Properties of Water WHATS SO GREAT ABOUT WATER

Properties of Water
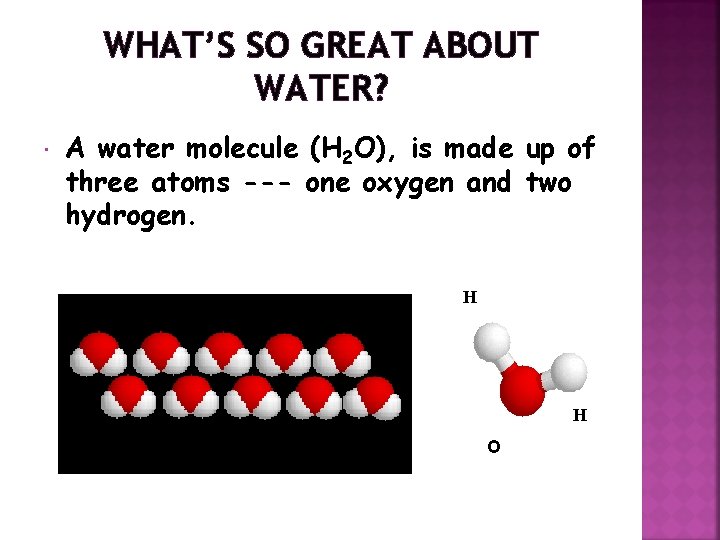
WHAT’S SO GREAT ABOUT WATER? A water molecule (H 2 O), is made up of three atoms --- one oxygen and two hydrogen. H H O


In each water molecule, the oxygen atom attracts more than its "fair share" of electrons The oxygen end “acts” negative The hydrogen end “acts” positive Causes the water to be POLAR However, Water is neutral (equal number of e- and p+) --- Zero Net Charge

HYDROGEN BONDS EXIST BETWEEN WATER MOLECULES Attraction between the hydrogen atom on one water molecule and the oxygen atom on another=Hydrogen Bond One hydrogen bond is weak , but many hydrogen bonds are strong

Single most abundant compound in most living things. Water expands as it freezes = meaning ice is less dense then liquid water. At sea level, pure water boils at 100 °C and freezes at 0 °C. The boiling temperature of water decreases at higher evaluations (lower atmospheric pressure). For this reason, an egg will take longer to boil at higher altitudes.

Cohesion Adhesion High Heat of Vaporization High Specific Heat Less Dense as a Solid

• Attraction between particles of the same substance ( why water is attracted to itself) Results in Surface tension (a measure of the strength of water’s surface) Wax Paper Activity

Adhesion (water to other) and Cohesion (water to water) Its almost like water forms a skin on the surface Pepper Activity

• • Attraction between two different substances. Water will make hydrogen bonds with other surfaces such as glass, soil, plant tissues, and cotton. Capillary action-water molecules will “tow” each other along when in a thin glass tube. Example: transpiration process which plants and trees remove water from the soil Examples: Microscope Slide/Penny Activity/Celery

ADHESION CAUSES CAPILLARY ACTION Which gives water the ability to “climb” structures

ADHESION ALSO CAUSES WATER TO … Form spheres & hold onto plant leaves Attach to a silken spider web

HIGH HEAT OF VAPORIZATION Amount of energy to convert 1 g or a substance from a liquid to a gas In order for water to evaporate, hydrogen bonds must be broken. As water evaporates, it removes a lot of heat with it.

Water vapor forms a kind of global ‘‘blanket” which helps to keep the Earth warm. Heat radiated from the sun warmed surface of the earth is absorbed and held by the vapor

HIGH SPECIFIC HEAT • Amount of heat needed to raise or lower 1 g of a substance 1° C. Water resists temperature change, both for heating and cooling. Water can absorb or release large amounts of heat energy with little change in actual temperature.

