Properties of Pure Substances Pure Substance w A

Properties of Pure Substances

Pure Substance w. A substance that has a fixed (homogeneous and invariable) chemical composition throughout is called a pure substance. w It may exist in more than one phase, but the chemical composition is the same in all phases.

Pure Substance w Pure means “…of uniform and invariable chemical composition (but more than one molecular type is allowed). ” This allows air to be a pure substance. w All our substances will be pure. We will drop the use of the word. When we refer to a simple system we mean one filled with a pure substance--a simple, pure system.

Examples of Pure Substance w Water (solid, liquid, and vapor phases) w Mixture of liquid water and water vapor w Carbon Dioxide w Nitrogen w Homogeneous mixture of gases, such as air, as long as there is no change of phases.
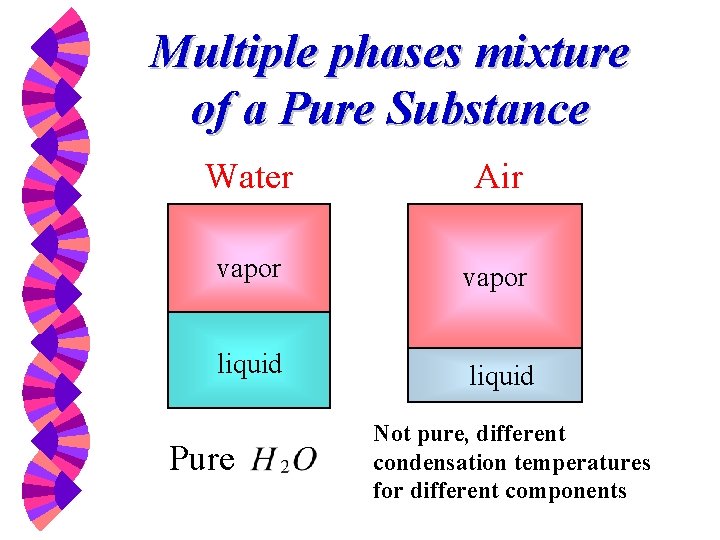
Multiple phases mixture of a Pure Substance Water Air vapor liquid Pure Not pure, different condensation temperatures for different components

Thermodynamic Properties w We have discussed extensive properties such as m, U, and V (for volume) which depend on the size or extent of a system, and w Intensive properties such as u, v, T, and P (sometimes we write a “p” for pressure, using P and p interchangeably) which are independent of system extent.

Important Questions. …. How many properties are needed to define the state of a system? w How do we obtain those properties? w Equation of State Property Tables

Review - State Postulate The number of independent intensive properties needed to characterize the state of a system is n+1 where n is the number of relevant quasiequilibrium work modes. w This is empirical, and is based on the experimental observation that there is one independent property for each way a system’s energy can be independently varied. w

Simple system w A simple system is defined as one for which only one quasiequilibrium work mode applies. Simple compressible systems Simple elastic systems w Simple magnetic systems w Simple electrostatic systems, etc. w w

Compressible w If we restrict our system to being compressible, we define what that quasiequilibrium work mode is:

For a simple system, w We may write P = P(v, T) w or v = v(P, T) perhaps T = T(P, v)

For a simple, pure substance w y 0 = y(y 1, y 2), or w P = P(v, T), v = v(P, T), and T = T(P, v) w What do these equations define, in space? w Equations used to relate properties are called “Equations of State”

Equation of State w Any two independent, intrinsic properties are sufficient to fix the intensive state of a simple substance. w One of the major task of Thermodynamics is to develop the equations of state which relate properties at a give state of a substance.

