Properties of Pure Substances first law of thermodynamics

-Properties of Pure Substances -first law of thermodynamics and applications

-Definition of Pure Substance A pure substance has a homogeneous and invariable chemical composition and may exist in more than one phase. -Phases of Pure Substance Solid Liquid Gas
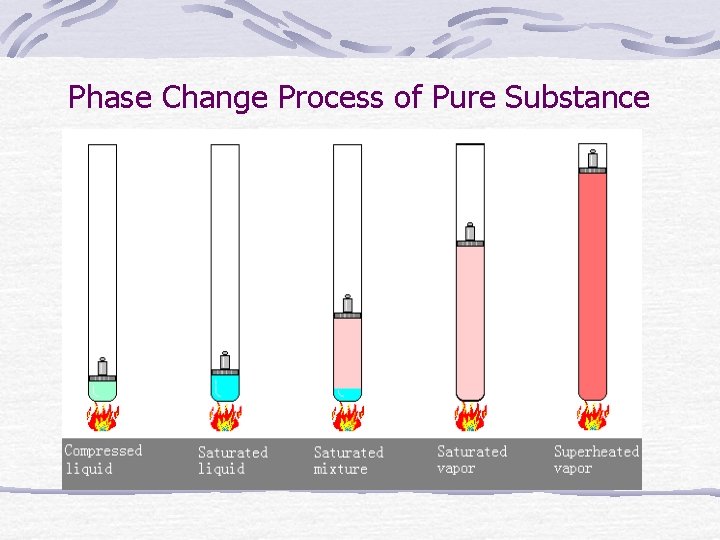
Phase Change Process of Pure Substance
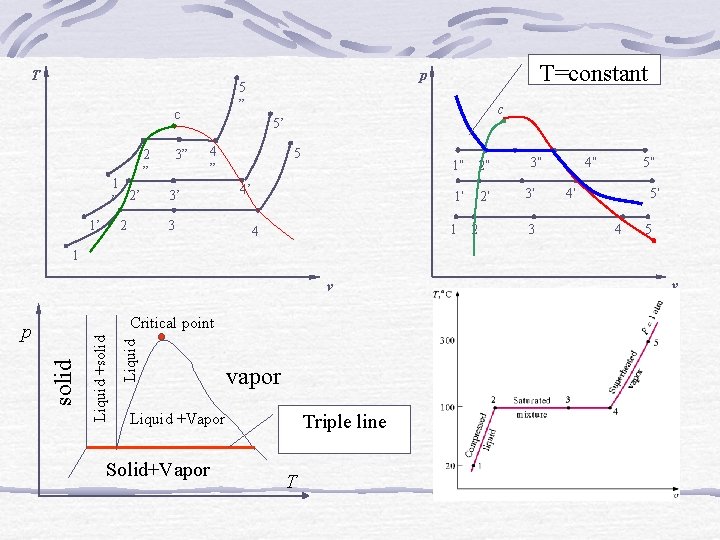
T c 1 ” 2’ 2 1’ 3” 2 ” T=constant p 5 ” c 5’ 4 ” 3’ 3 5 1” 2” 4’ 1’ 1 4 2’ 2 3” 3’ 3 4” 5” 4’ 5’ 4 5 1 v Liquid solid Liquid +solid Critical point p vapor Liquid +Vapor Solid+Vapor Triple line T v
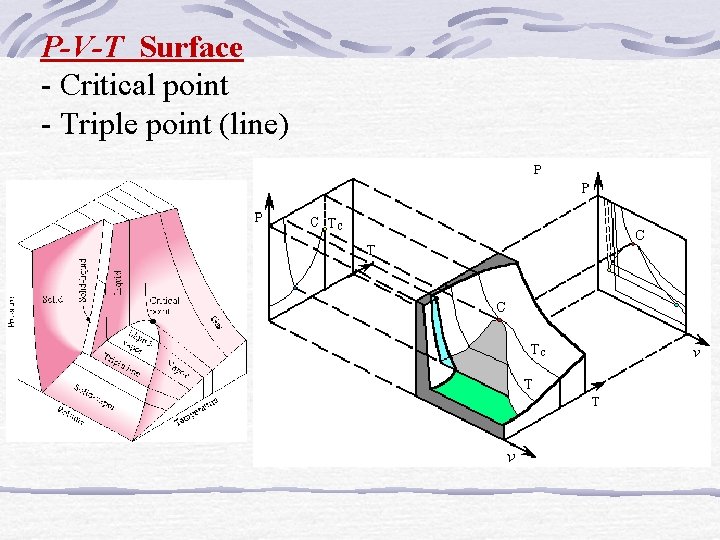
P-V-T Surface - Critical point - Triple point (line)