WATER IS LESS DENSE AS A SOLID • • • Ice is less dense as a solid than as a liquid (ice floats) Liquid water has hydrogen bonds that are constantly being broken and reformed. Frozen water forms a crystal-like lattice whereby molecules are set at fixed distances.

Water is Less Dense as a Solid • Which is ice and which is water?

Water is Less Dense as a Solid Water Ice
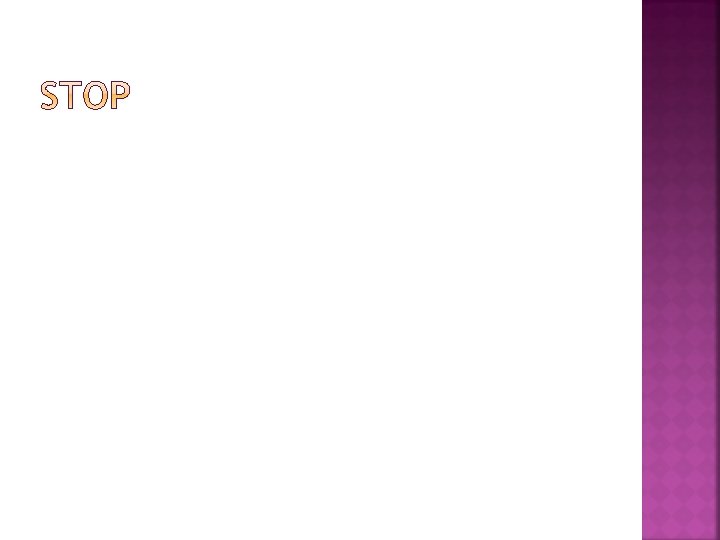

Why are carbon atoms so common in living things? One carbon atom can form 4 covalent bonds with other atoms. Covalent bonds (share electrons) enable the carbon atoms to bond to each other, which results in a variety of important organic compounds. Can be straight chained, branched, or in a ring shape that lead to the diversity of life. Figure 25

Ase-enzyme Hydrate-water Ose-full of Philic-loving Phobic-fearing

Carbo-coal Di-two Hydro-water Mono-one Poly-many

Most carbon based molecules are classified as ORGANIC Molecules In contrast-non-carbon based molecules such as water, oxygen, and ammonia are classified as INORGANIC Molecules

HYDROCARBONS-organic molecules that are composed of only carbon and hydrogen Example: Methane (CH 4)

Functional Group-A group of atoms within a molecule that interacts in predictable ways with other molecules Example: Hydroxyl groups are HYDROPHILIC (literally meaning “water-loving”) meaning they attract water molecules However, if they are not “waterloving” they are HYDROPHOBIC (literally meaning “water fearing”)

Large molecules are built from many similar, smaller molecular units called MONOMERS When monomers link together and make a long chain these are called MACROMOLECULES or POLYMERS. Example: The protein that makes up a spiders web, every living cell has thousands of different kinds of polymers

IMPORTANT: Life’s large molecules are classified into four main categories: 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids

