Properties of Metallic Ionic Bonds Properties electricity flows

Properties of Metallic & Ionic Bonds

ØProperties: Ø electricity flows through conductive them: _____ Ø can be hammered into sheets: malleable _______ Øcan be drawn into a wire: _______ ductile

ØAttraction of a metallic cation (+ ion) and valence electrons that are free to move (delocalized) ØDelocalized electrons ØElectrons do not belong to any one atom ØSometimes called a “sea of electrons”

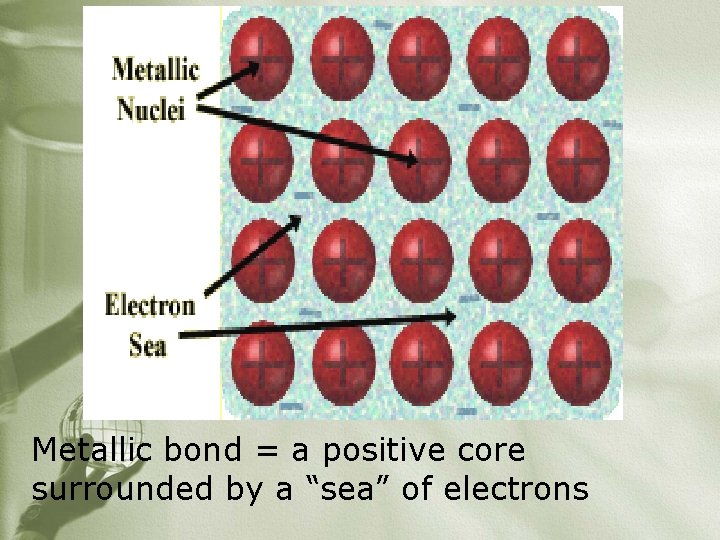
Metallic bond = a positive core surrounded by a “sea” of electrons

Electrical Conductivity • Since the electrons are free to move, if electrons from an outside source are added to a metal wire at one end, the electrons will move through the wire and come out at the other end at the same rate. Thus, this model accounts for the electrical conductivity of metals.

Luster • The free electrons in the "sea" can absorb photons, so metals are opaque. Electrons on the surface can bounce back light at the same frequency as the light that hits the surface. Therefore, the metal appears to be shiny. This model accounts for the luster of metals.

Malleability • The sea of electrons surrounding the cations act like a cushion and so, when the metal is hammered on, the overall composition of the structure of the metal is not harmed or changed. The cations may slide past each other, but the sea of electrons will adjust to the new formation of cations and keep the metal intact. Thus, this model accounts for the malleability and ductility of metals.

Heat Conductivity • Metals usually have high densities. Hence the atoms are closely packed. That makes heat transfer by conduction more efficient. Conduction is the transfer of heat energy by vibration of particles to transfer energy from one end of the metal to the other end. Metals also have the sea of electrons to help them in this conduction. Since electrons are very small compared to the metal ions, they can easily pass through the spaces between the cations and help to transfer heat energy along. Thus, this model accounts for thermal conductivity of metals.

Properties of Ionic Compounds Ø Most form solid crystals (called “salts”) at room temperature Ø Have High melting points (strength of electrostatic attraction) Ø Form electrolytes (ions in solution) which can conduct electricity when dissolved in water (aqueous). Ø When dissolved the electrons are permitted to move freely (compare to ionic compound crystal structure)

Ionic vs Metallic + + = =
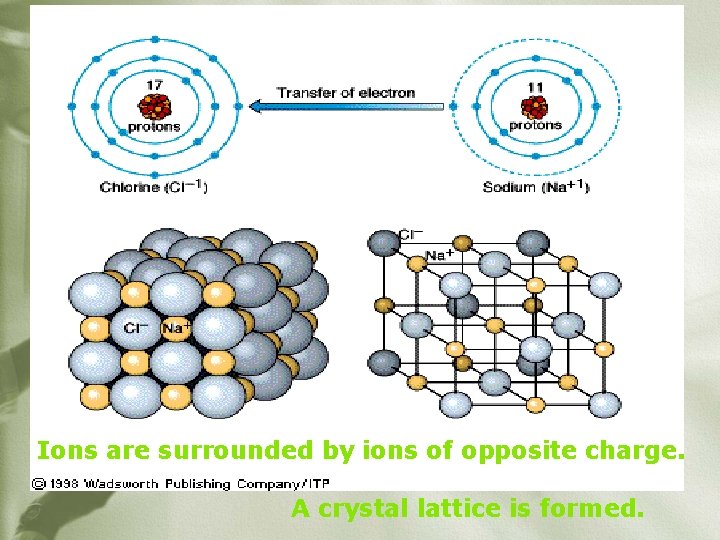
Ions are surrounded by ions of opposite charge. A crystal lattice is formed.

