Properties of matter Pure Substances Mixtures Solutions Separation

Properties of matter Pure Substances Mixtures Solutions Separation Methods Chapter 2

Properties of matter Chapter 2

Properties of Matter Properties of matter can be classified as EXTENSIVE or INTENSIVE properties. INTENSIVE Extensive Property: depends on the amount of matter present. Example: weight. Extensive Intensive Property is which is INDEPENDENT of the amount of substance but depends Intensive on the identity of substance. Example: Boiling Temperature. Properties of matter can be classified as PHYSICAL or CHEMICAL properties. CHEMICAL Physical Property: Can be observed without changing the identity of the substance. Physical Example: mass, length, color, boiling point. Chemical Property: Can be observed only by testing the substance in a chemical reaction Chemical or change, and so, the substance will change its identity. Example: Flammability, reaction with water. 3

Classify the following Properties of Matter Property Intensive or Extensive Property Boiling Point Melting point Volume Flammability Mass Density Tarnish in air Conductivity Rusting 4 Chemical or Physical

Changes of Matter Changes in matter can be classified as PHYSICAL or CHEMICAL changes. CHEMICAL Physical change: It is the one that changes the form of a substance Physical without changing the identity of the substance. Example: freezing, dissolving, boiling, cutting, etc. Chemical change: It is the one that changes the identity of the substance Chemical through a chemical reaction Example: rust, tarnish, burning, oxidation, etc. 5

Signs of a chemical change If a chemical change takes place you will observe at least one of the following signs. Change in color Production of smell Formation of Gas Formation of precipitate (solid) Change in light or heat 6

Classify the following Changes of Matter Change Boiling alcohol Dissolving salt in water Burning a match Melting ice Grinding spices 7 Physical of chemical

Pure Substances Mixtures Chapter 2

Pure Substances Pure substances have a definite composition (The same intensive properties throughout the sample) They can be classified in two types B Elements: Elements Formed by a unique type of atoms Examples: H 2, O 2, Cl 2, Na, Ne, Ar, etc. Compounds: Compounds Formed by a combination of different elements Examples: H 2 O, Ca. CO 3, Na. Cl, H 2 SO 4, etc C N O F Ne Cl Ar Br I
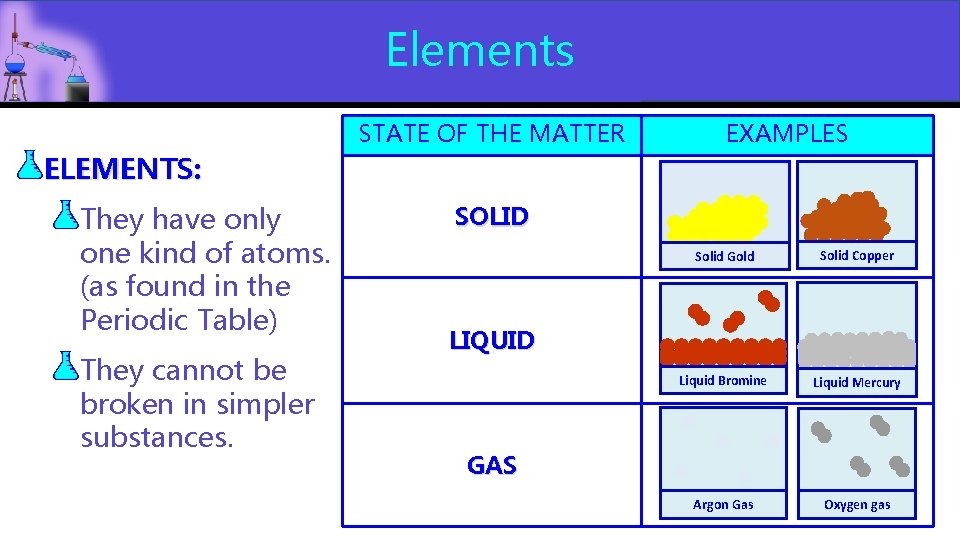
Elements STATE OF THE MATTER EXAMPLES ELEMENTS: They have only one kind of atoms. (as found in the Periodic Table) They cannot be broken in simpler substances. SOLID Solid Gold Solid Copper Liquid Bromine Liquid Mercury Argon Gas Oxygen gas LIQUID GAS

