages I-III: 508, 000 Metastatic (M+): 183, 000 Castrate Sensitive*: 80, 000 Castrate Resistant*: 103, 000 Market Research Symptomatic M+ CRPC Asymptomatic M+ CRPC *Estimated based on physician reported percentages among 200 community oncologists and urologists. (2005).

Life After Traditional Androgen Deprivation Failure • Other Hormonal Therapy • Chemo Therapy • Immunotherapy

Rationale For Immunotherapy • Cancer • Cellular dysfunction • Resistance to or escape from host defenses • Active immunotherapy • • • Suitable target antigen Effective presentation of antigen Mobilization of immune cascade

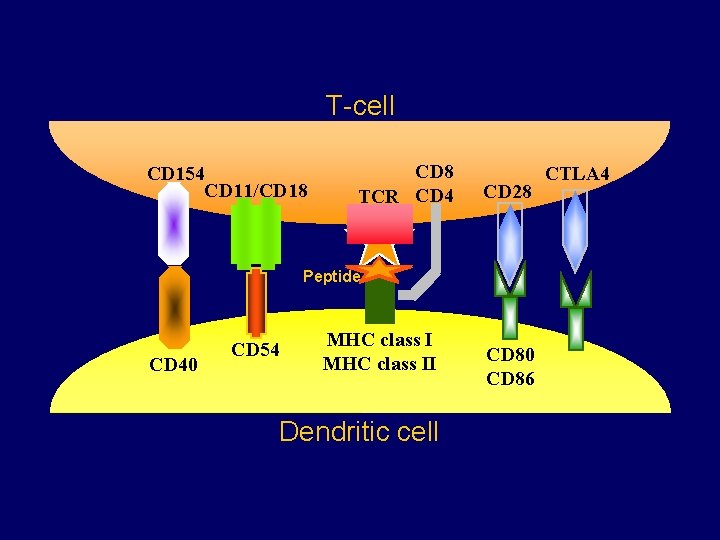
Antigen Presenting Cells (APCs) § Dendritic cells (DCs) are antigen-presenting cells that process and present MHC-peptide complexes to activate a T-lymphocyte response

T-cell CD 154 CD 11/CD 18 CD 8 TCR CD 4 CD 28 Peptide CD 40 CD 54 MHC class II Dendritic cell CD 80 CD 86 CTLA 4

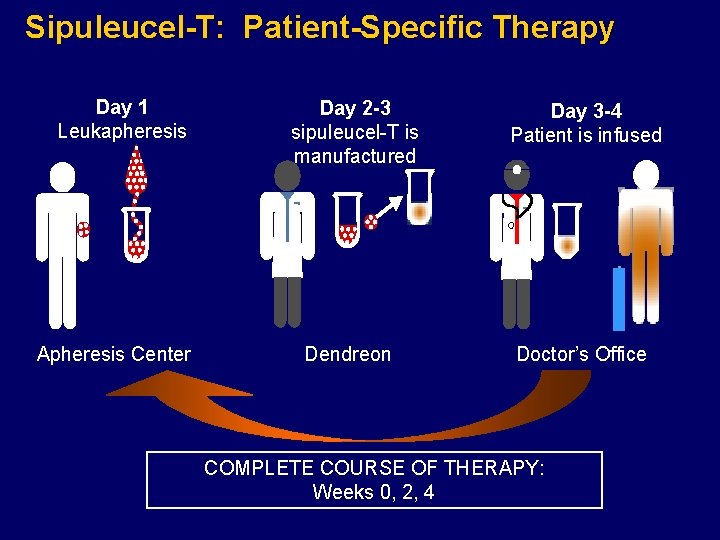
sipuleucel-T (Provenge®) sipuleucel-T is an autologous active cellular immunotherapy that activates the immune system against prostate cancer

Sipuleucel-T: Patient-Specific Therapy Day 1 Leukapheresis Apheresis Center Day 2 -3 sipuleucel-T is manufactured Dendreon Day 3 -4 Patient is infused Doctor’s Office COMPLETE COURSE OF THERAPY: Weeks 0, 2, 4