ØInvolves the transfer of electron(s) from a metal to a nonmetal. ØThe metal _____ loses electrons becoming positive and is called a _______. cation ____ gains electrons ØThe non-metal _____ negative and is called an becoming _______ anion _______.

Electrostatic Interactions “Opposites attract” Ø Cations and anions are attracted to each other through strong electrostatic attractions – stronger than metallic bonds Ø The result is a stable, low energy ionic compound Ø Ionic compounds are neutral since the charges of the cation and anion cancel

Properties of Covalent Bonds

Covalent Compound Properties of Covalent Compounds… • Have low melting and boiling points • Do not dissolve easily in water • Do not conduct electricity in solutions • Exist as gases, liquids, or solids • Have low electronegativity differences

Common Covalent Compounds Nitrous oxide (N 2 O) is laughing gas used as an anesthetic and to boost auto engine power Methane (CH 4) is a flammable gas used as fuel and in homes for domestic heating and cooking purposes. Sugars, like sucrose (C 12 H 22 O 11) and glucose (C 6 H 12 O 6), are used in food and energy production Hydrogen peroxide (H 2 O 2) is used as a bleaching agent, emetic (induces vomiting), and an antiseptic (clean cuts and scrapes)

Common Covalent Compounds Carbon dioxide (CO 2) is a gas used by plants during photosynthesis, produced in respiration; it is a byproduct of burning fossil fuels, and it has many uses as a fire extinguisher, refrigerant (dry ice), and carbonation in drinks

Common Covalent Compounds Water (H 2 O) is a liquid that is vital for life. It covers about 71% of earth’s surface, and makes up about 60% of the human body. It is known as the universal solvent, and has many unique properties and uses.

Diatomic Elements • Some nonmetal elements on the periodic table exist in nature only as pairs called diatomic molecules; bonded covalently – H, O, F, Br, I, N, Cl Hydrogen Molecule (H 2) http: //web. jjay. cuny. edu/~acarpi/NSC/5 -bonds. htm

IONIC FORMULAS AND LEWIS DOT STRUCTURES

Review of valence electrons • All electrons that fill the outside energy level are considered, “Valence Electrons” • How do you determine the number of valence electrons based upon position in the PT? • How do you determine the charge based on position in the PT?

Valence Electron Dot Structures for Ionic Compounds Example 1: Na & Cl 1. Determine the number of valence electrons for each element 2. From the valence count, draw the Lewis valence electron dot structures of Na and Cl 3. Show the electron is transferred from Na to Cl to form ions

So what IS an ionic bond? Ionic compounds do not actually bond. Electrons are completely gained or lost (transferred), NOT shared!!!! The electrostatic attraction is the ionic “bond. ”

Valence Electron Dot Structures for Ionic Compounds Example 2: Mg & O Example 3: Ca & Cl

Why do atoms gain or lose electrons? more stable Ø In order to become _____ Ø What makes something stable? ØIt is lower in energy ØIt has a full valence shell ØIt’s electron configuration is the same as a _____ noble ____ gas

Stability: Atom vs. Ion • Which is more stable, Na or Na+1? Every atom wants to be like a noble!

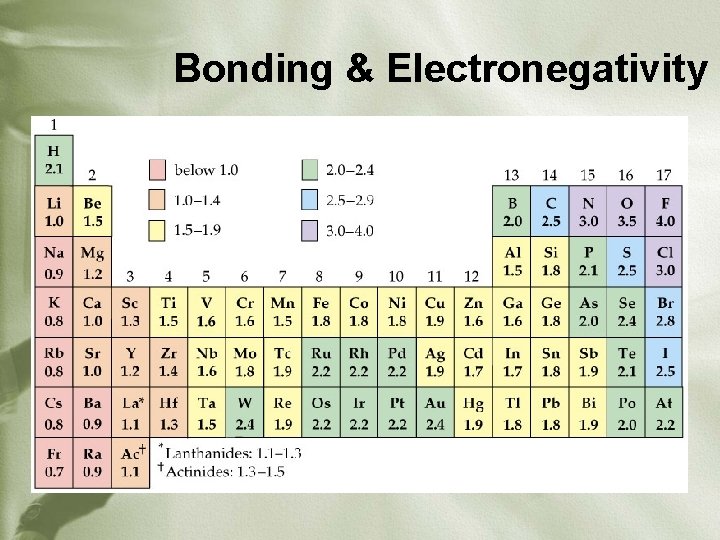
Electronegativity in Ionic Bonds • What does electronegativity mean? – Attraction for valence electrons in a bond • What element is the MOST electronegative? – Fluorine (F) • What element is the LEAST electronegative? – Francium (Fr)

Electronegativity in Ionic Bonds Ø The larger the difference between the EN values in a compound, the stronger the ionic bond. • What compound would have the STRONGEST ionic bond that is possible? • Fr. F I’m strong; does that mean I’m ionic?

