Properties of Light Electromagenetic Spectrum Electromagnetic Spectrum the











































- Slides: 54

Properties of Light Electromagenetic Spectrum

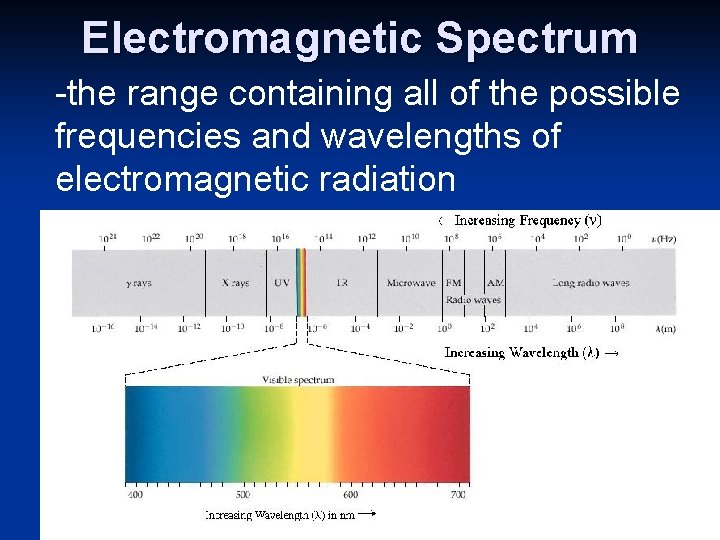
Electromagnetic Spectrum -the range containing all of the possible frequencies and wavelengths of electromagnetic radiation Types of electromagne tic radiation

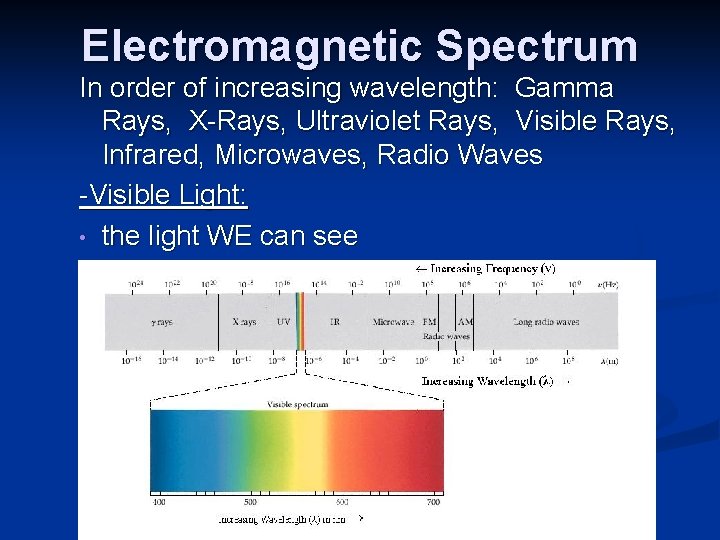
Electromagnetic Spectrum In order of increasing wavelength: Gamma Rays, X-Rays, Ultraviolet Rays, Visible Rays, Infrared, Microwaves, Radio Waves -Visible Light: • the light WE can see

Properties of Light -Wave description of Light - light exhibits wavelike behavior as it travels through space. - - - Wavelength (λ): distance between two peaks; unit of measure – meter, cm, nanometer Frequency (f): number of waves that pass a given point in a specific time; unit of measure – Hertz (Hz) Speed of light (c ): c= λf c=3. 00 X 108 m/s

Properties of Light - Particle description of light: - light can be thought of as being composed of particles (Planck) - Photon: particle of light. Has zero mass and a quantum of energy (Einstein) - Quantum: minimum quantity of energy that can be lost or gained by an atom

Photoelectric effect - Photoelectric effect- refers to the emission of electrons from a metal when a light of sufficient energy shines on the metal - Wave theory of light couldn’t explain this effect

E = hf E - energy in Joules of a quantum of energy h - Planck’s constant = 6. 626 x 10 -34 J·s f - frequency

Problems 1. Calculate the wavelength if the frequency of radiation is 5. 10 x 1014 Hz.

Problems 2. Find the energy in a quantum of light that has a frequency of 1. 7 x 1014 Hz

Problems 3. Find the wavelength of a photon of light with an energy of 3. 26 x 10 -19 J.

Violet Indigo Blue Green Yellow Orange Red 400 -424 nm 425 -454 nm 455 -489 nm 490 -574 nm 575 -579 nm 580 -649 nm 650 -700 nm Find the color of a photon of light if it is known to have an energy of 4. 75 x 10 -19 J.