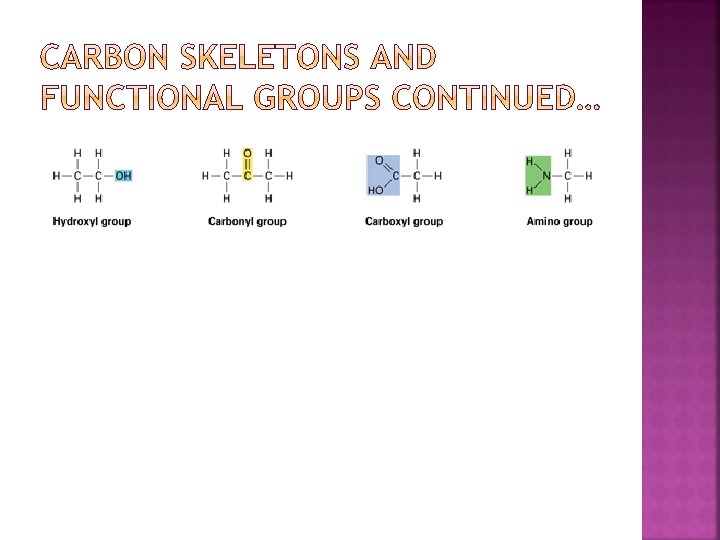
Each time a monomer is added to a chain, a water molecules is released; this is called a DEHYDRATION REACTION Cells break bonds between monomers by adding water to them, the reverse of dehydration; HYDROLYSIS REACTION
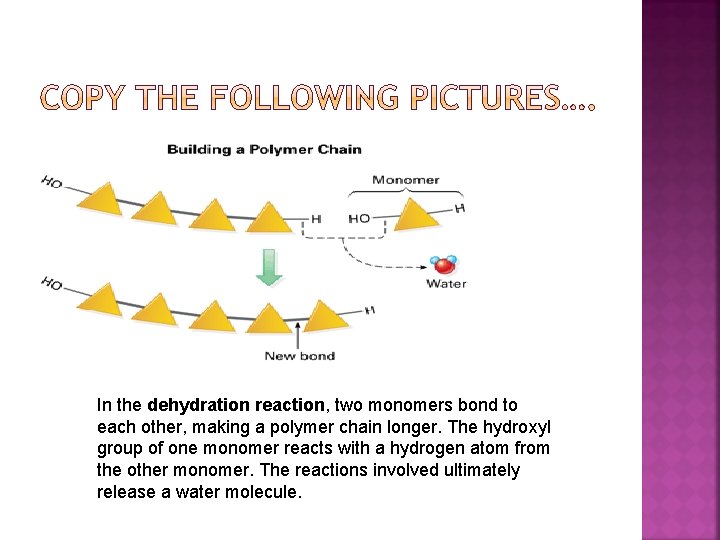
In the dehydration reaction, two monomers bond to each other, making a polymer chain longer. The hydroxyl group of one monomer reacts with a hydrogen atom from the other monomer. The reactions involved ultimately release a water molecule.
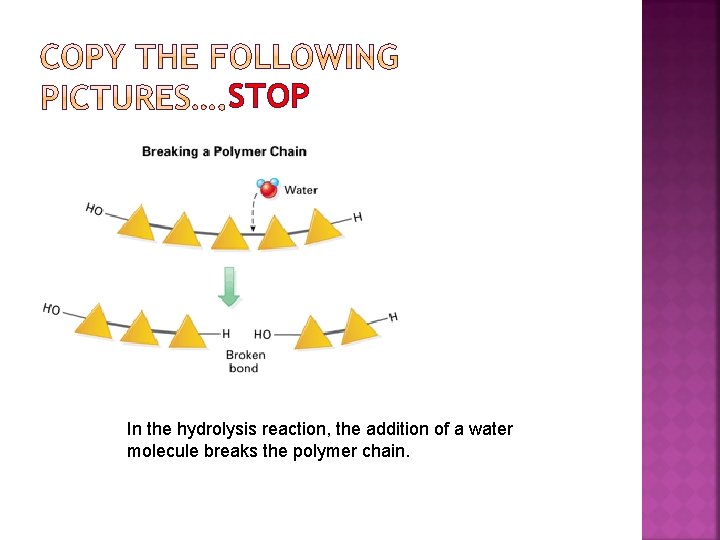
STOP In the hydrolysis reaction, the addition of a water molecule breaks the polymer chain.
1. p. H scale ranges from ___ to ____ 2. Acid range? H or OH ions? 3. Base range? H or OH ions? 4. Neutral? 5. How could you improve the lab? Regular or Extra Strength?

Line Graph…. be sure to include title, labels and a key Date Table 1…needs to be retyped or done neatly on a new sheet of paper…DO NOT just staple the paper from your lab Graph and Table DO NOT have to be typed but they need to be neat and legible 1 paragraph describing data…what were your results. . be sure to include some numbers
- Slides: 33