Compounds STATE OF THE MATTER COMPOUNDS: COMPOUNDS Can be broken into simpler SOLID substances. They are formed by 2 or more LIQUID different atoms combined together. H 2 O, Ca. CO 3, Na. Cl, GAS H 2 SO 4, etc EXAMPLES Sodium Chloride Water Ethanol Carbon Dioxide Hydrogen Chloride

Mixtures vs. Impurities If one of the substance in a mixture is in a very small proportion, proportion it is considered Impurity refers to the presence of a not desirable substance in a pure substance, which contaminates it , and compromises its purity. In everyday life, we do not care about how pure a substance is, but most Industries reliability depend on the purity of the substances that they produce or sell. Food must be tested to ensure they do not have impurities that can harm us. When taking a medicine, it is very important to know that there are no harmful impurities that can get us sick.

Mixtures are formed when two or Mixtures more pure substances mix together without a chemical reaction. Mixtures can be formed by Atoms and atoms = brass (copper and zinc) Atoms and compounds= iodine dissolved in ethanol Compounds and compounds = water and Copper (II) Sulfate

Classification of Mixtures can be classified in two types Heterogeneous mixtures: These are the ones in mixtures which their properties vary throughout the substance. Heterogeneous mixtures are solids or liquids, never gases. Examples: marble, water/chalk, sand/salt, etc. MIXTURES HETEROGENEOUS Note: If the substances are two liquids which do not mix – immiscible – (water and oil) each liquid immiscible forms a FASE. In the example we see how oil “floats” on water Homogeneous mixtures: also called Solutions: mixtures Solutions Have the same properties in any point of the substance. Homogeneous mixtures can be solids, liquids or gases Examples: tea, water and salt, the air we breath. Solutions are a very important part of chemistry EXAMPLES Sulfur powder and iron fillings Marble HOMOGENEOUS Water/oil Water/Na. Cl not completely dissolved Cl 2 / F 2 Gaseous solution Water/ Na. Cl Aqueous solution Ethanol/ Iodine Alcoholic solution Copper/Zinc Brass

Pure substances or mixtures? element The examples below are pure substances element Monoatomic Element Diatomic Elements Compound The examples below are mixtures 1 element & 1 compound 2 elements 2 compounds

Elements, compounds and mixtures Classify each of the pictures below by placing the correct label in the blank space below. Each circle represents an atom and each different color represents a different kind of atom. If two atoms are touching together, they are bonded together A= ELEMENT B=COMPOUND C= MIXTURE OF ELEMENTS D= MIXTURE OF COMPOUNDS E = MIXTURE OF ELEMENTS AND COMPOUNDS

Solutions Chapter 2

Solutions SOLUTIONS: Are homogeneous mixtures. SOLUTIONS Solutions can be solids, liquids or gaseous. In chemistry, we use liquid solutions most of the time. The particles in solutions are so small that we cannot see them with the naked eye. That’s why solutions are translucent. (clear, not cloudy)

Components of a solution Solutions are formed by two substances Solvent: The substance in higher proportion. Solute: The chemical in lower proportion. This can be a gas, liquid or solid. Example: Volatile liquids: liquids These are solvents that evaporate easily, like alcohol or acetone. The force that keeps the molecules together in these compounds are weak so the molecules escape the liquids more easily. Water is NOT a very volatile liquid. For dissolving the following solute Sugar, salt, etc Use the following solvent Water Ink, iodine Alcohol Nail polish Acetone, Thinner