Properties of Light Therefore, light has dual wave- particle properties - It behaves like a wave AND a particle

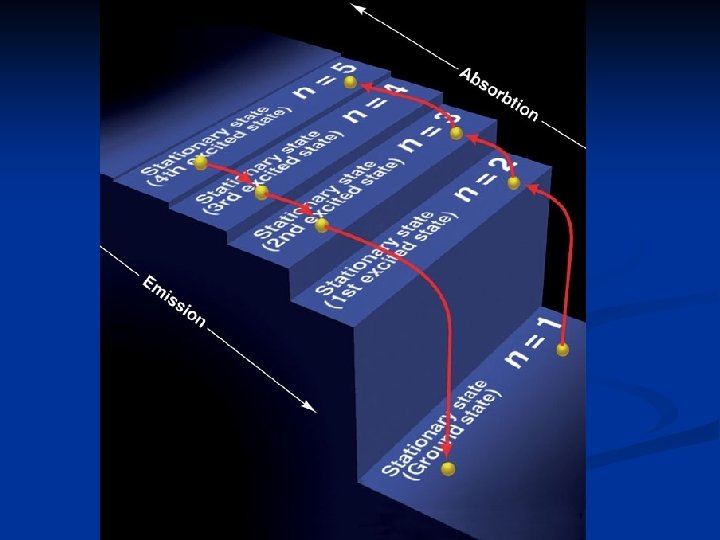
H atom Line-Emission Spectra When you pass a current through a gas at low pressure, the energy of some of the gas atoms increases. n Ground State - lowest energy state for an electron. n Excited state - An electron that has a higher potential energy n When an electron drops down from an excited state to its ground state, light of a certain wavelength is emitted n

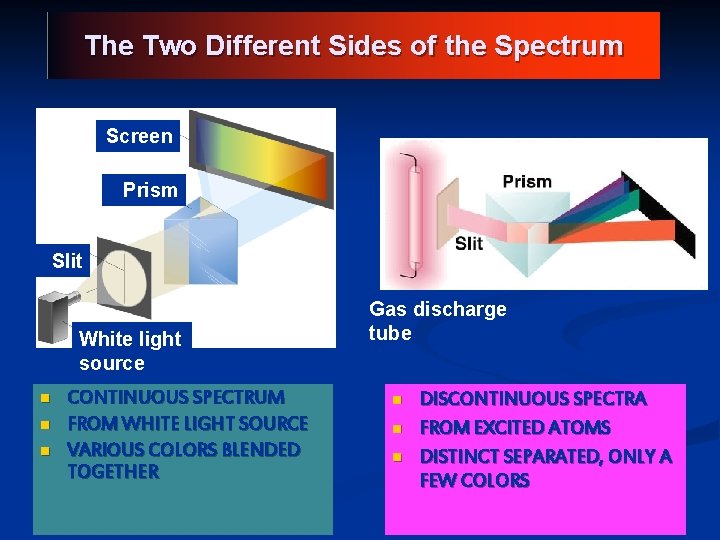
The Two Different Sides of the Spectrum Screen Prism Slit White light source n n n CONTINUOUS SPECTRUM FROM WHITE LIGHT SOURCE VARIOUS COLORS BLENDED TOGETHER Gas discharge tube n n n DISCONTINUOUS SPECTRA FROM EXCITED ATOMS DISTINCT SEPARATED, ONLY A FEW COLORS

2

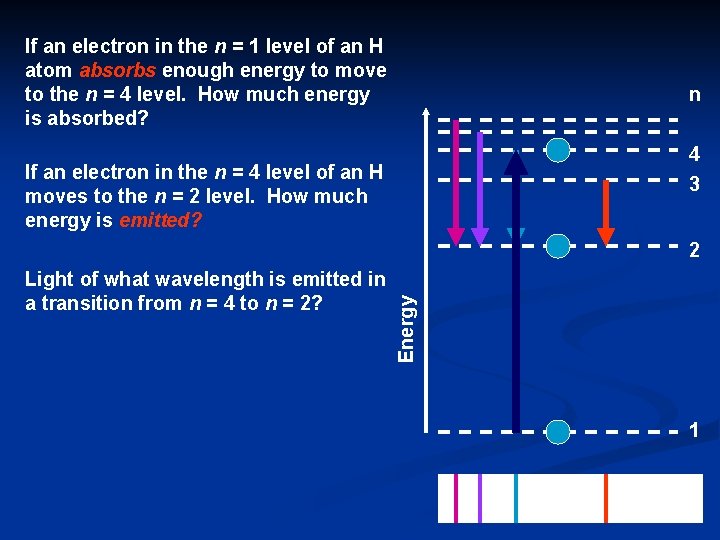
If an electron in the n = 1 level of an H atom absorbs enough energy to move to the n = 4 level. How much energy is absorbed? n 4 3 If an electron in the n = 4 level of an H moves to the n = 2 level. How much energy is emitted? Light of what wavelength is emitted in a transition from n = 4 to n = 2? Energy 2 1