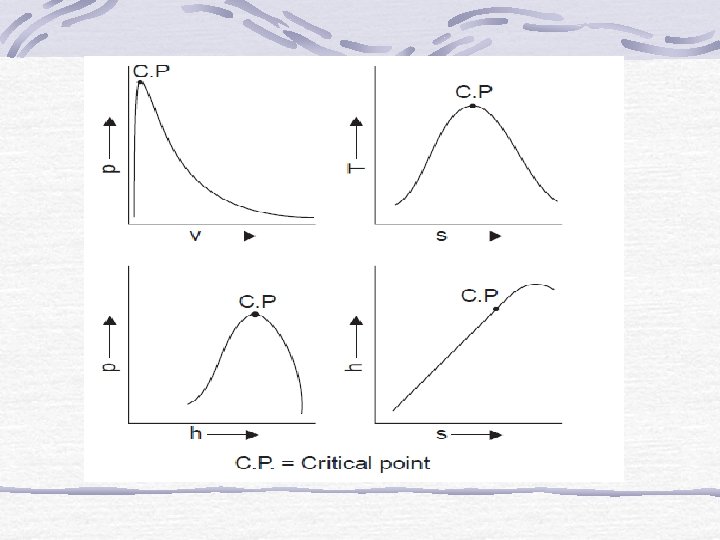
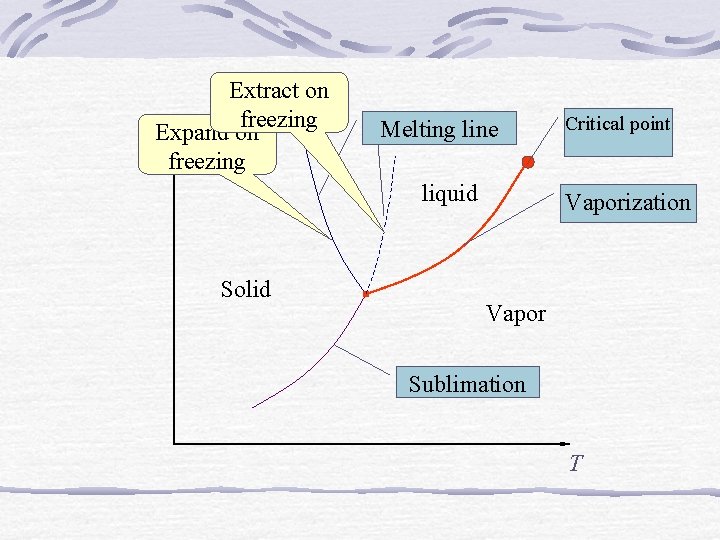
Extract on freezing Expand on P freezing Melting line liquid Solid Critical point Vaporization Vapor Sublimation T

Property In addition to the temperature, pressure, and volume data, contain the data for the specific internal energy u the specific enthalpy h and the specific entropy s. The enthalpy is a convenient grouping of the internal energy, pressure, and volume and is given by The enthalpy per unit mass is
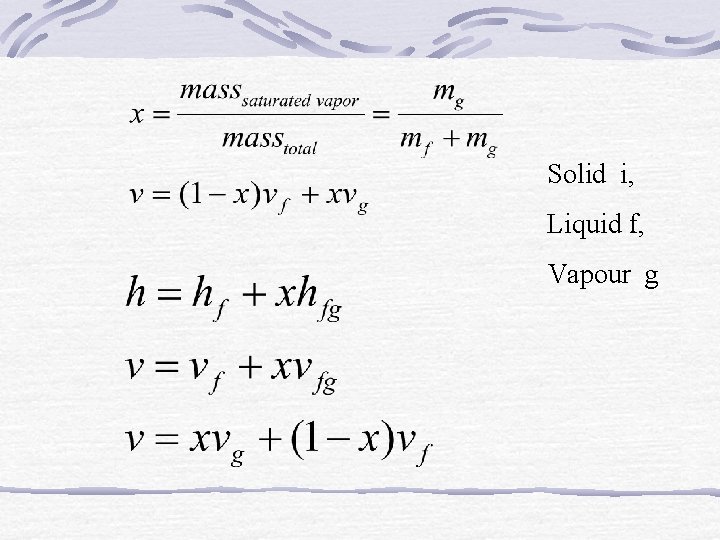
Solid i, Liquid f, Vapour g

First law of thermodynamics -Energy can be changed from one form to another, but it cannot be created or destroyed. The