What substances are present in a solution? Solutions are formed by two substances Solvent: The substance in higher proportion. Depending on the kind of solvent, we have: Aqueous Solution: These are the ones where the solvent is water. Like tea or coffee. If nothing is stated, Aqueous Solution we will assume that all solutions will be aqueous. Alcoholic Solution: These are solutions where the solvent is alcohol. Like Iodine dissolved in alcohol for disinfection purposes Solute: The chemical in lower proportion. This can be a gas, liquid or solid. Some solutes dissolve easier than others. The amount of solute that can be dissolved in 100 g of water is a very good source of information about how soluble a compound is. Electrolytes: Sometimes the solute is: Electrolytes • • An Ionic compound (formed by a metal+ + nonmetal-) that separate in their ions when dissolve in water An acid (Compounds formed by hydrogen+ + nonmetal-+)hat release hydrogen ions in solution acid The charge of those ions will allow the solution to conduct electricity. These substances are called electrolytes. (Yes, the same as in Gatorade) (
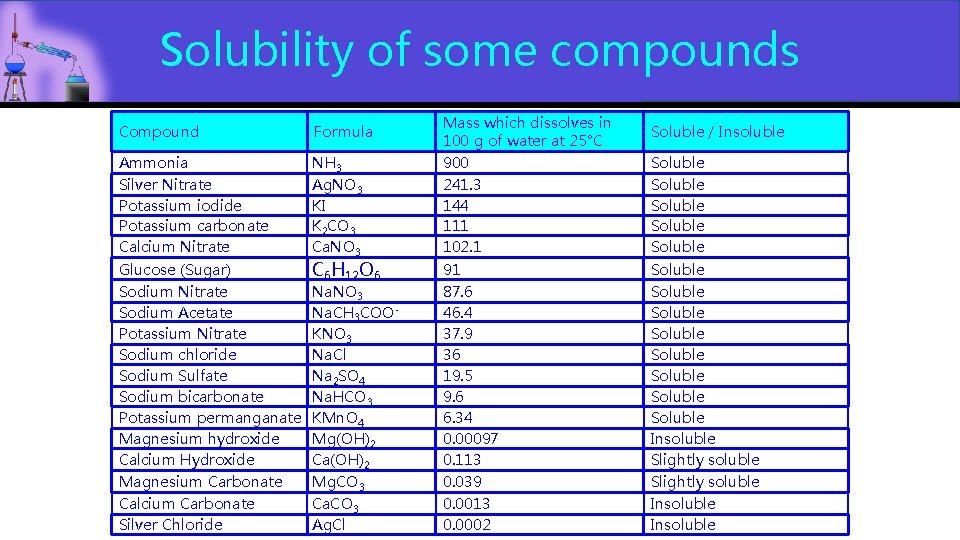
Solubility of some compounds Compound Ammonia Silver Nitrate Potassium iodide Potassium carbonate Calcium Nitrate Glucose (Sugar) Sodium Nitrate Sodium Acetate Potassium Nitrate Sodium chloride Sodium Sulfate Sodium bicarbonate Potassium permanganate Magnesium hydroxide Calcium Hydroxide Magnesium Carbonate Calcium Carbonate Silver Chloride Formula NH 3 Ag. NO 3 KI K 2 CO 3 Ca. NO 3 C 6 H 12 O 6 Na. NO 3 Na. CH 3 COO KNO 3 Na. Cl Na 2 SO 4 Na. HCO 3 KMn. O 4 Mg(OH)2 Ca(OH)2 Mg. CO 3 Ca. CO 3 Ag. Cl Mass which dissolves in 100 g of water at 25°C 900 241. 3 144 111 102. 1 91 87. 6 46. 4 37. 9 36 19. 5 9. 6 6. 34 0. 00097 0. 113 0. 039 0. 0013 0. 0002 Soluble / Insoluble Soluble Soluble Soluble Soluble Insoluble Slightly soluble Insoluble
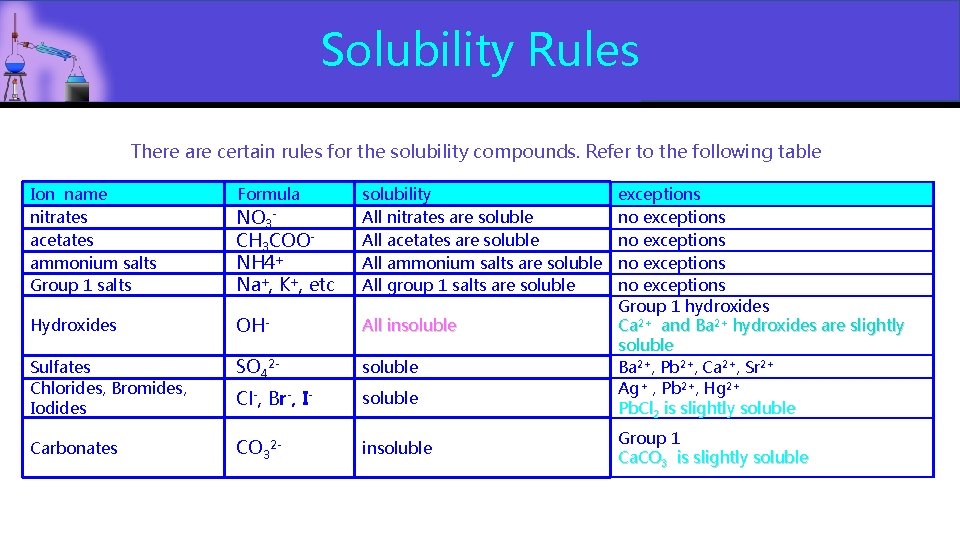
Solubility Rules There are certain rules for the solubility compounds. Refer to the following table Ion name nitrates acetates ammonium salts Group 1 salts Formula NO 3 CH 3 COO NH 4+ Na+, K+, etc solubility All nitrates are soluble All acetates are soluble All ammonium salts are soluble All group 1 salts are soluble Hydroxides OH- All insoluble Sulfates Chlorides, Bromides, Iodides SO 42 - soluble Cl-, Br-, I- soluble Carbonates CO 32 - insoluble exceptions no exceptions Group 1 hydroxides Ca 2+ and Ba 2+ hydroxides are slightly soluble Ba 2+, Pb 2+, Ca 2+, Sr 2+ Ag+ , Pb 2+, Hg 2+ Pb. Cl 2 is slightly soluble Group 1 Ca. CO 3 is slightly soluble