Electronegativity Values

What is a formula unit? Ø One single unit of the ionic compound Ø Smallest ratio of cations to anions that form an electrically neutral compound Ex. Na. Cl vs. Mg. Cl 2

Common Polyatomic Ions Ammonium Acetate Carbonate Hydroxide Nitrate Nitrite Permanganate Phosphate Sulfite • • • (NH 4)+1 (C 2 H 3 O 2)-1 (CO 3)-2 (OH)-1 (NO 3)-1 (NO 2)-1 (Mn. O 4)-1 (PO 4)-3 (SO 4)-2 (SO 3)-2

How does the charge work? Ignore the subscripts and treat the group as one thing: (NO 3) -1 total charge is -1 (SO 4) -2 total charge is -2 (PO 4) -3 total charge is -3

Write the formula for these compounds: 1. Ca 2+ + (NO 3) 2. Cr 4+ + Cl 3. NH 4 + O 24. Cu 2+ + S 2+ 2 3 5. Ni + N + 26. Ag + O Ca(NO 3)2 Cr. Cl 4 (NH 4)2 O Cu. S Ni 3 N 2 Ag 2 O

Ionic Bonding Recap Questions Copy the question and answer in complete sentences 1. 2. 3. What is the total charge on an ionic compound? What holds the anions to the cations? In other words, how is an ionic “bond” formed? List the pair(s) of elements below that are likely to form ionic compounds? Remember that ionic compounds are formed between metals and nonmetals. Write the balanced formula. Cl, Br Na 4. 5. 6. Li, Cl K, He I, What properties characterize ionic compounds? Why do ionic compounds conduct electricity when they are melted or dissolved in water? What is a formula unit? What does it represent?

COVALENT BONDSDOT STRUCTURES AND MOLECULAR GEOMETRY

Covalent Bonds • Occurs when valence electrons are shared between atoms – usually between nonmetallic elements • Covalently bonded compound known as a molecule • These shared electrons are part of the valences of all atoms involved (satisfies octet rule) • Since electrons are shared, no charges appear • Many combinations can occur between two nonmetals – Example: carbon and oxygen can form carbon monoxide (CO) and carbon dioxide (CO 2) Carbon Dioxide

Rules for Lewis Structures • 1. Make certain that the bond is a covalent bond by checking the electronegativity difference. Then set up the skeleton structure as follows: – The atom with the lowest electronegativity will tend to go in middle – Place all the other atoms around this central atom – Attach these atoms to the central atom in reasonable fashion with single bonds

Rules for Lewis Structures ► 2. Sum valence electrons ► 3. Complete octets of peripheral atoms ► 4. Place leftover e- on central atom ► 5. If necessary use multiple bonds to fill the center atom's octet.
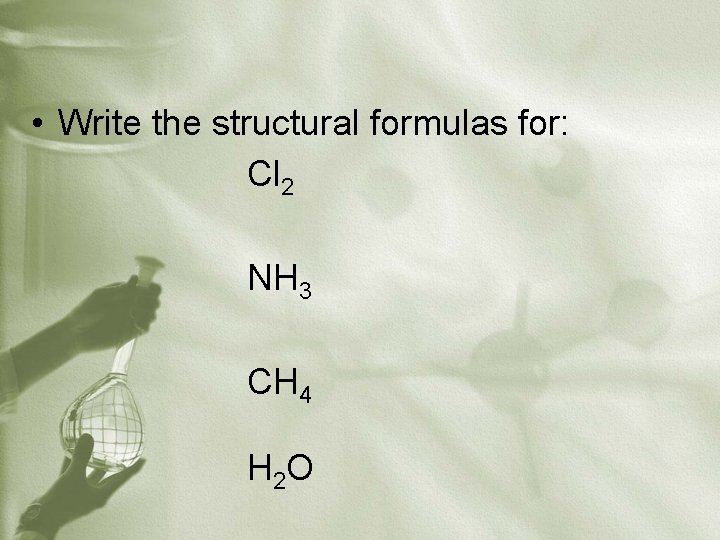
• Write the structural formulas for: Cl 2 NH 3 CH 4 H 2 O

