Properties of Metals and Nonmetals Periodic Table Classification

Properties of Metals and Nonmetals Periodic Table Classification
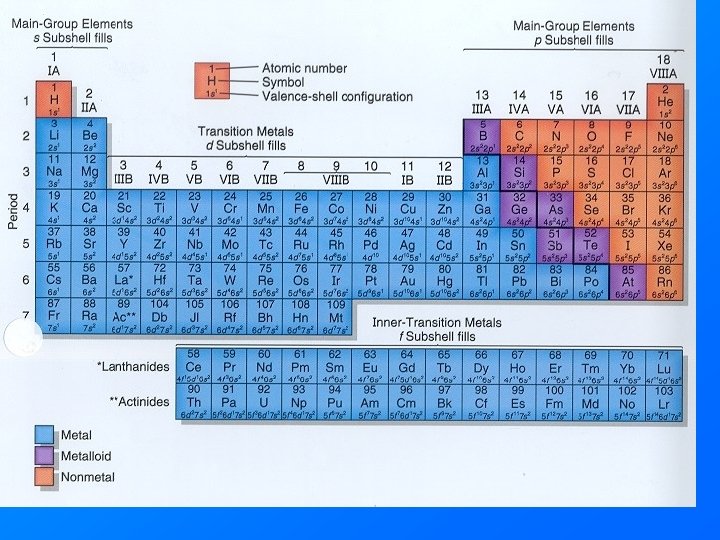

Physical Properties of Metals • Solids at room temperature, except Hg. • Malleable – pounded into sheets • Ductile – drawn into wires • Have Luster (shine) • Good Conductors of Heat & Electricity • High Melting/Boiling Points

Chemical Properties of Metals • Metals lose electrons to form positive ions. “Metals are losers” • Metals have positive oxidation numbers. • Low Ionization Energy • Low Electronegativity Ca+2

Physical Properties of Nonmetals • Generally gases or solids, except Br 2. • Solids are brittle. • Solids are dull. • Poor conductors of heat & electricity. • Low Melting and Boiling Points

Chemical Properties of Nonmetals • Nonmetals gain electrons to form negative ions. “Nonmetals are winners. ” • Nonmetals have negative oxidation numbers -1 F • High Ionization Energy. • High Electronegativity. • Properties are the OPPOSITE of metals.

Properties of Metalloids • Each metalloid has some metallic and some non-metallic properties. • For example, silicon is shiny like a metal, but brittle like a nonmetal.

Transition Metals • Elements in Columns 3 through 12 • Transition elements form brightly colored salts and brightly colored solutions. • Actinide and Lanthanide series are called the inner transition elements.
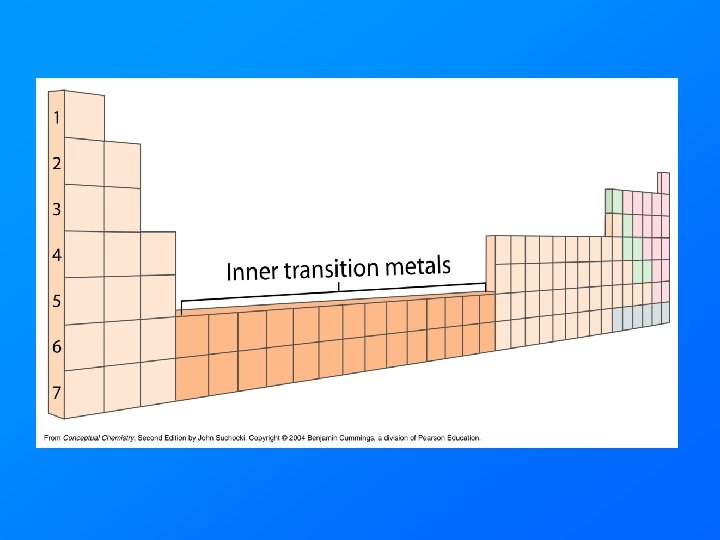
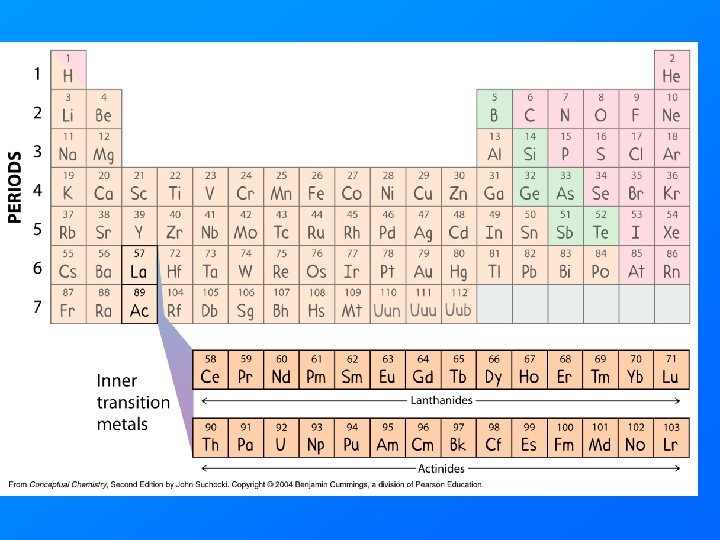

Valence Electrons and Periodicity electrons

Phases of the Elements • SOLIDS – Most elements • LIQUIDS – Bromine (non-metal) – Mercury (metal) • GASES – H 2, N 2, O 2, F 2, Cl 2 (diatomics) – Noble Gases (He, Ne, Ar, Kr, Xe, Rn)

Summary • Three general categories of classification: • # Valence Electrons play a key role
- Slides: 13