Solutions Concentration: is a measurement of how much solute is Concentration present in the solution. The more solute a solution has, the more CONCENTRATED it is. CONCENTRATED The less solute it has, the more DILUTED it is. DILUTED More concentrated More diluted

How to prepare a solution To prepare STANDARD solutions in chemistry, we use a VOLUMETRIC FLASK. Most of the time in the lab we work with AQUEOUS solutions, AQUEOUS where the solvent is water We will get back to solutions and make calculations about them later in the year. SOLVENT: The SOLVENT chemical in bigger proportion CAPACITY MARK: The level MARK that marks exactly the volumetric flask capacity SOLUTE: the chemical in SOLUTE smaller proportion

Separation Methods Chapter 2

Methods of separation SEPARATION METHOD WHAT TO SEPARATE FILTRATION A solid from a liquid CRYSTALLISATION A Solute from its solution EVAPORATION A solute from its solution SIMPLE DISTILLATION A solvent from a solution FRACTIONAL DISTILLATION Two miscible liquids PAPER CHROMATOGRAPHY Different substances from a solution

FILTERING When separating a mixture, you need to know the properties of each substance present in the mixture. If one of them is soluble in water and the other is not, you can use FILTERING The substance left in the filter paper is the RESIDUE and the liquid that passes the filter is the FILTRATE

Crystallisation After the filtration takes place, you need to evaporate as much water as you can so the crystals of the substance begin forming. This happens because most of the substances are less soluble at lower temperatures. This process is called crystallization+ Steps: 1 2 3 4 5 6
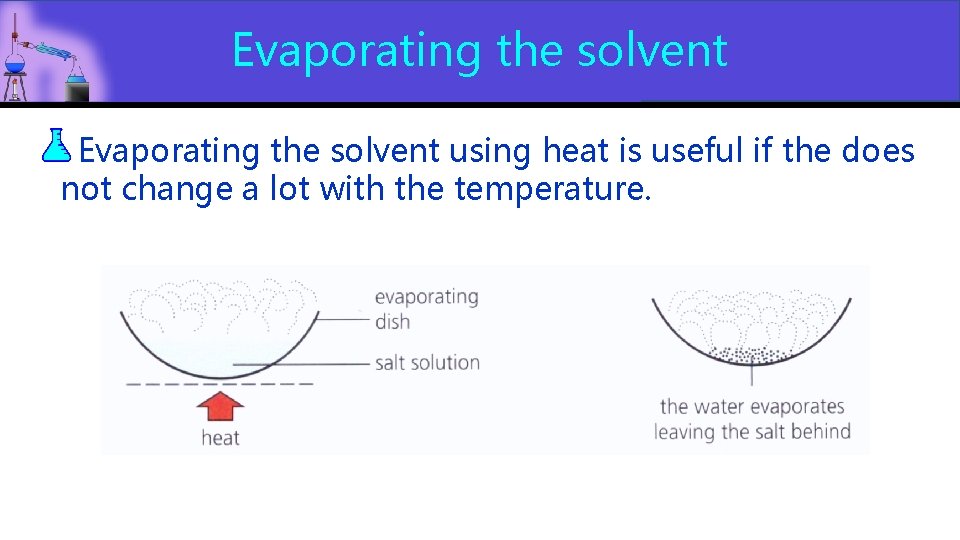
Evaporating the solvent using heat is useful if the does not change a lot with the temperature.
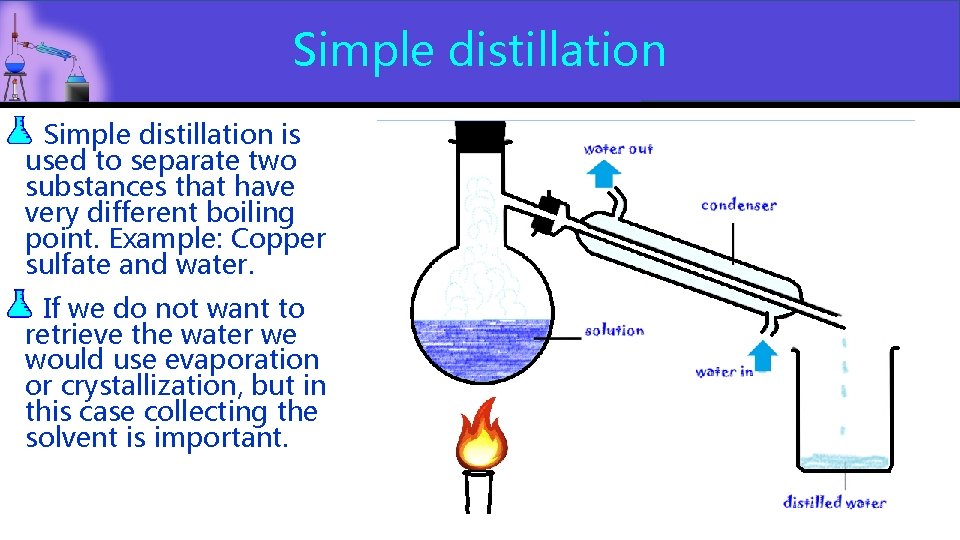
Simple distillation is used to separate two substances that have very different boiling point. Example: Copper sulfate and water. If we do not want to retrieve the water we would use evaporation or crystallization, but in this case collecting the solvent is important.