Ideal gas “law” is a simple equation of state Ru = universal gas constant = 8. 3144 (k. Pa-m 3)/(kgmol-K) = 1. 545 (ft-lbf)/(lbmol-R)

Phases of a Pure Substance Solid phase -- molecules are arranged in a 3 D pattern (lattice). w Liquid phase -- chunks of molecules float about each other, but maintain an orderly structure and relative positions within each chunk. w Gas phase -- random motion, high energy level. w
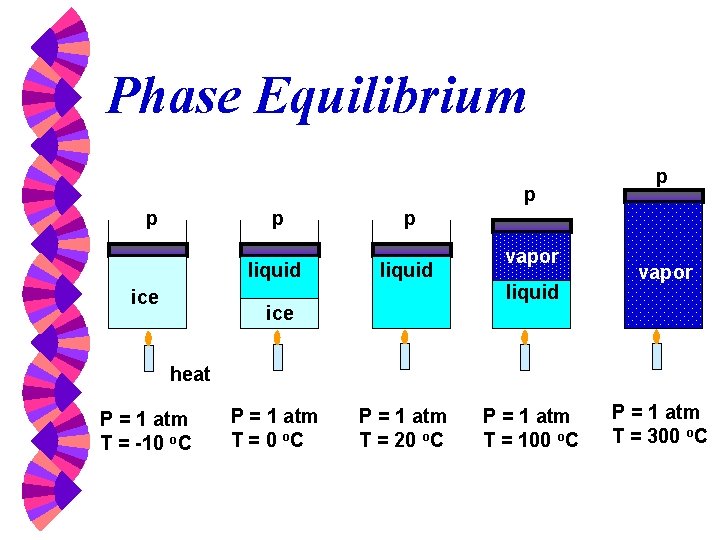
Phase Equilibrium p p p liquid ice vapor liquid vapor heat P = 1 atm T = -10 o. C P = 1 atm T = 20 o. C P = 1 atm T = 100 o. C P = 1 atm T = 300 o. C