Polyatomic Ions (K ONLY) • Draw the electron dot structures for: CO 32 NO 3 SO 32 SO 42 -
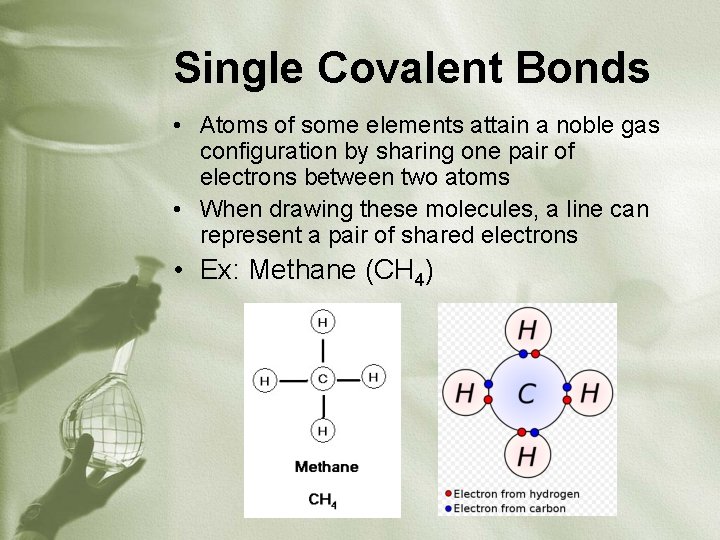
Single Covalent Bonds • Atoms of some elements attain a noble gas configuration by sharing one pair of electrons between two atoms • When drawing these molecules, a line can represent a pair of shared electrons • Ex: Methane (CH 4)
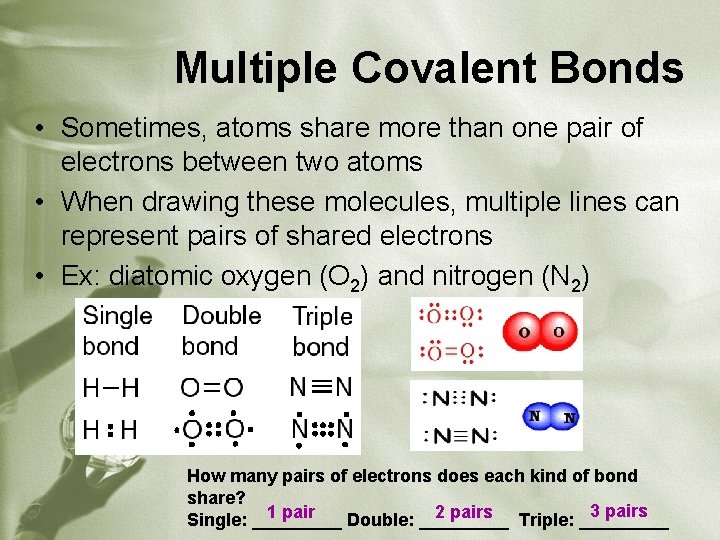
Multiple Covalent Bonds • Sometimes, atoms share more than one pair of electrons between two atoms • When drawing these molecules, multiple lines can represent pairs of shared electrons • Ex: diatomic oxygen (O 2) and nitrogen (N 2) How many pairs of electrons does each kind of bond share? 3 pairs 1 pair 2 pairs Triple: _____ Single: _____ Double: _____
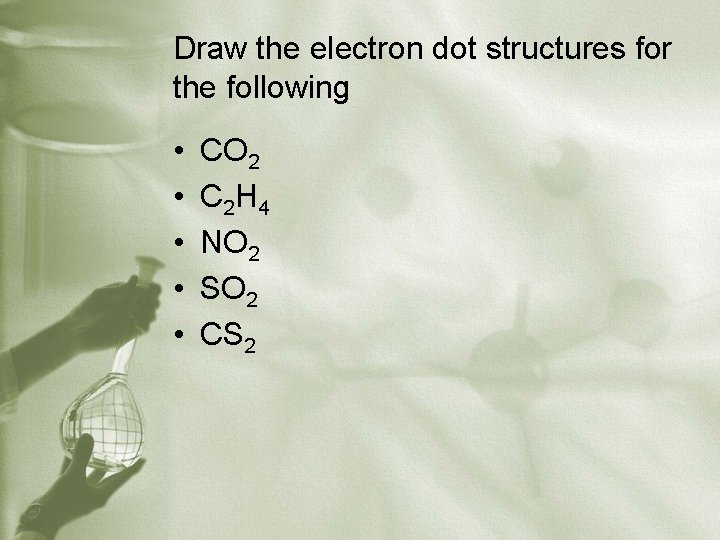
Draw the electron dot structures for the following • • • CO 2 C 2 H 4 NO 2 SO 2 CS 2

Exceptions to Octet Rule • Beryllium – Has 2 valence electrons – Full with 4 valence electrons – Ex: Be. I 2 • Aluminum – 3 valence electrons – Full with 6 valence electrons – Ex: Al. Cl 3 • Boron – 3 valence electrons – Full with 6 valence electrons – Ex: BH 3