Fractional Distillation Fractional distillation is used to separate a mixture of miscible liquids that have different boiling points. Example: water and alcohol. points Alcohol will distill first since the Boiling Point is around 78°C. The temperature in the column will stay at 78°C while alcohol is distilling. Water will not distill since the column will be at a lower temperature than 100°C and it will fall down back to the round bottom flask After all the alcohol has passed through, the temperature of thermometer will rise again until it reaches 100°C.

Fractional distillation in industry CRUDE OIL: Fractional distillation is largely used in to separate the OIL components of crude oil which are compounds made of only carbon and hydrogen (hydrocarbons). The bigger and more (hydrocarbons) complex the molecules, the higher the melting and boiling point. ETHANOL: Alcohol is produced using fermentation of sugary ETHANOL plants. The fermentation takes place in vessels where water and plants are mixed and let to “ferment”. This process produces alcohol which has to be distilled to separate it from the mixture. AIR: AIR Air is cooled until liquid and then separated by warming it up slowly to separate the gases with different boiling points.
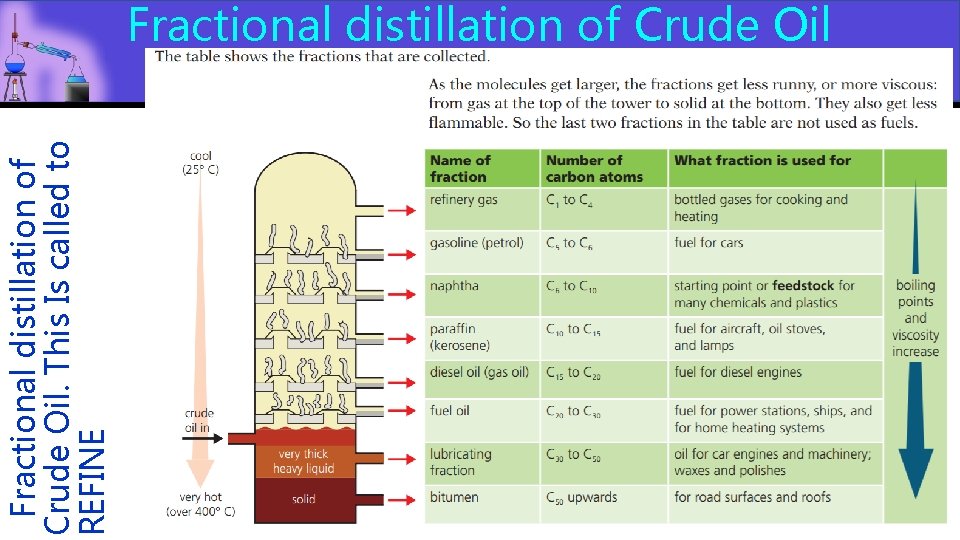
Fractional distillation of Crude Oil. This Is called to REFINE Fractional distillation of Crude Oil