Phase-change Process w w w Compressed liquid -- not about to evaporate Saturated liquid -- about to evaporate Saturated liquid-vapor mixture --two phase Saturated Vapor -- about to condense Superheated Vapor -- not about to condense
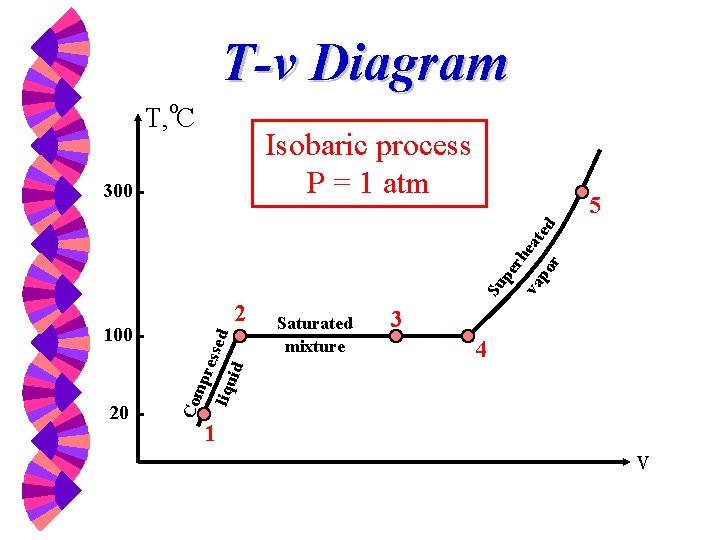
T-v Diagram T, o. C Isobaric process P = 1 atm 5 Su pe rh va eat po ed r 300 3 4 id ssed Saturated mixture liqu 20 Com pre 100 2 1 v
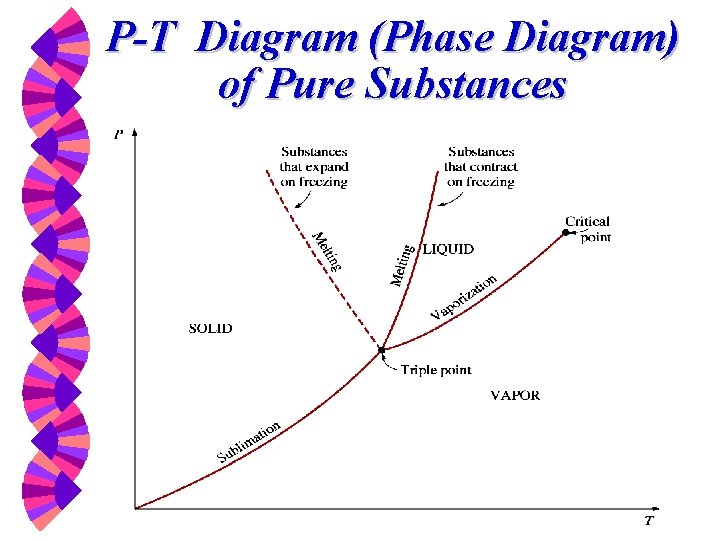
P-T Diagram (Phase Diagram) of Pure Substances
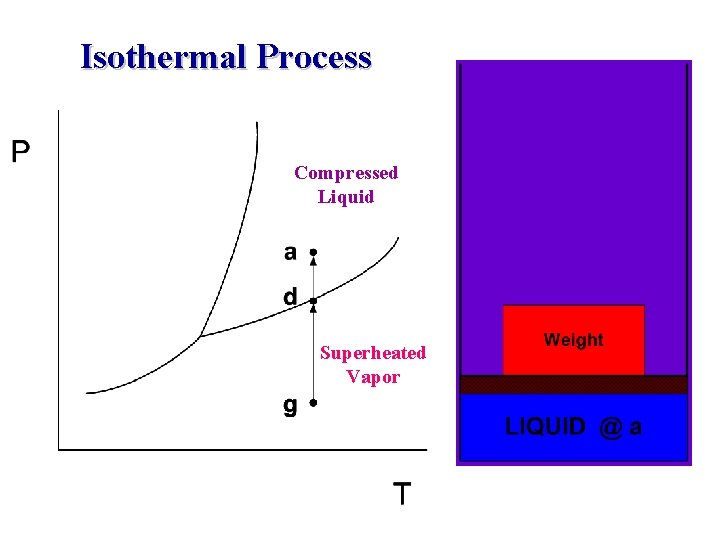
Isothermal Process Compressed Liquid Superheated Vapor
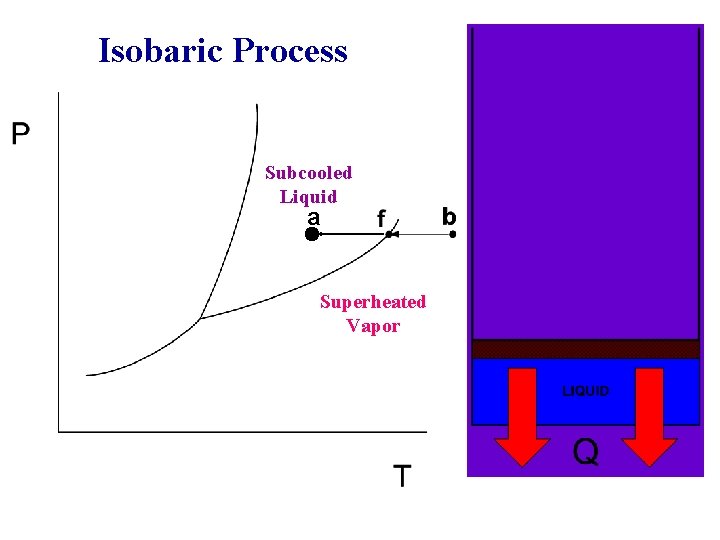
Isobaric Process Subcooled Liquid a Superheated Vapor

Water Expands on Freezing! Ice floats on top of the water body (lakes, rivers, oceans, soft drinks, etc. ). w If ice sinks to the bottom (contracts on freezing), the sun’s ray may never reach the bottom ice layers. w This will seriously disrupt marine life. w

Saturation Temperature and Pressure w Tsat -- Temperature at which a phase change takes place at a given pressure. w Psat -- Pressure at which a phase change takes place at a given temperature.

Saturation Temperature Tsat = f (Psat) p = 1 atm = 101. 3 k. Pa, p = 500 k. Pa, o T = 100 C o T = 151. 9 C *T and P are dependent during phase change *Allow us to control boiling temperature by controlling the pressure (i. e. , pressure cooker).