Exceptions to Octet Rule Cont’d • Expanded octets – Some atoms bond so they have more than 8 electrons in their valence – Occurs only around a central nonmetallic atom from period 3 or higher (most commonly sulfur & phosphorous) – Ex: PCl 5 and SF 6 Trigonal Bipyramidal Octahedral
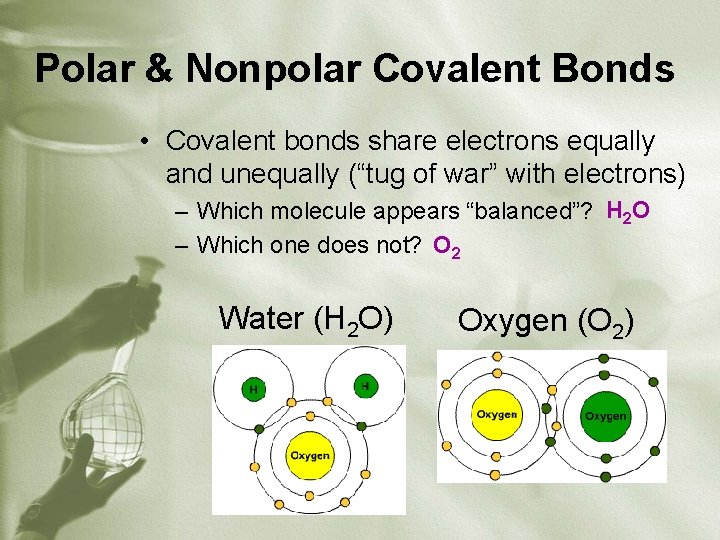
Polar & Nonpolar Covalent Bonds • Covalent bonds share electrons equally and unequally (“tug of war” with electrons) – Which molecule appears “balanced”? H 2 O – Which one does not? O 2 Water (H 2 O) Oxygen (O 2)

Polar Covalent Bonds • Unequal sharing of electrons • Occurs when atoms that share electrons have different electronegativities – Shared electrons spend a greater amount of time at the more electronegative atom • Example: water (H 2 O)
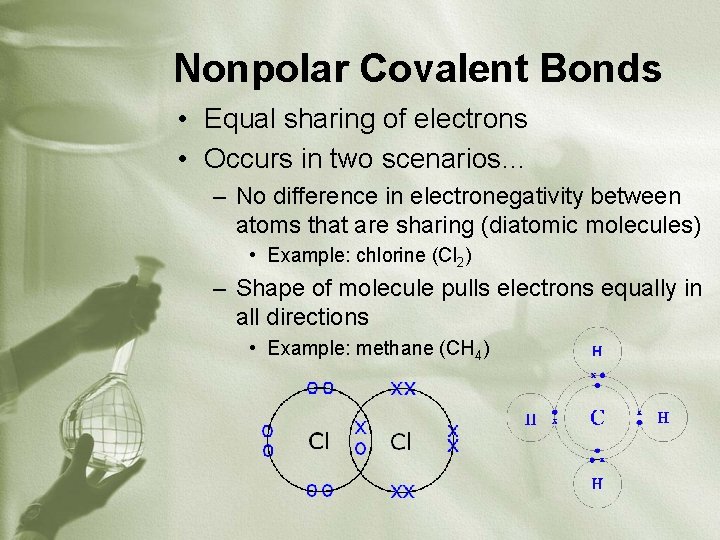
Nonpolar Covalent Bonds • Equal sharing of electrons • Occurs in two scenarios… – No difference in electronegativity between atoms that are sharing (diatomic molecules) • Example: chlorine (Cl 2) – Shape of molecule pulls electrons equally in all directions • Example: methane (CH 4)

Bonding & Electronegativity • Atoms bond according to the difference in their electronegativities • Look at table of EN values and find the difference between the two atoms involved (absolute value) • If EN difference is… • < 0. 5 = nonpolar covalent bond • 0. 5 – 1. 7 = polar covalent bond • >1. 7 = ionic bond
Bonding & Electronegativity
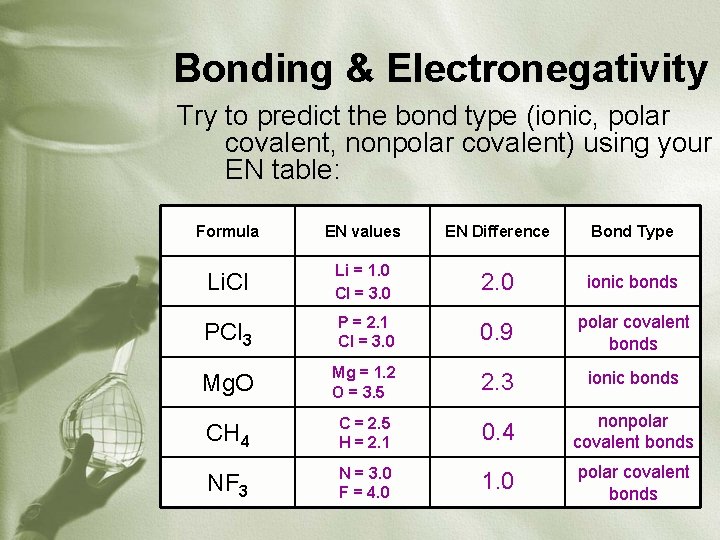
Bonding & Electronegativity Try to predict the bond type (ionic, polar covalent, nonpolar covalent) using your EN table: Formula EN values EN Difference Bond Type Li. Cl Li = 1. 0 Cl = 3. 0 2. 0 ionic bonds PCl 3 P = 2. 1 Cl = 3. 0 0. 9 polar covalent bonds Mg. O Mg = 1. 2 O = 3. 5 2. 3 ionic bonds CH 4 C = 2. 5 H = 2. 1 0. 4 nonpolar covalent bonds NF 3 N = 3. 0 F = 4. 0 1. 0 polar covalent bonds

