Properties of Water S Hydrogen Bonds S Hydrogen

Properties of Water S

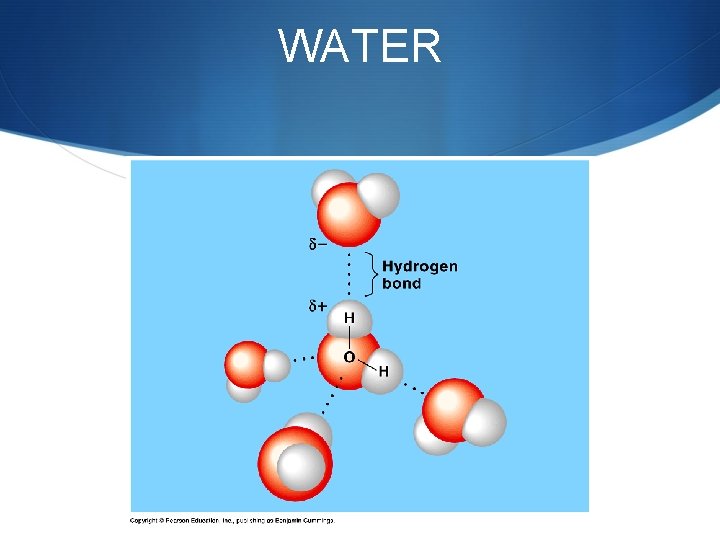
Hydrogen Bonds S Hydrogen bonds do not join atoms … S Hydrogen bonds are attractions between compounds
WATER

WATER Activity: (5 minutes) Create the structure of water, including the hydrogen bonds between molecules (drawing, model, skit)

Hydrogen Bonds Give Water Unique Properties (Part I) S Heat Storage S A large input of thermal energy is required to disrupt the organization of liquid water S This minimizes temperature changes S High Heat of Vaporization S At high temperatures, hydrogen bonds do break S Water is changed into vapor resulting in a cooling effect

Heat Storage & High Heat of Vaporization • Water’s heat storage ability keeps temp of coastal areas more stable!

Properties of Water Heat Storage & High Heat of Vaporization • These properties allow water to trap lots of heat and take it away by evaporative cooling, like when you sweat! • Ahhhhh …

Questions to Ponder (Think, Pair, Share) • Where would you feel cooler on a 100 degree day: Las Vegas or Leesburg? Why? • Why do athletes like polyester more than cotton? • Where and why was Gatorade developed? • Why does it seldom (if ever) snow in San Francisco, even though it is similar in latitude to Leesburg?

Ice Formation • At low temperatures, hydrogen bonds don’t break • Water forms a regular crystal structure that floats

Ice is less dense than water, so ice floats! • This is super important because it prevents bodies of water from freezing solid and killing all life on the surface and underneath!

Question to Ponder Think-pair-share • At what temperature is the ice cover? • Estimate the temperature of the “blue water”. Which layers are warmer? Which are colder?

Hydrogen Bonds Give Water Unique Properties (Part II) S Cohesion S Attraction of water molecules to other water molecules S Example: Surface tension S Adhesion • Attraction of water molecules to other polar molecules • Example: Capillary action Water strider

Surface Tension! S Cohesion (water to water) makes the surface of water like a “solid” film!

Surface Tension! S Demonstration

Capillary Action! S Cohesion and Adhesion Allows for Transpiration - The movement of water up a plant (by “capillary action”) because of pull from evaporation through the leaves.

Capillary Action! S demonstration

Question to Ponder Think-pair-share S Why does it hurt more to dive into the pool “spread eagle” (belly flop) than to dive in head first, and why? S Which is easier to siphon, water or diesel fuel, and why?

Water is a Great Solvent: Hydrophilic & Hydrophobic Substances S Hydrophilic (“water-loving”) substances S Polar S Hydrogen bond with water S Example: sugar S Hydrophobic (“water-fearing”) substances S Nonpolar S Repelled by water S Example: oil

Water is a Great Solvent: Hydrophilic & Hydrophobic Substances S demostration

Solution Solute + Solvent = Solution S Solute- solid particles that dissolve S Solvent- what causes dissolving S Solution- homogenous mixture made from solute and solvent S Concentrated: when there is more solute than solvent S Diluted: when there is more solvent than solute

Solution Solute + Solvent = Solution S demonstration

The p. H Scale S Measures H+ concentration of fluid S Change of 1 on scale means 10 X change in H+ concentration Highest H+ Lowest H+ 0 -----------7 ----------14 Acidic Neutral Basic S BUFFERS - Buffers are substances that resist p. H changes!

Acids & Bases S Acids S These substances cause water to release its H+ S Acidic solutions have p. H < 7 S Bases S These substances grab H+ from water, leaving behind OH- ions S Basic solutions have p. H > 7
- Slides: 23