Latent Heat w Latent heat is the amount of energy absorbed or released during phase change Latent heat of fusion -- melting/freezing =333. 7 k. J/kg for 1 atm H 2 O w Latent heat of vaporization -boiling/condensation =2257. 1 k. J/kg for 1 atm H 2 O w
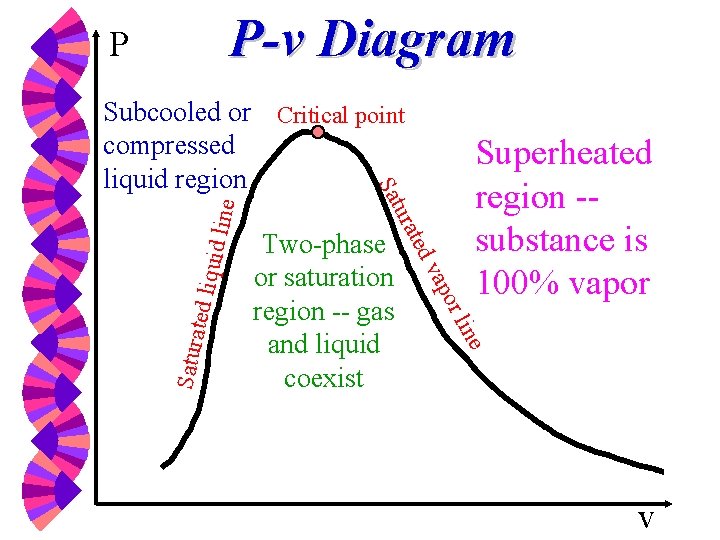
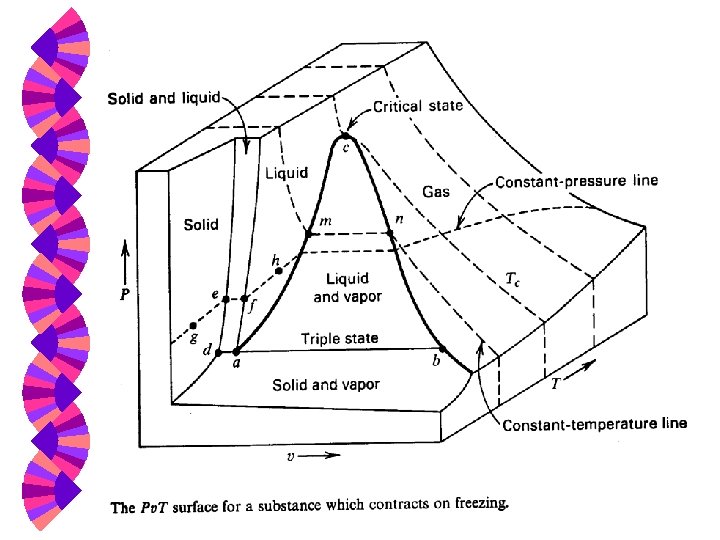
P-v Diagram P e uid lin Superheated region -substance is 100% vapor e lin ted liq or Satura vap Two-phase or saturation region -- gas and liquid coexist ted ura Sat Subcooled or Critical point compressed liquid region v
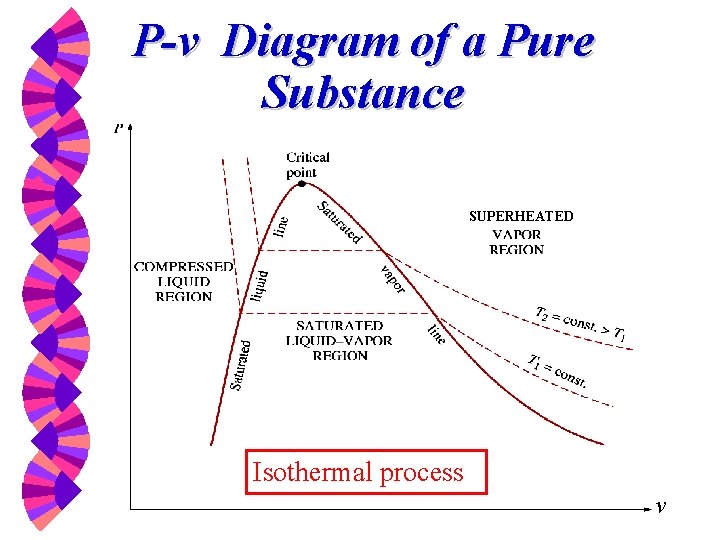
P-v Diagram of a Pure Substance w SUPERHEATED Isothermal process v
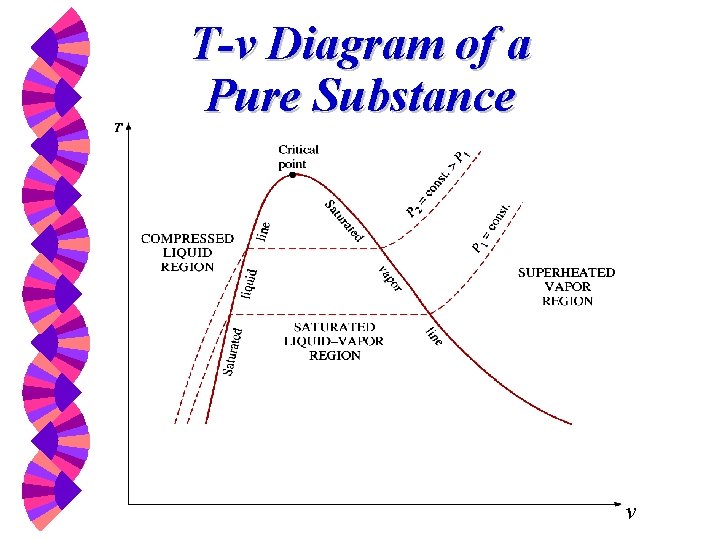
T-v Diagram of a Pure Substance v