total amount of energy and matter in the Universe remains constant, merely changing from one form to another.
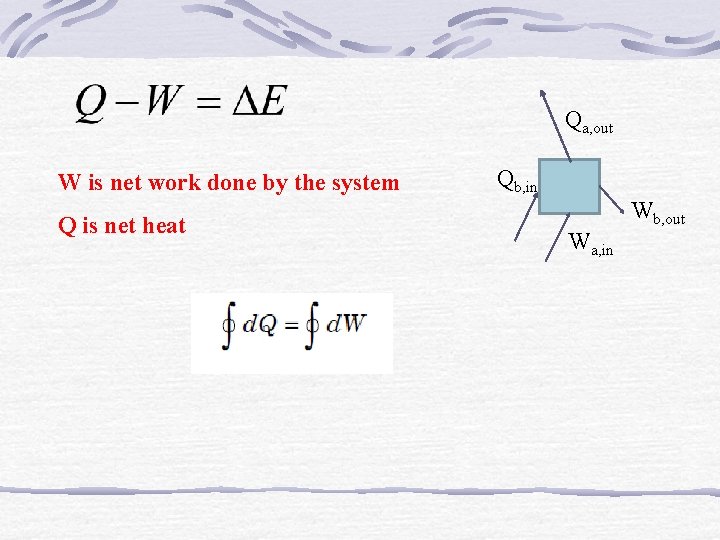
Qa, out W is net work done by the system Q is net heat Qb, in Wb, out Wa, in

If the work is done by the system on the surroundings, e. g. , when a fluid expands pushing a piston outwards, the work is said to be positive. i. e. , Work output of the system = + W If the work is done on the system by the surroundings, e. g. , when a force is applied to a rotating handle, or to a piston to compress a fluid, the work is said to be negative. i. e. , Work input to system = – W Heat received by the system = + Q Heat rejected or given up by the system = – Q.