Molecular Shapes # of Electron Groups 2 Number of Lone Pairs Electron Pair Arrangement 0 0 3 linear trigonal planar 1 0 1 Molecular Geometry bent tetrahedral Approximate Bond Angles 180° 120° <120° 109. 5° trigonal pyramid <109. 5° (~107°) 4 2 bent <109. 5°(~105°)
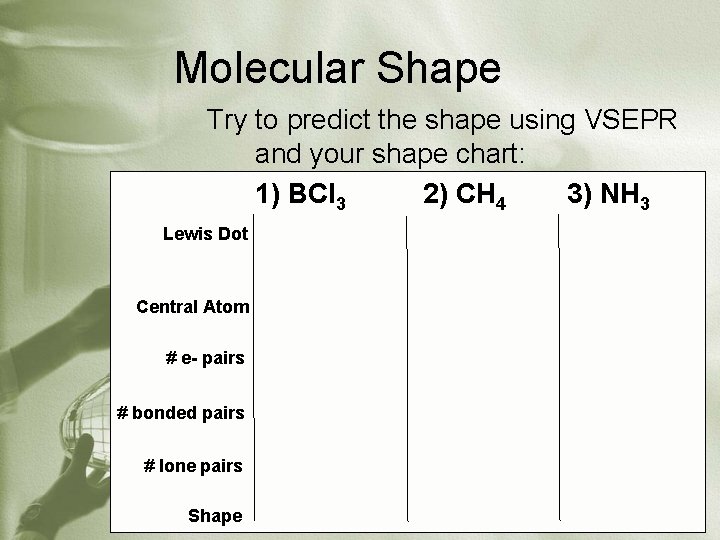
Molecular Shape Try to predict the shape using VSEPR and your shape chart: 1) BCl 3 2) CH 4 3) NH 3 Lewis Dot Central Atom # e- pairs # bonded pairs # lone pairs Shape
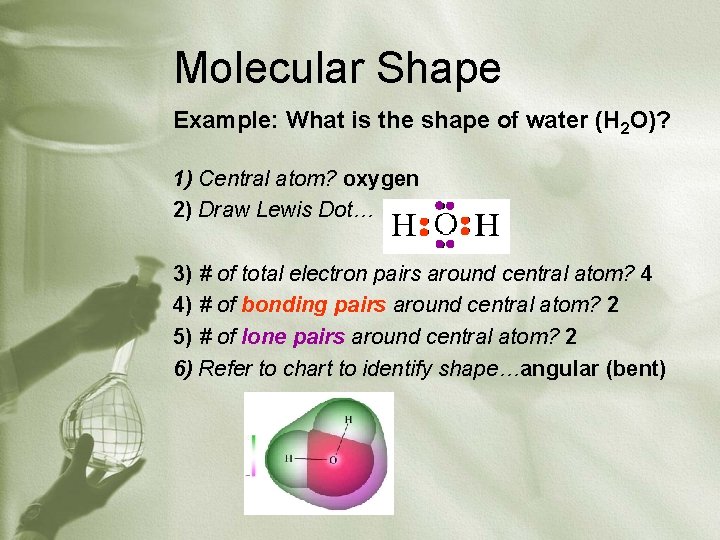
Molecular Shape Example: What is the shape of water (H 2 O)? 1) Central atom? oxygen 2) Draw Lewis Dot… 3) # of total electron pairs around central atom? 4 4) # of bonding pairs around central atom? 2 5) # of lone pairs around central atom? 2 6) Refer to chart to identify shape…angular (bent)

Molecular Shapes Applies to covalent compounds only. Use VSEPR steps (valence shell electron pair repulsion): 1) Identify the central atom as the element that can form the most bonds 2) Draw the Lewis dot structure for the molecule 3) Count total # of electron pairs around the central atom 4) Count # of bonding pairs of electrons around the central atom 5) Count # of lone pairs of electrons around the central atom 6) Look at summary chart, identify shape

Polar & Nonpolar Molecules • Polar molecules have a partial positive and partial negative charge – called a dipole • Dependent upon location and nature of the covalent bonds they contain Molecules are considered polar if they: • Have a polar covalent bond - EN difference between 0. 5 - 1. 7 • Have an asymmetrical shape - trigonal pyramidal - linear (with 2 different elements) - angular (bent) If both criteria are not met, it is either a nonpolar molecule or an ionic compound