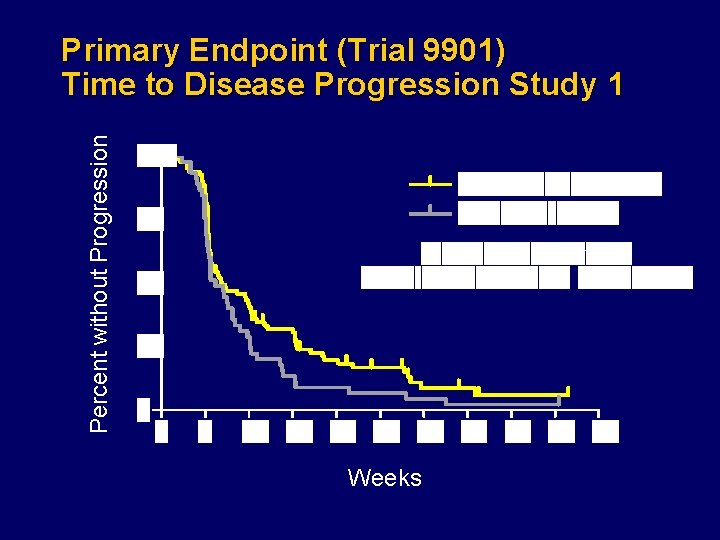
Percent without Progression Primary Endpoint (Trial 9901) Time to Disease Progression Study 1 Weeks

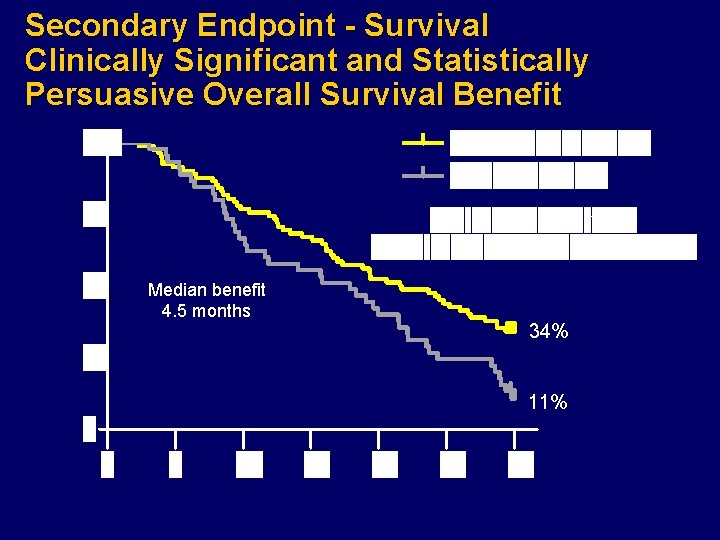
Secondary Endpoint - Survival Clinically Significant and Statistically Persuasive Overall Survival Benefit Median benefit 4. 5 months 34% 11%

Vote of FDA Expert Panel 17 – 0 for safety 13 – 4 for substantial evidence of efficacy FDA Requires More Data (<2% Event)

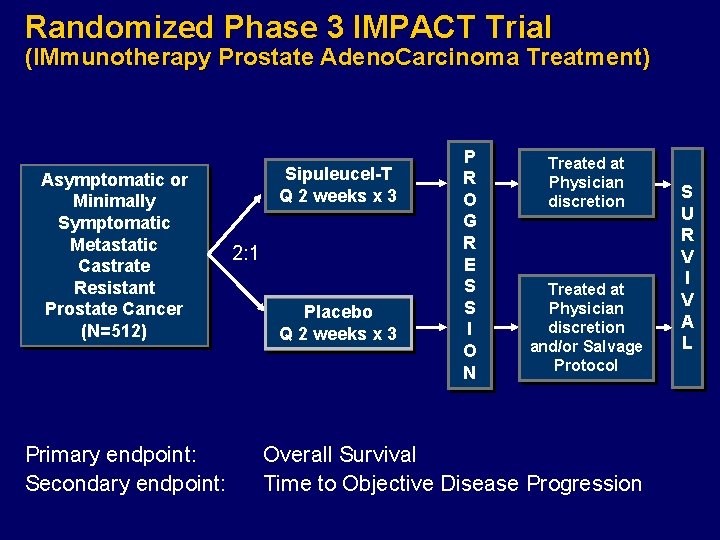
Randomized Phase 3 IMPACT Trial (IMmunotherapy Prostate Adeno. Carcinoma Treatment) Asymptomatic or Minimally Symptomatic Metastatic Castrate Resistant Prostate Cancer (N=512) Primary endpoint: Secondary endpoint: Sipuleucel-T Q 2 weeks x 3 2: 1 Placebo Q 2 weeks x 3 P R O G R E S S I O N Treated at Physician discretion and/or Salvage Protocol Overall Survival Time to Objective Disease Progression S U R V I V A L