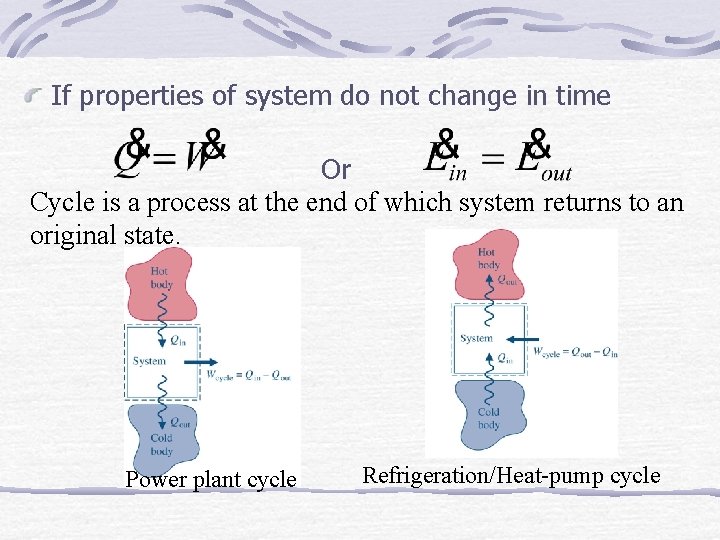
If properties of system do not change in time Or Cycle is a process at the end of which system returns to an original state. Power plant cycle Refrigeration/Heat-pump cycle

2 -5 The Ideal-Gas Equation of State 2 -5 -1 The Ideal-Gas The molecules of ideal-gas have no volume There are no attraction among molecules of ideal-gas 2 -5 -2 The Ideal-Gas Equation of State (1). p. V = m. RT pv=RT R------The gas constant (2) p. Vm = μRμT pvm=RμT Rμ-----The universal gas constant = 8. 314 k. J/kmol. K
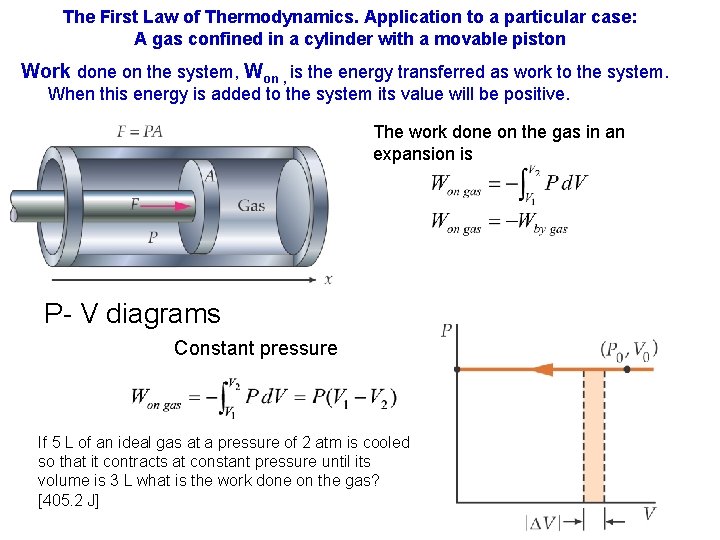
The First Law of Thermodynamics. Application to a particular case: A gas confined in a cylinder with a movable piston Work done on the system, Won , is the energy transferred as work to the system. When this energy is added to the system its value will be positive. The work done on the gas in an expansion is P- V diagrams Constant pressure If 5 L of an ideal gas at a pressure of 2 atm is cooled so that it contracts at constant pressure until its volume is 3 L what is the work done on the gas? [405. 2 J]
