Proposal To Schedule Fioricet Frederick Nichols Hope Barnes

Proposal To Schedule ® Fioricet Frederick Nichols Hope Barnes Todd Nitta Marketa Lanier Radee Bun Han Nguyen Mike Sylva

Outline • Introduction • Stakeholders & Alternatives • Technical & Political Feasibility, Viability • Evidence of Schedule III status • Recommended Change to the Law

Introduction • Fioricet® Unscheduled Ø Acetaminophen 325 mg Ø Butalbital 50 mg Ø Caffeine 40 mg • Fiorinal® Scheduled Ø Aspirin 325 mg Ø Butalbital 50 mg Ø Caffeine 40 mg **The only difference is Aspirin & Acetaminophen

Current Problems • Since 2000/List of Abused Compounds Ø Fioricet® – 50 to 74 place Ø Scheduled Butalbital Compounds – 149 to 194 place • Pizza Ambush • Conclusion: Ø Fioricet® is more abused because it is more available

Current Law • 21 CFR Part 1308 Ø What are the criteria for scheduling Ø 1988 – Except for certain mixtures of scheduled + non-narcotic substances Ø Fioricet® Included

Drug Abuse and Dependence (for both Fioricet® and Fiorinal®) Abuse and Dependence Butalbital Barbiturates may be habit-forming: Tolerance, psychological dependence, and physical dependence may occur especially following prolonged use of high doses of barbiturates. The average daily dose of the barbiturate addict is usually about 1500 mg. As tolerance to barbiturates develops, the amount needed to maintain the same level of intoxication increases: tolerance to a fatal dosage, however, does not increase more than two-fold. As this occurs, the margin between an intoxication dose and a fatal dosage becomes smaller. The lethal dose of a barbiturate is far less if alcohol is ingested. Major withdrawal symptoms (convulsions and delirium) may occur within 16 hours and last up to 5 days after abrupt cessation of these drugs.

Overdosage (Fioricet®) Following an acute overdose of butalbital, acetaminophen, and caffeine, toxicity may result from the barbiturate or the acetaminophen. Signs and Symptoms Toxicity from barbiturate poisoning includes drowsiness, confusion, and coma; respiratory depression; hypotension; and hypovolemic shock. In acetaminophen overdose: dose-dependent, potentially fatal hepatic necrosis is the most serious adverse effect. In adults hepatic toxicity has rarely been reported with acute overdoses of less than 10 grams, or fatalities with less than 15 grams. Toxic Doses (for adults) Butalbital Toxic dose 1 g (20 tablets Fioricet ®) Acetaminophen Toxic dose 10 g (30 tablets Fioricet ®) Caffeine Toxic dose 1 g (25 tablets Fioricet®)

How patients become addicted to Fioricet® • Prescribers/patients may be unaware that this drug is potentially habit forming because it is not listed as a controlled substance. • This may lead to unmanaged use of this drug for the following reasons:

How patients become addicted to Fioricet • Patients who experience tension headache and migraines are at risk of taking excessive amounts of medication in hope to facilitate immediate relief from their symptoms. • This habit can often lead to the development of rebound/interdose headaches and over a period of time these headaches may become chronic daily headaches. • Tolerance can develop in patients, therefore they need to consume more drug to experience relief from their headache.