Eligibility Criteria Metastatic castrate resistant prostate cancer Life expectancy of at least 6 months Serum PSA ≥ 5. 0 ng/m. L Castrate level of testosterone (< 50 ng/d. L) achieved via medical or surgical castration Adequate hematologic, renal, and liver function Negative serology for HIV 1 & 2, HTLV-1, and Hepatitis B & C

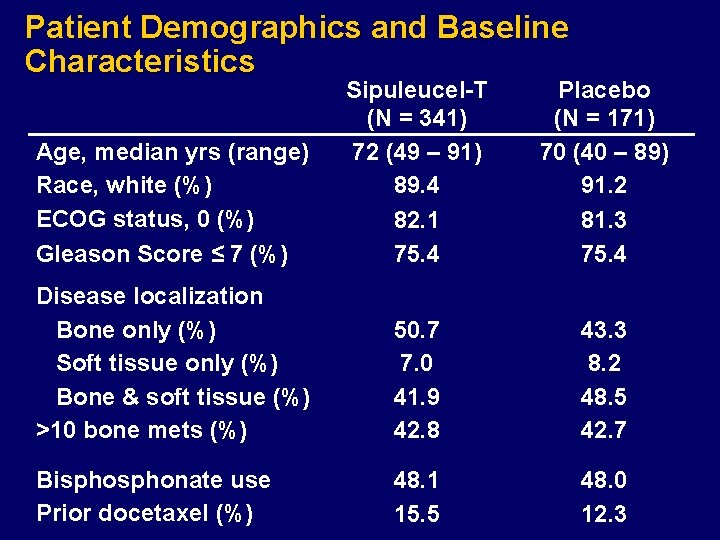
Patient Demographics and Baseline Characteristics Age, median yrs (range) Race, white (%) ECOG status, 0 (%) Gleason Score ≤ 7 (%) Sipuleucel-T (N = 341) 72 (49 – 91) 89. 4 82. 1 75. 4 Placebo (N = 171) 70 (40 – 89) 91. 2 81. 3 75. 4 Disease localization Bone only (%) Soft tissue only (%) Bone & soft tissue (%) >10 bone mets (%) 50. 7 7. 0 41. 9 42. 8 43. 3 8. 2 48. 5 42. 7 Bisphonate use Prior docetaxel (%) 48. 1 15. 5 48. 0 12. 3

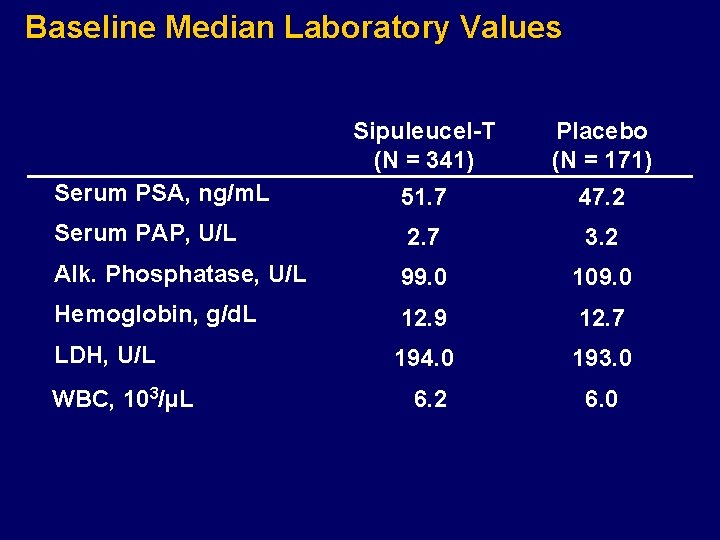
Baseline Median Laboratory Values Sipuleucel-T (N = 341) Placebo (N = 171) Serum PSA, ng/m. L 51. 7 47. 2 Serum PAP, U/L 2. 7 3. 2 Alk. Phosphatase, U/L 99. 0 109. 0 Hemoglobin, g/d. L 12. 9 12. 7 LDH, U/L 194. 0 193. 0 6. 2 6. 0 WBC, 103/µL

