Properties of Matter Matter always has mass and









- Slides: 10

Properties of Matter

• Matter always has mass and volume. • Mass and volume are properties of matter. These are specific aspects that matter has. Types of properties • Mass and volume are non-characteristic properties because they do not provide enough information to identify an unknown substance. • For example, knowing a person has brown hair is not enough information to identify them, because many people have brown hair. • Colour is therefore non-characteristic as well.

Temperature • Temperature is a measure of the energy level of particles. • Determining the temperature of a substance tells us how hot or cold that substance is. • The hotter a substance is, the faster its particles are moving, and the higher its temperature. • We measure temperature with thermometers, and the unit of measurement is °C (degrees Celsius).

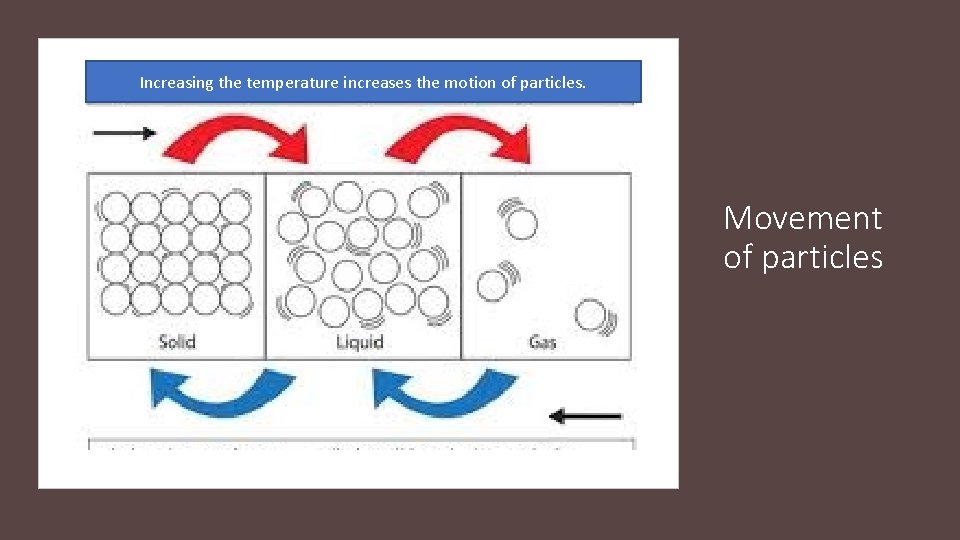
Increasing the temperature increases the motion of particles. Movement of particles

Thermometers

How to measure temperature • http: //www. alloprof. qc. ca/BV/Pages/s 1503. asp x

Melting Point • The temperature where a solid transforms into a liquid is known as the substance’s melting point. • Ice melts into water at 0°C, so its melting point is 0°C. • Solid gold melts at 1 064°C, so gold’s melting point is 1 064°C. • Melting point is a characteristic property because it specifically allows us to identify a substance.

Boiling point • When a liquid transforms into a gas, this is called vaporization. If this happens rapidly, we call it boiling. • The temperature at which a liquid turns into a gas is known as its boiling point. • Water turns into water vapour at 100°C, so water’s boiling point is 100°C. • Liquid gold transforms into a gas at 2 856°C, so gold’s boiling point is 2 856°C. • Boiling point is also a characteristic property.

Characteristic Properties • Melting point is a characteristic property, because only gold turns from a solid to a liquid at 1064°C. This temperature is unique to gold. • Boiling point is a characteristic property because only gold turns from a liquid to a gas at 2 856°C. This temperature is unique to gold. • Characteristic properties help us identify unknown substances. If you are presented with an unknown substance that melts at 1064°C, you know it must be gold.

Summary • https: //youtu. be/G 2 x. PR 3 g. UFpw