How patients become addicted to Fioricet • Case reports of patients who suffered from severe migraines developed a chemical dependence to butalbital containing products used for legitimate treatment purposes. • Withdrawal symptoms experiences by these patients were grand real seizures and associated behavioral disorders. • These patients also developed drug seeking behaviors to overcome their prescription limit.
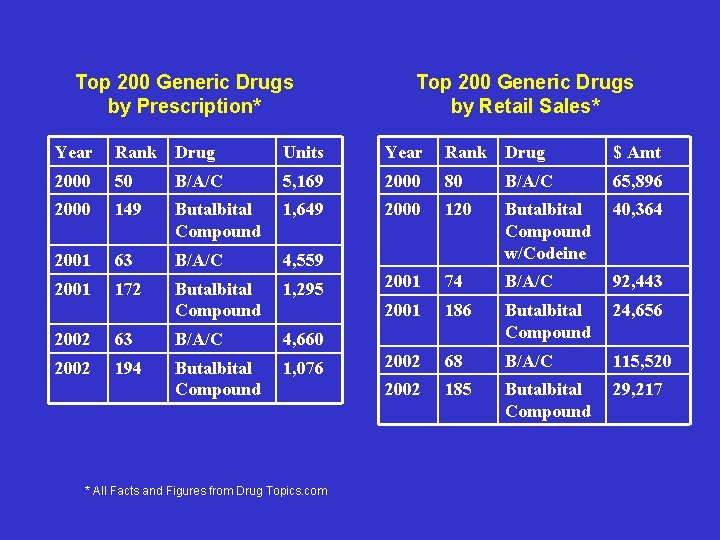
Top 200 Generic Drugs by Prescription* Top 200 Generic Drugs by Retail Sales* Year Rank Drug Units Year Rank Drug $ Amt 2000 50 B/A/C 5, 169 2000 80 B/A/C 65, 896 2000 149 Butalbital Compound 1, 649 2000 120 40, 364 2001 63 B/A/C 4, 559 Butalbital Compound w/Codeine 2001 172 Butalbital Compound 1, 295 2001 74 B/A/C 92, 443 2001 186 Butalbital Compound 24, 656 2002 68 B/A/C 115, 520 2002 185 Butalbital Compound 29, 217 2002 63 B/A/C 4, 660 2002 194 Butalbital Compound 1, 076 * All Facts and Figures from Drug Topics. com

Other stake holders include: Pharmacists – divided • scheduled in the best interests of the patient to prevent abuse • double-counting, responding to more frequent refill requests, and having to inventory another controlled substance Physicians – divided Patients – depend on their affiliation with the drug Wholesalers – in favor to divert sales to comparable and more expensive drugs such as sumatriptan (Imitrex®) Overall, we believe that the stakeholders in favor of scheduling have more valid opinions and sincere intentions regarding the outcomes of scheduling.

Alternatives • Change Fioricet® to a C-IV or C-V, which would at least give it some recognition as a substance with potential for abuse • Propose for Fioricet ® to be Scheduled as CIII at the Federal level • Pull Fioricet® from the market

Alternatives • Our least favored alternative: Do Nothing

Controlled Substance Scheduling: Exemption Criteria from Federal Register: • 15 mg intermediate-acting barbiturate: + 70 mg APAP or + 188 mg ASA = Exempt • for 50 mg Butalbital, drug must contain at least: 233 mg APAP or 627 mg ASA = Exempt • Fioricet® = 325 mg APAP = Exempt • Fiorinal® = 325 mg ASA = C-III

Technical Feasibility • Determine whether scheduling Fioricet will/will not lead to reduction in abuse • Collect data from: – Rehab Programs – Medical Establishments – WA State Law Enforcement Agencies • Compare yearly data and identify actual effects – DAWN Reports
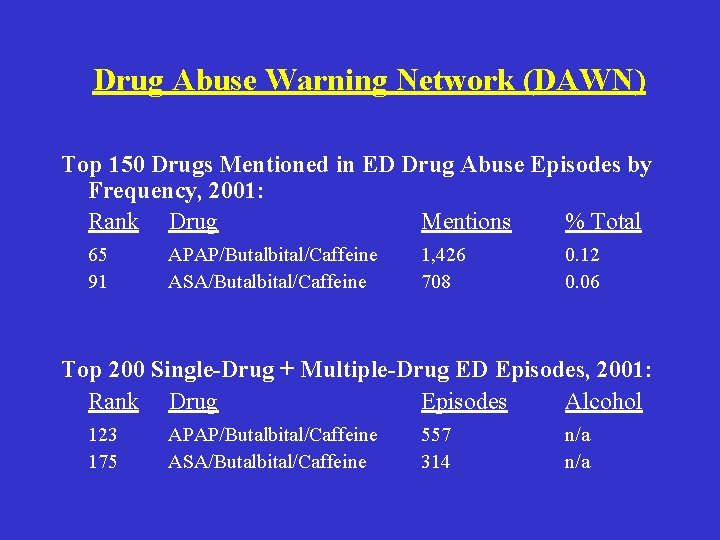
Drug Abuse Warning Network (DAWN) Top 150 Drugs Mentioned in ED Drug Abuse Episodes by Frequency, 2001: Rank Drug Mentions % Total 65 91 APAP/Butalbital/Caffeine ASA/Butalbital/Caffeine 1, 426 708 0. 12 0. 06 Top 200 Single-Drug + Multiple-Drug ED Episodes, 2001: Rank Drug Episodes Alcohol 123 175 APAP/Butalbital/Caffeine ASA/Butalbital/Caffeine 557 314 n/a