Critical & Supercritical w The state beyond which there is no distinct vaporization process is called the critical point. w At supercritical pressures, a substance gradually and uniformly expands from the liquid to vapor phase. w Above the critical point, the phase transition from liquid to vapor is no longer discrete.

Critical Point at which the saturated vapor and saturated liquid lines coincide. w If T Tc or P Pc there is no clear distinction between the superheated vapor region and the compressed liquid region. w

Critical Point A point beyond which T Tc and a liquid-vapor transition is no longer possible at constant pressure. If T Tc , the substance cannot be liquefied, no matter how great the pressure. w Substances in this region are sometimes known as “fluids” rather than as vapors or liquids. w

Vapor (Steam) Dome The dome-shaped region encompassing the two-phase, vapor-liquid equilibrium region. w It is bordered by the saturated liquid line and the saturated vapor line, both of which end at the triple line and end at the critical point. w The region below the vapor dome is also called: saturated liquid-vapor region, wet region, two-phase region, or saturation region. w

THERMODYNAMIC TABLES

STEAM IS NOT AN IDEAL GAS!

Steam Tables w w w Table A-1. 1 Saturation water -- temperature table Table A-1. 2 Saturation water -- pressure table Table A-1. 3 Superheated vapor
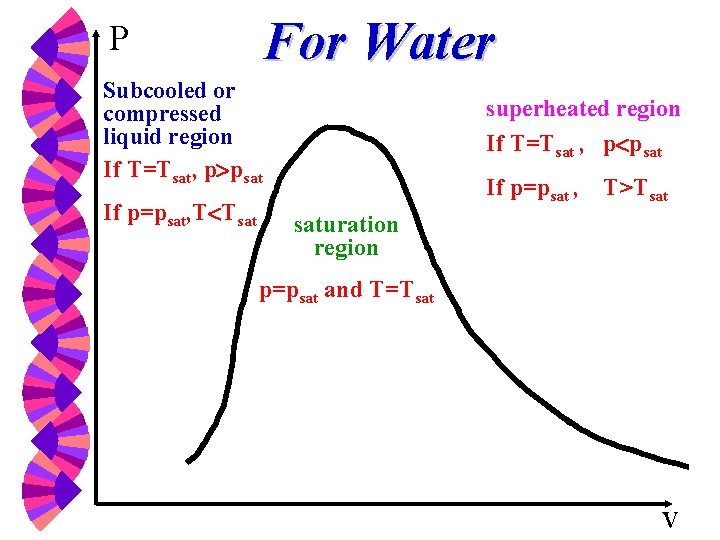
For Water P Subcooled or compressed liquid region If T=Tsat, p psat If p=psat, T Tsat superheated region If T=Tsat , p psat If p=psat , T>Tsat saturation region p=psat and T=Tsat v