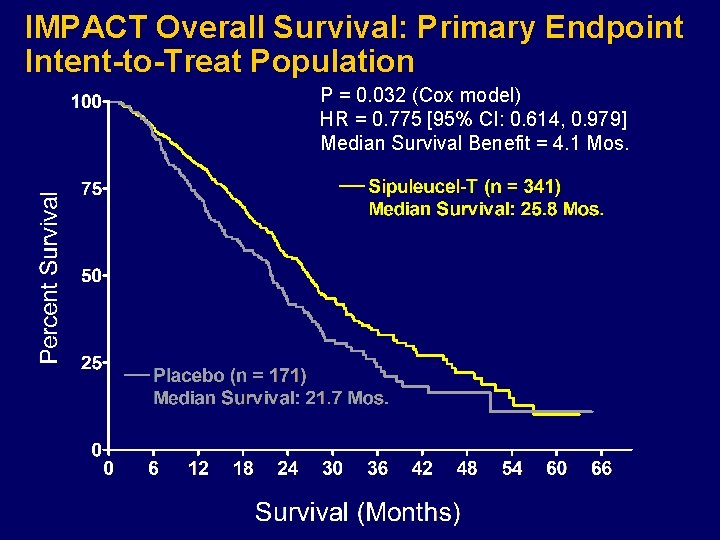
IMPACT Overall Survival: Primary Endpoint Intent-to-Treat Population P = 0. 032 (Cox model) HR = 0. 775 [95% CI: 0. 614, 0. 979] Median Survival Benefit = 4. 1 Mos.

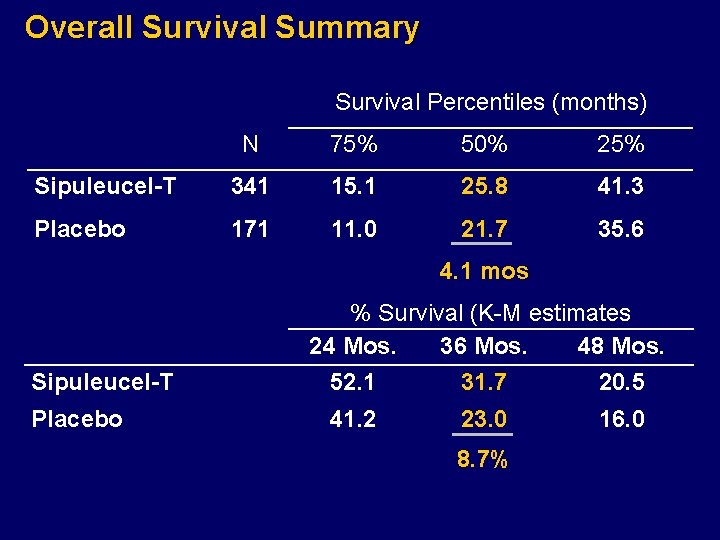
Overall Survival Summary Survival Percentiles (months) N 75% 50% 25% Sipuleucel-T 341 15. 1 25. 8 41. 3 Placebo 171 11. 0 21. 7 35. 6 4. 1 mos Sipuleucel-T Placebo % Survival (K-M estimates 24 Mos. 36 Mos. 48 Mos. 52. 1 31. 7 20. 5 41. 2 23. 0 8. 7% 16. 0

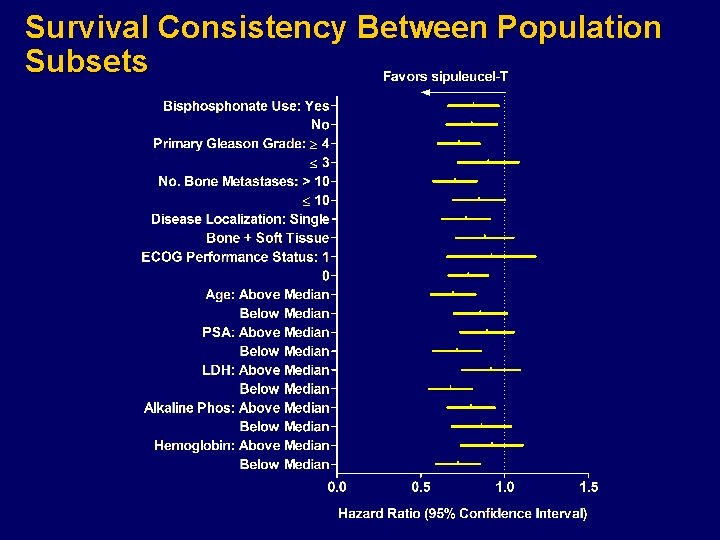
Survival Consistency Between Population Subsets