Polar & Nonpolar Molecules *IMPORTANT: Polar & nonpolar molecules are different from polar & nonpolar bonds!!! Non-polar molecules have charges that are evenly distributed, due to shape (ex: any diatomic molecule, gasoline) Polar molecules have a partial positive and a partial negative charge (ex: water)
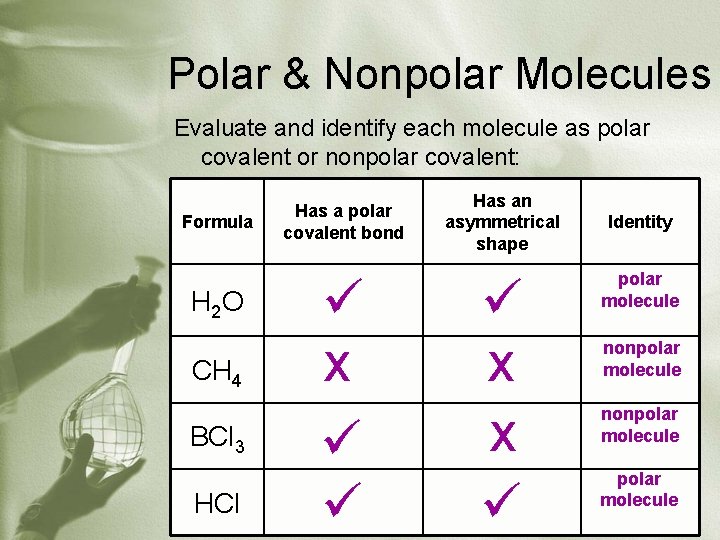
Polar & Nonpolar Molecules Evaluate and identify each molecule as polar covalent or nonpolar covalent: Formula H 2 O CH 4 BCl 3 HCl Has a polar covalent bond x Has an asymmetrical shape x x Identity polar molecule nonpolar molecule
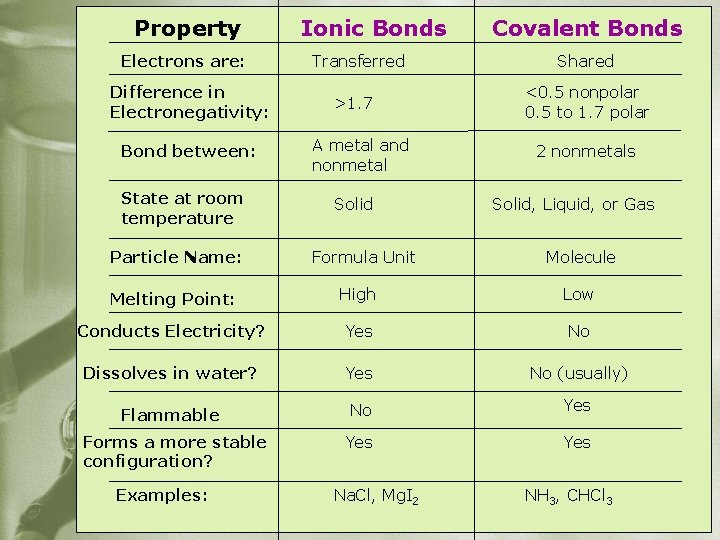
Property Electrons are: Difference in Electronegativity: Bond between: State at room temperature Particle Name: Ionic Bonds Transferred >1. 7 A metal and nonmetal Solid Formula Unit Covalent Bonds Shared <0. 5 nonpolar 0. 5 to 1. 7 polar 2 nonmetals Solid, Liquid, or Gas Molecule Melting Point: High Low Conducts Electricity? Yes No Dissolves in water? Yes No (usually) Flammable No Yes Forms a more stable configuration? Yes Examples: Na. Cl, Mg. I 2 NH 3, CHCl 3