Two properties are not independent in the vapor dome (the two-phase region) w The temperature and pressure are uniquely related. Knowing a T defines the P and vice versa. w Use quality to determine the state in two-phase region.
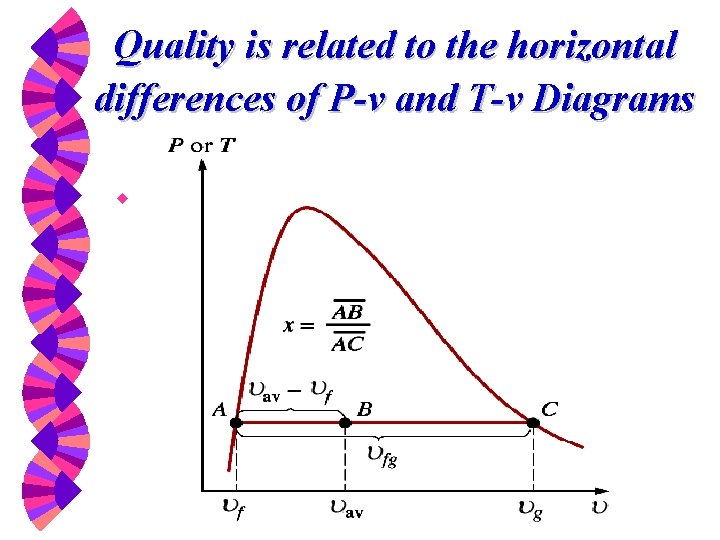
Quality is related to the horizontal differences of P-v and T-v Diagrams w

Quality w In a saturated liquid-vapor mixture, the mass fraction (not volume fraction) of the vapor phase is called the quality and is defined as w The quality may have values between 0 (saturated liquid) and 1 (saturated vapor). It has no meaning in the compressed liquid or superheated vapor regions.

What is v for something in the two-phase region? v = (1 -x)vf + xvg = vf + x(vg - vf) = vf + xvfg

Enthalpy in Two-Phase Region w H = U + p. V w h = u + pv h = (1 -x)hf + xhg = hf + x(hg - hf) = hf + xhfg

Saturated Mixture w In the saturated mixture region, the average value of any intensive property y is determined from where f stands for saturated liquid and g for saturated vapor.