Fractional distillation in Alcohol Production ETHANOL: Ethanol is made in two ways, one biological and one chemical. We will ETHANOL cover the biological only for now. By fermentation – the biological way : Ethanol is made from glucose using yeast, in the absence of air Yeast is a mass of living cells. The enzymes in it catalyse the reaction. The process is called fermentation, and it is exothermic. fermentation exothermic Ethanol can be made in this way from any substance that contains sugar, starch, or cellulose. (These break down to glucose. ) For example it can be made from sugarcane, maize, potatoes, and wood. The yeast stops working when the % of ethanol reaches a certain level, or if the mixture gets too warm. The ethanol is separated from the final mixture by fractional distillation.
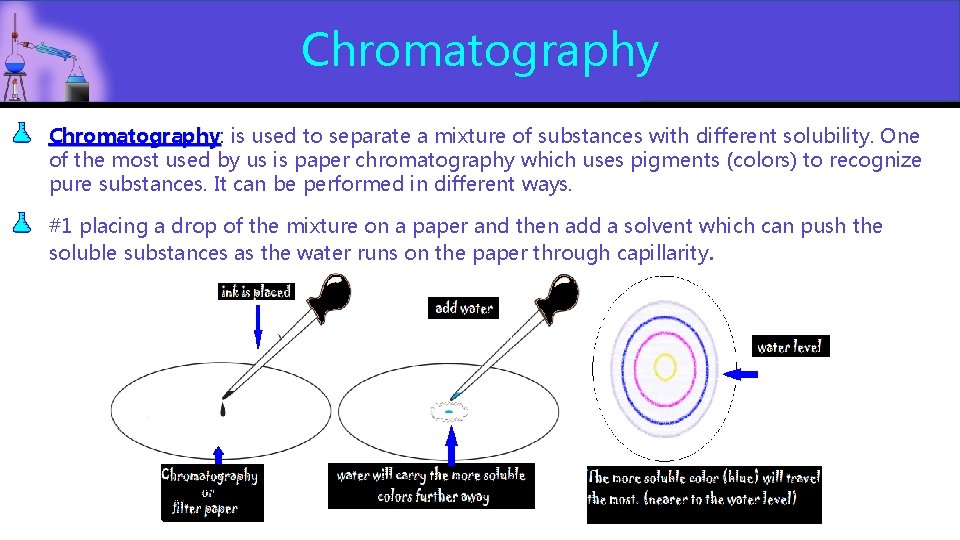
Chromatography: is used to separate a mixture of substances with different solubility. One Chromatography of the most used by us is paper chromatography which uses pigments (colors) to recognize pure substances. It can be performed in different ways. #1 placing a drop of the mixture on a paper and then add a solvent which can push the soluble substances as the water runs on the paper through capillarity.

Chromatography: In the lab we will perform the paper Chromatography chromatography as follows.

Facts about Paper Chromatography The paper is called the Static Phase The solvent (water in this case)is called the Moving Phase Substances that run the same distance correspond to the same compound. In the example, we can say that the sample contains substances w, x and z but it does not contain y The graph with the information is called a chromatogram

Retardation Factor (Rf) Retardation factor Is the ratio between the distance covered by one pure substance and the distance covered by the solvent Retardation factor is an intensive property. We can use it to identify substances. Rf red = dr/dw = around 0. 5 (no units) Rf yellow = dy/dw= around 0. 9 Rf blue = db/dw = around 0. 3 Yellow is most soluble blue is the least soluble

Possible values of Rf If the pigment travels with the water front, the Rf value will be equals to 1, because both distances are the same. If the pigment does not move at all, the pigment distance will be zero, so the Rf value will be zero. Those are the maximum and minimum values posibles of Rf. Rf does not have any unit. It is only a number.

Separating aminoacids Paper chromatography is also used to separate aminoacids, which are colorless. After the separation, Ninhydrin, a Locatin Agent must be applied to be able to “see” the distance covered by each one.

Exercise
- Slides: 42