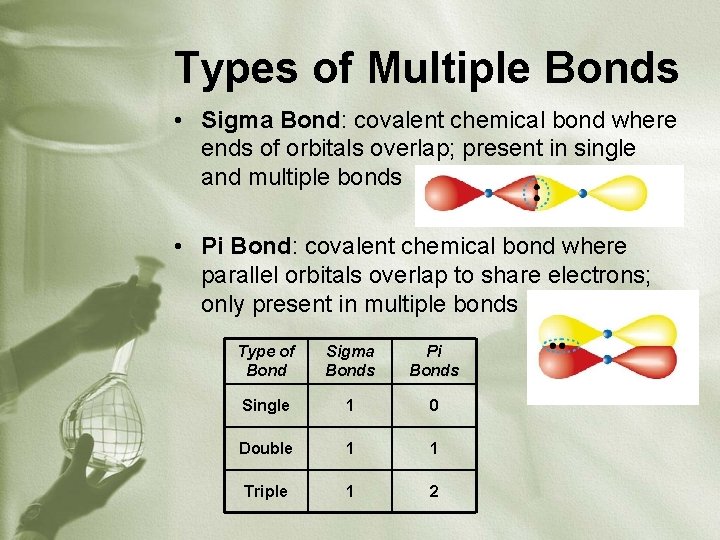
Types of Multiple Bonds • Sigma Bond: covalent chemical bond where ends of orbitals overlap; present in single and multiple bonds • Pi Bond: covalent chemical bond where parallel orbitals overlap to share electrons; only present in multiple bonds Type of Bond Sigma Bonds Pi Bonds Single 1 0 Double 1 1 Triple 1 2
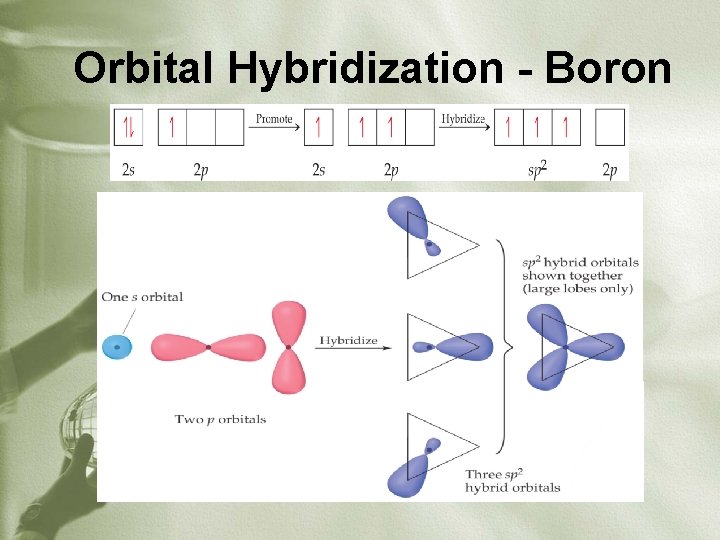
Orbital Hybridization - Boron
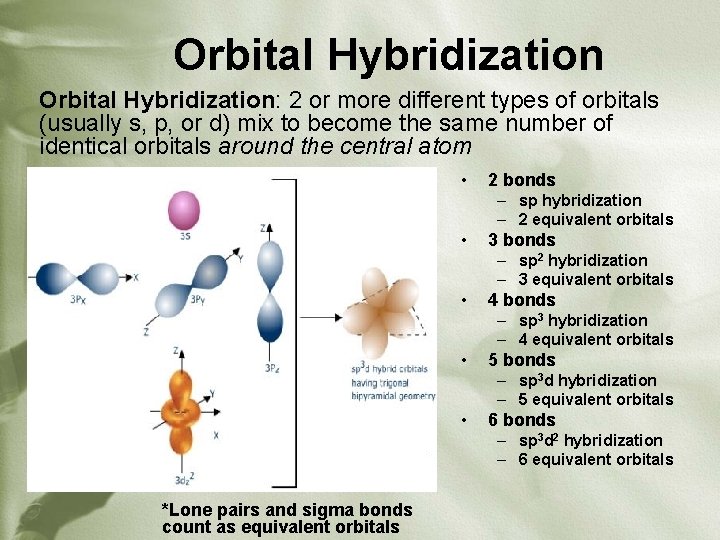
Orbital Hybridization: 2 or more different types of orbitals (usually s, p, or d) mix to become the same number of identical orbitals around the central atom • 2 bonds – sp hybridization – 2 equivalent orbitals • 3 bonds – sp 2 hybridization – 3 equivalent orbitals • 4 bonds – sp 3 hybridization – 4 equivalent orbitals • 5 bonds – sp 3 d hybridization – 5 equivalent orbitals • 6 bonds – sp 3 d 2 hybridization – 6 equivalent orbitals *Lone pairs and sigma bonds count as equivalent orbitals
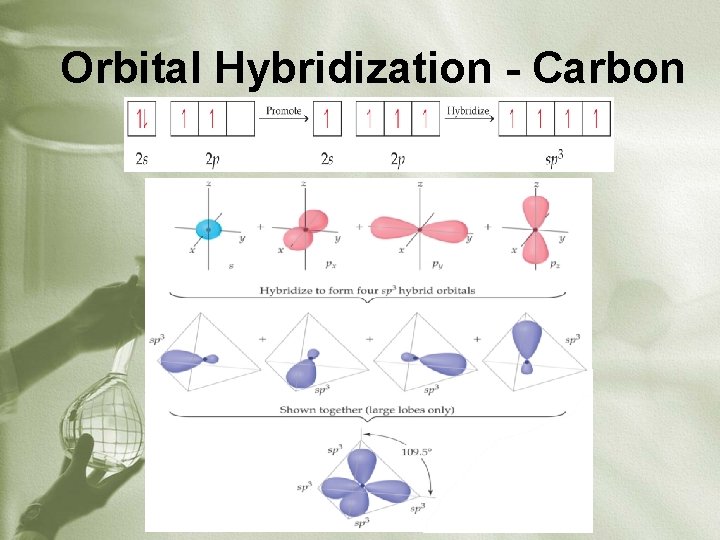
Orbital Hybridization - Carbon
- Slides: 64