“Neon Lights”

Electron -Is a particle & also has wave properties -Bohr’s Model of Atom: -Electrons circle around nucleus in allowed paths (orbits) -Electrons placed in different orbits based on energy levels of the electron -One electron can not be in two orbits at once and can not be between orbits *** Bohr’s model still has shortcomings… we will discuss later)

Take Home Message -Science is CONSTANTLY changing -New research may disprove old findings -New models and thoughts may be created as a result

Review 1) Name the two properties that light has. 2) What is the type of light that we can see called? 3) Visible light is found on the _____ spectrum. 4) A particle of light that has zero mass and a quantum of energy is called a ____. 5) The distance between two peaks is called_____ 6) Name the model of an atom that said electrons circle around nucleus in allowed paths based on energy level. 7) The number of waves that pass a given point in a specific time is called ____ 8) The minimum quantity of energy that can be lost or gained by an atom is a______. 9) The _______ refers to the emission of electrons from a metal when a light of sufficient energy shines on the metal

Quantum Numbers

Review 1) Name the two properties that light has. 2) A particle of light that has zero mass and a quantum of energy is called a ____. 3) The minimum quantity of energy that can be lost or gained by an atom is a______. 4) Name the model of an atom that said electrons circle around nucleus in allowed paths based on energy level.

Electron -Is a particle & also has wave properties -Bohr’s Model of Atom: -Electrons circle around nucleus in allowed paths (orbits) -Electrons placed in different orbits based on energy levels of the electron -One electron can not be in two orbits at once and can not be between orbits *** Bohr’s model still has shortcomings… we will discuss later)

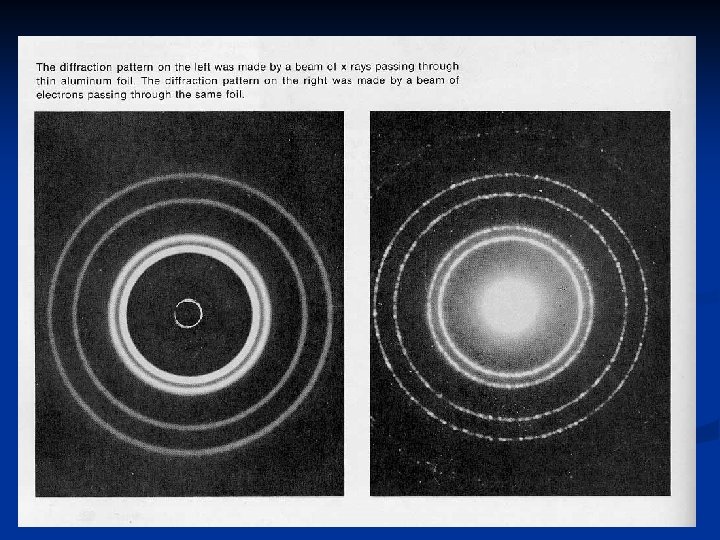
Electrons as Waves Early 20 th century: -De Broglie: believed electrons have dual wave-particle properties -Scientists demonstrated that electrons could be bent or diffracted -Diffraction: bending of a wave as it passes by the edge of an object


Electron “Detection” - - - Electrons are detected with interaction with photons Photons can knock an electron off its course Heisenberg Uncertainty Principle: - It is impossible to determine simultaneously both the position and velocity of an electron or any other particle

Quantum Model - Schrödinger: wave equation - Quantum Theory: - - Electrons can exist in atoms only at specific energy levels Wave equations only tell us probability of finding electron in certain position Currently it is believed: - Electrons do not travel in orbits, but orbitals Orbital: 3 -D region around nucleus. Indicates probable location of finding an electron

Quantum numbers - Quantum numbers: specify the properties of atomic orbitals and the electrons in orbitals - Four different Quantum Numbers - Principal Quantum Number Angular Momentum Quantum Number Magnetic Quantum Number Spin Quantum Number

Principal Quantum Number - Symbol: n - Main energy level occupied by the electron - Whole numbers (1, 2, 3, …) - n=1 : Lowest energy level (closest to nucleus) - Gives an indication of the size of the orbital