Saturated Mixture v = vf u = uf h = hf s = sf + x(vg - vf) = vf + xvfg + x(ug - uf) = uf + xufg + x(hg - hf) = hf + xhfg + x(sg - sf) = sf + xsfg f : saturated liquid g : saturated vapor
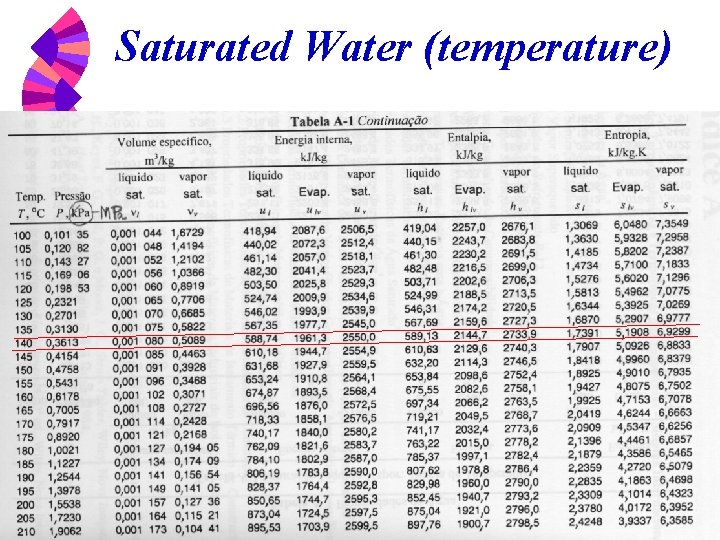
Saturated Water (temperature)
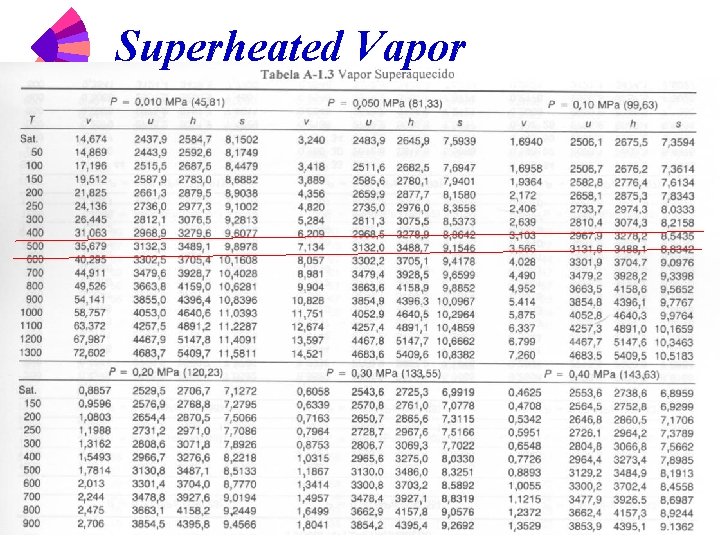
Superheated Vapor
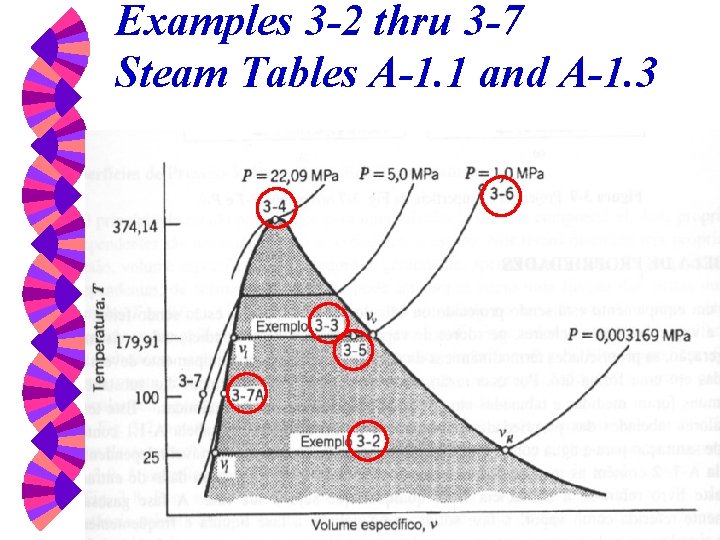
Examples 3 -2 thru 3 -7 Steam Tables A-1. 1 and A-1. 3

TEAMPLAY Complete the table below as a team. The substance is water. Make sure everybody understands how to do it! P (MPa) T( C) v(m 3/kg) 300 0. 15 0. 50 1. 0 0. 65 300 x (if appl. )
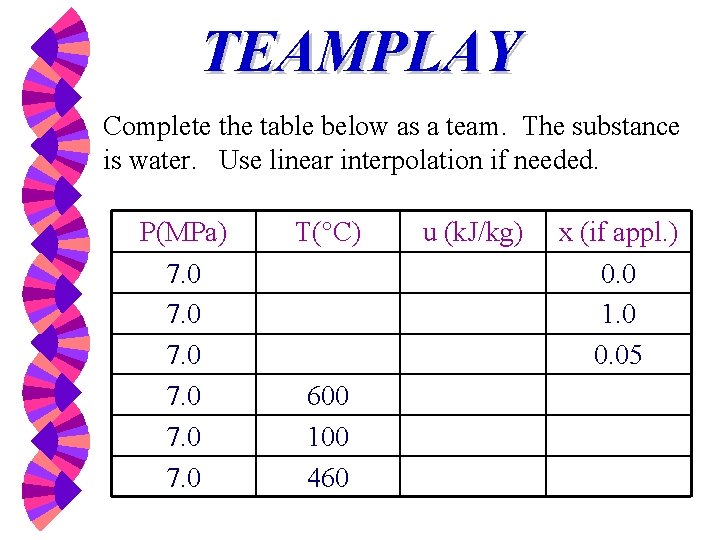
TEAMPLAY Complete the table below as a team. The substance is water. Use linear interpolation if needed. P(MPa) 7. 0 7. 0 T( C) 600 100 460 u (k. J/kg) x (if appl. ) 0. 0 1. 0 0. 05
- Slides: 50