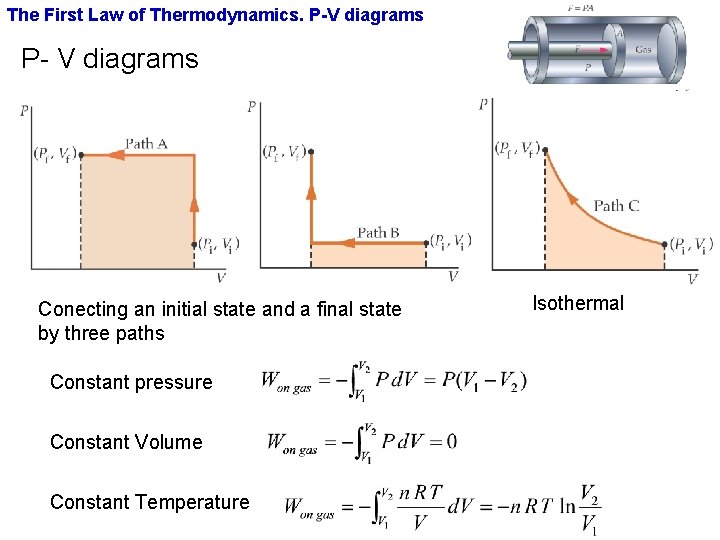
The First Law of Thermodynamics. P-V diagrams P- V diagrams Conecting an initial state and a final state by three paths Constant pressure Constant Volume Constant Temperature Isothermal
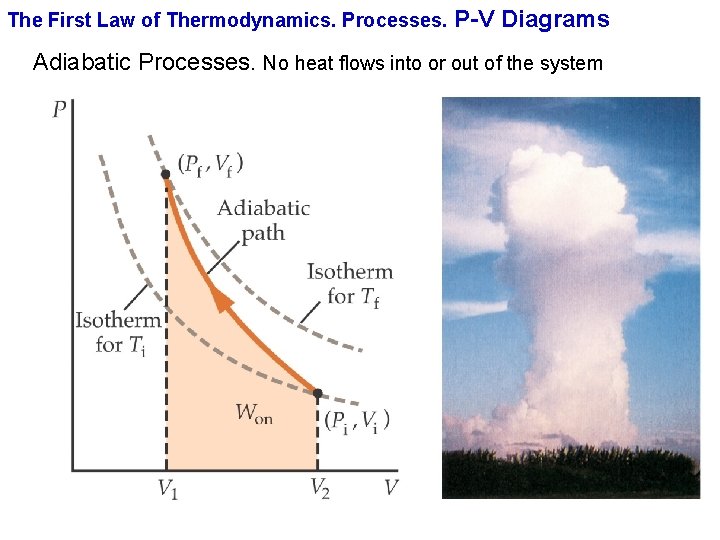
The First Law of Thermodynamics. Processes. P-V Diagrams Adiabatic Processes. No heat flows into or out of the system
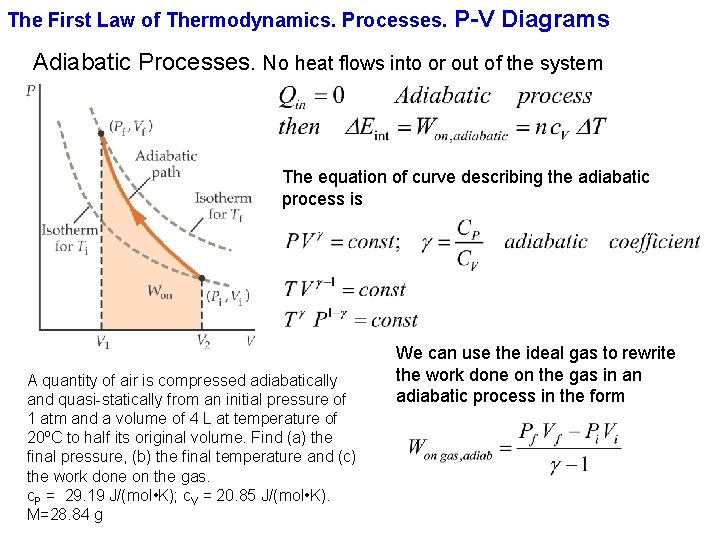
The First Law of Thermodynamics. Processes. P-V Diagrams Adiabatic Processes. No heat flows into or out of the system The equation of curve describing the adiabatic process is A quantity of air is compressed adiabatically and quasi-statically from an initial pressure of 1 atm and a volume of 4 L at temperature of 20ºC to half its original volume. Find (a) the final pressure, (b) the final temperature and (c) the work done on the gas. c. P = 29. 19 J/(mol • K); c. V = 20. 85 J/(mol • K). M=28. 84 g We can use the ideal gas to rewrite the work done on the gas in an adiabatic process in the form

Thank you
- Slides: 21