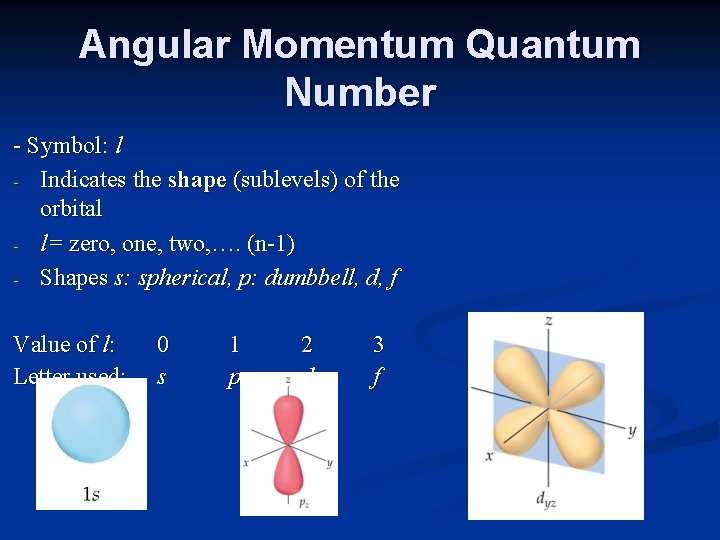
Angular Momentum Quantum Number - Symbol: l - Indicates the shape (sublevels) of the orbital - l= zero, one, two, …. (n-1) - Shapes s: spherical, p: dumbbell, d, f Value of l: Letter used: 0 s 1 p 2 d 3 f

Magnetic Quantum Number - Symbol: m - Indicates the orientation of an orbital in space - s orbital has 1 orientation, p orbital has 3, d orbital has 5, f has 7


Spin Quantum Number - Indicates the spin of an electron in an orbital - A single orbital can hold a maximum of 2 electrons with OPPOSITE spins - Practice: - How many orbitals are in n=3? How many electrons are in n=3?

Electron Configuration

Objectives -List the total number of electrons needed to fully occupy each main energy level -State the Aufbau principle, Pauli exclusion principle, and Hund’s Rule -Draw the electron configuration and orbital notation to show the location of electrons in an atom.

Electron Configuration - Electron configuration: the arrangement of electrons in an atom - Rules for electron filling of orbitals: - - - Aufbau Principle: e- will occupy the lowest energy level orbital available Pauli Exclusion Principle: no two e- in the same orbital can have the same spin Hund’s Rule: orbitals of equal energy will fill singularly before a second e- can occupy the same orbital

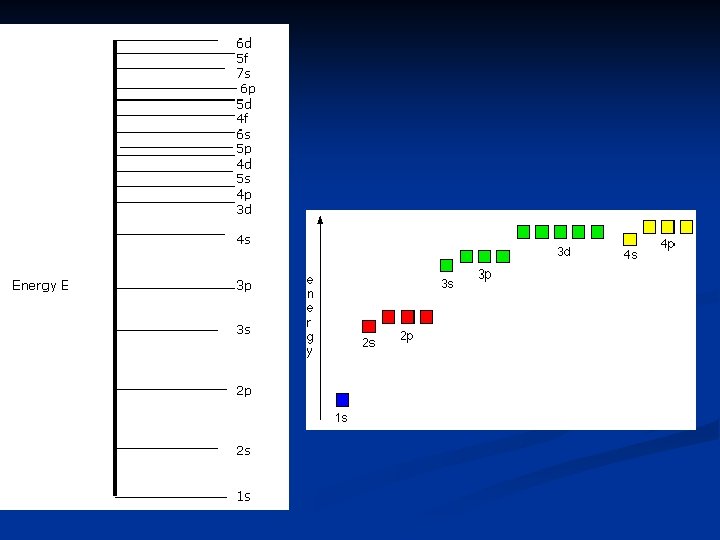
The Order of Electron Filling 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 3 p 2 p 7 d 6 d 5 d 4 d 3 d 7 f 6 f 5 f 4 f Remember the Aufbau Principle


The Order of Electron Filling 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 3 p 2 p 7 d 6 d 5 d 4 d 3 d 7 f 6 f 5 f 4 f Remember the Aufbau Principle