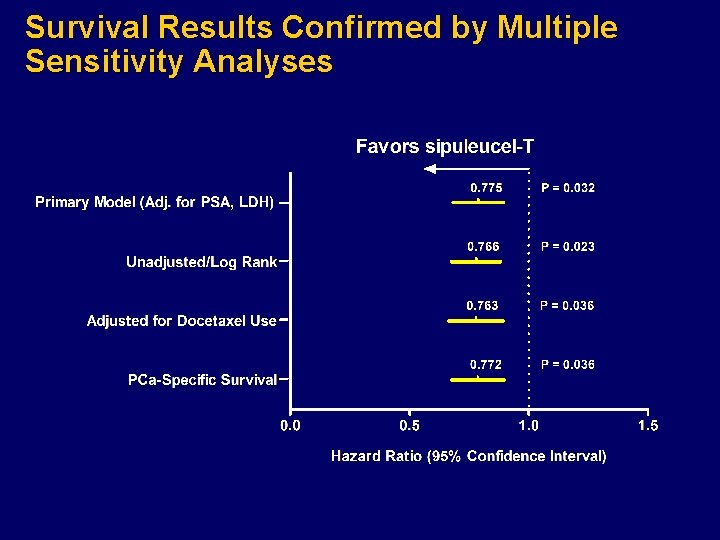
Survival Results Confirmed by Multiple Sensitivity Analyses

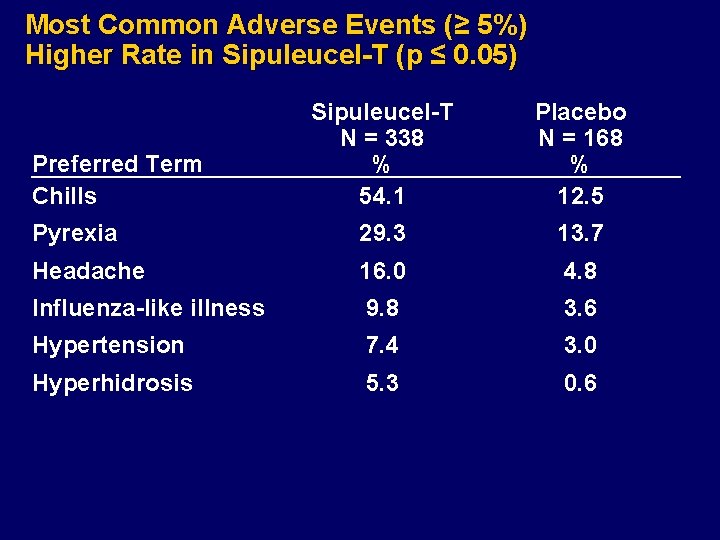
Most Common Adverse Events (≥ 5%) Higher Rate in Sipuleucel-T (p ≤ 0. 05) Sipuleucel-T N = 338 % 54. 1 Placebo N = 168 % 12. 5 Pyrexia 29. 3 13. 7 Headache 16. 0 4. 8 Influenza-like illness 9. 8 3. 6 Hypertension 7. 4 3. 0 Hyperhidrosis 5. 3 0. 6 Preferred Term Chills

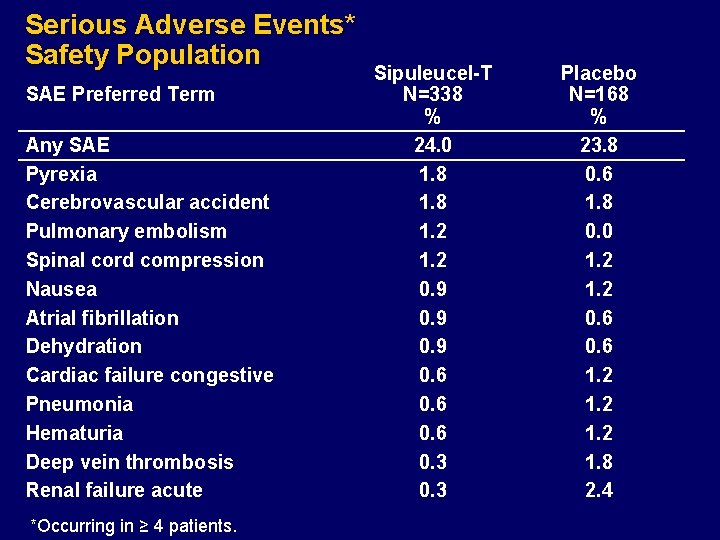
Serious Adverse Events* Safety Population SAE Preferred Term Any SAE Pyrexia Cerebrovascular accident Pulmonary embolism Spinal cord compression Nausea Atrial fibrillation Dehydration Cardiac failure congestive Pneumonia Hematuria Deep vein thrombosis Renal failure acute *Occurring in ≥ 4 patients. Sipuleucel-T N=338 % 24. 0 1. 8 1. 2 0. 9 0. 6 0. 3 Placebo N=168 % 23. 8 0. 6 1. 8 0. 0 1. 2 0. 6 1. 2 1. 8 2. 4