Political Viability • Determine if proposal will survive “Political Test” • Involves decision makers, officials, voters • Perspectives: – Supporters – Non-Supporters • Manufacturers • Patients

Administrative Operability • Determine if there is an existing administrative system to put our proposal into effect • RCW 69. 50. 201 – BOP has authority and responsibility • To schedule, drug must meet 8 criteria – These relate to: • • Potential for drug abuse Patterns of abuse Risk to public health Psychic or physiological dependence, etc… • Consideration of FDA and DEA findings

Micki Kedzierski, substance abuse expert · Fioricet Migraine Headache Frequent dosing · No maintenance therapy · Rehab is difficult · Cost · So should Fioricet it be scheduled?

Recommendation of Changes to WAC 246 -887 -160: Schedule III The board finds that the following substances have potential for abuse less than substances listed in Schedules I and II, and have currently accepted medical use in treatment in the United States and that the abuse of the substances may lead to moderate or low physical dependency to high physiological dependency. The board, therefore, places each of the following substances In Schedule III.

Recommendation of Changes to WAC 246 -887 -160: Schedule III (a) (No changes) (b) (No changes)

Recommendation of Changes to WAC 246887 -160: Schedule III (c) Depressants. Unless specifically excepted or unless listed in another schedule, any material, compound, or preparation which contains any quantity of the following substances having a depressant effect on the central nervous system: (1) Any compound, mixture, or preparation containing: (i) Amobarbital; (ii) Butalbital; (iii) (iv) Secobarbital; Phenobarbital; (No changes after this)

Conclusion • Based on the evidence presented, we believe that Fioricet® should be placed in Schedule III so that it may be more tightly controlled. • To facilitate this change, the Board of Pharmacy should use its vested power under the Administrative Procedures Act and place Fioricet® on the list of drugs under WAC 246 -887 -160.
![References [1] Chichowski, John. Addiction to painkiller blamed for pizza delivery ambush. North Jersey References [1] Chichowski, John. Addiction to painkiller blamed for pizza delivery ambush. North Jersey](http://slidetodoc.com/presentation_image_h2/6afe4ea7f3897c7e7939482b867b159c/image-25.jpg)
References [1] Chichowski, John. Addiction to painkiller blamed for pizza delivery ambush. North Jersey Media Group. 2002; News; p A 03. [2] Physicians’ Desk Reference, 48 th Ed. Montvale, NJ. © 1996: 2050 -2052 [3] The Online Newsmagazine for Pharmacists. Copyright © 2003 Advanstar Medical Economics at Montvale, NJ: http: //www. drugtopics. com. [4] Young WB, Siow HC. Should butalbital-containing analgesics be banned? Yes. Curr Pain Headache Rep. 2002; 6(2): 151 -5. [5] Drug Abuse Warning Network (DAWN). US Dept. of Health and Human Services. Rockville, MD: http: //www. dawninfo. net [6] Revised Code of Washington 69. 50. 201, Uniform Controlled Substance Act, Article II: Standards and Schedules; 2002. [7] Revised Code of Washington 34. 05, Administrative Procedures Act; 2002. [8] Revised Code of Washington 69. 50. 201, Uniform Controlled Substance Act, Article II: Standards and Schedules; 2002. [9] Physicians’ Desk Reference, 48 th Ed. Montvale, NJ. © 1996: 2050 -2052. [10] Raja M, Altavista M, Azzoni A, Albanese A. Sever barbiturate withdrawal syndrome in migrainous patients. Headache. 1996; 36: 119 -121 [11] Revised Code of Washington 69. 50. 201, Uniform Controlled Substance Act, Article II: Standards and Schedules; 2002.
- Slides: 25