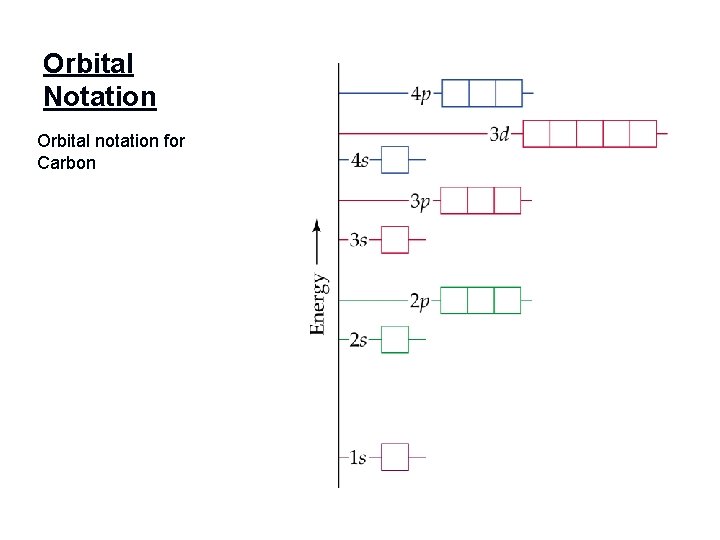
Electron Configuration - - Electron configuration shows the energy level number, the orbital sublevel, and the number of electrons in each orbital # of e- are represented by a superscript e- config for carbon - - 1 s 22 p 2 Write the e-config for fluorine. Write the e-config for silver.

Orbital Notation Orbital notation for Carbon

Orbital Notation - Orbital notation shows e- in their orbitals - First write the electron configuration _____ This is the orbital notation for Lithium 1 s 2 s Remember the Pauli Exclusion Principle & Hund’s Rule

Orbital Notation -Draw the orbital notation for fluorine -Draw the orbital notation for nitrogen

Review - - - What rule says orbitals of equal energy will fill singly before a second e- can occupy the same orbital What rule says that e- will occupy the lowest energy level orbital available What rule says that no two e- in the same orbital can have the same spin

Chapter 4 -3

The Order of Electron Filling 7 s 6 s 5 s 4 s 3 s 2 s 1 s 7 p 6 p 5 p 4 p 3 p 2 p 7 d 6 d 5 d 4 d 3 d 7 f 6 f 5 f 4 f Remember the Aufbau Principle

Objectives - - - Write the noble-gas notation of a given element Determine the element given the electron configuration or orbital notation. Determine the highest energy level that is occupied and the last orbital that is filled of a given element.

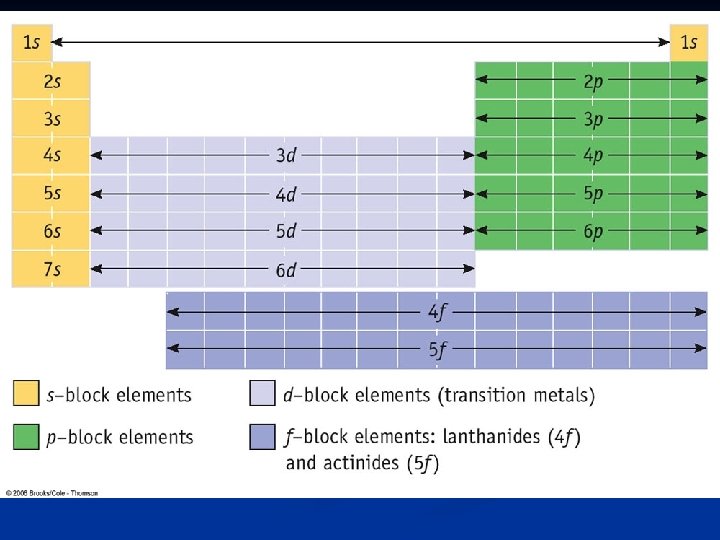
Noble Gases - Gases like helium, neon, and argon Where are these on the periodic table? - - - Group 18 The outer shell (main energy level) is completely filled with 8 electrons We can use their electron configuration to help abbreviate the e- config. of other elements

Noble Gas Configuration - The electron configuration of Manganese is: - - - Manganese is in period 4…. Look at the noble gas in the previous period (period 3) --- Argon’s e- config. is: - - 1 s 22 p 63 s 23 p 64 s 23 d 5 1 s 22 p 63 s 23 p 6 We can shorten the e- config of Mn by writing:

Practice - Write the Noble Gas Configuration of Na, Sr, and P

Short cuts with the Periodic Table n Look at the period number, group number, and block


Identify the following elements A) B) C) D) 1 s 22 p 63 s 23 p 3 [Ar]4 s 1 1 s 22 p 1 [Ne]3 s 2

Review - What is the last orbital occupied by an electron in the elements: - - He Al What is the highest energy level occupied by the following elements: - Ca Be