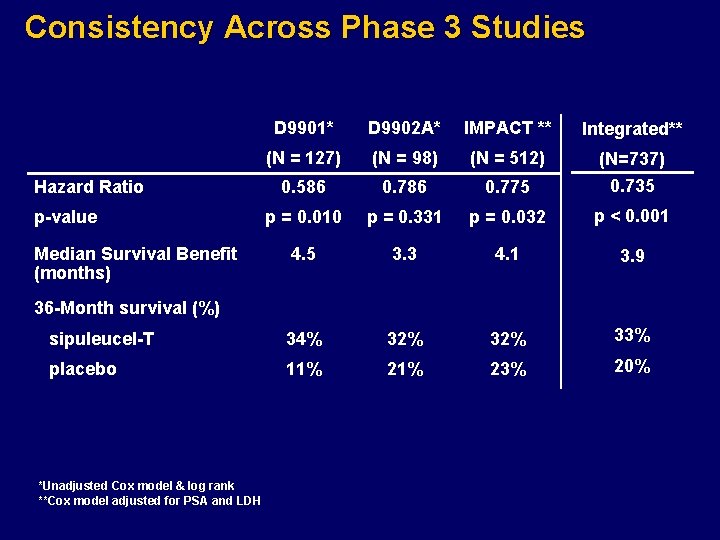
Consistency Across Phase 3 Studies D 9901* D 9902 A* IMPACT ** Integrated** (N = 127) (N = 98) (N = 512) 0. 586 0. 775 (N=737) 0. 735 p = 0. 010 p = 0. 331 p = 0. 032 p < 0. 001 4. 5 3. 3 4. 1 3. 9 sipuleucel-T 34% 32% 33% placebo 11% 23% 20% Hazard Ratio p-value Median Survival Benefit (months) 36 -Month survival (%) *Unadjusted Cox model & log rank **Cox model adjusted for PSA and LDH

Summary First active immunotherapy to demonstrate improvement in overall survival for cancer Highly favorable benefit to risk profile Short duration of therapy Potential to create new paradigm in treatment of metastatic, castrate resistant prostate cancer

Time to Objective Disease Progression Secondary endpoint Result – Independent radiologic review – HR=0. 951 (95% CI: 0. 77, 1. 17); P=0. 628 (log rank) Consistent with other trials in advanced prostate cancer Difficult endpoint to measure reliably and doesn’t correlate with overall survival

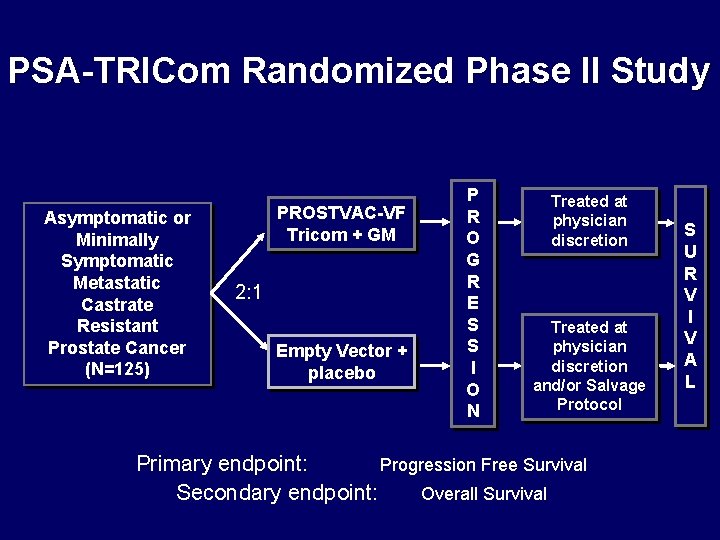
PSA-TRICom Randomized Phase II Study Asymptomatic or Minimally Symptomatic Metastatic Castrate Resistant Prostate Cancer (N=125) PROSTVAC-VF Tricom + GM 2: 1 Empty Vector + placebo P R O G R E S S I O N Treated at physician discretion and/or Salvage Protocol Primary endpoint: Progression Free Survival Secondary endpoint: Overall Survival S U R V I V A L

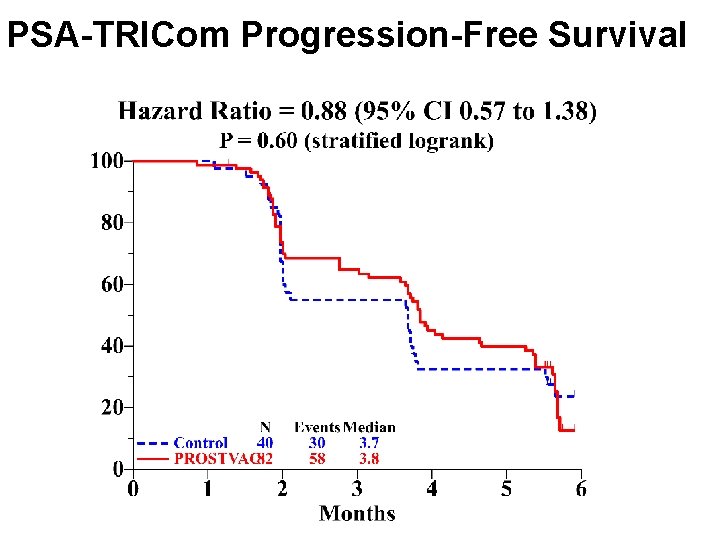
PSA-TRICom Progression-Free Survival 25

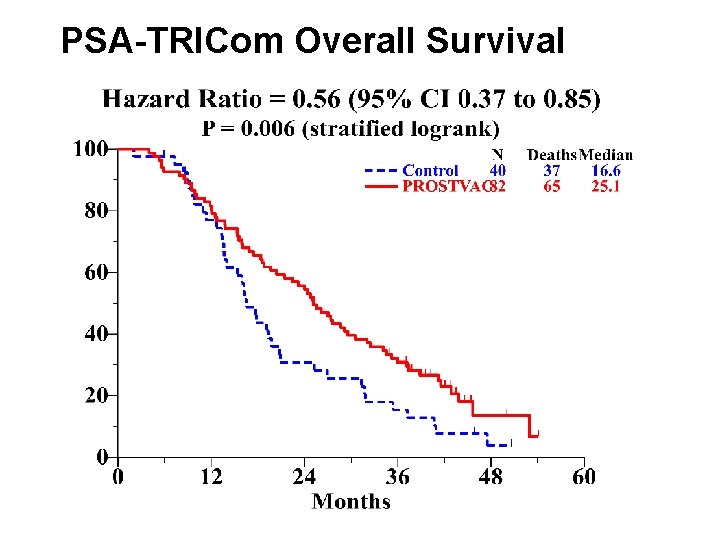
PSA-TRICom Overall Survival 26

Background-The Development of PROSTVAC-VF-Tricom Vaccinia – Potent immunological priming agent – Derived from wild-type Wyeth strain (used in millions of immunizations) Fowlpox – Minimally/non-cross-reactive with vaccinia q Enables boosting Slightly altered PSA transgene Tricom – Lymphocyte function-associated antigen LFA-3 (CD 58) – Intercellular adhesion molecule ICAM-1 (CD 54) – Costimulatory molecule for the T-cell receptor B 7. 1 (CD 80) 27

PROSTVAC-PSA-Tricom Vaccinia-PSA-Tricom Fowlpox-PSA-Tricom Prime Boosts 28

Conclusions Poxviral-based immunotherapy is feasible and safe Primary endpoint of prolonged PFS was not met Secondary endpoint of prolonged median OS – 8. 5 month difference in OS, HR 0. 56 (95% CI 0. 37 -0. 85) with p=0. 0061 Baseline characteristics of patients in both arms similar Need to validate these findings in large Phase III study with survival endpoint