Properties of Gases Common Gases Of the elements























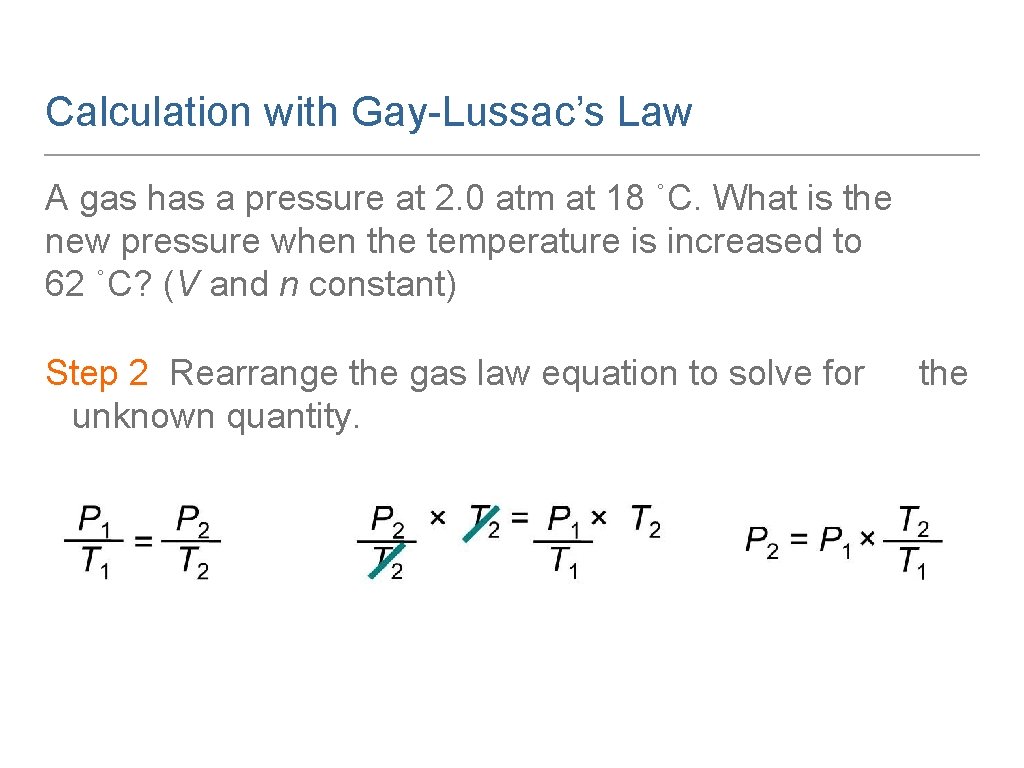
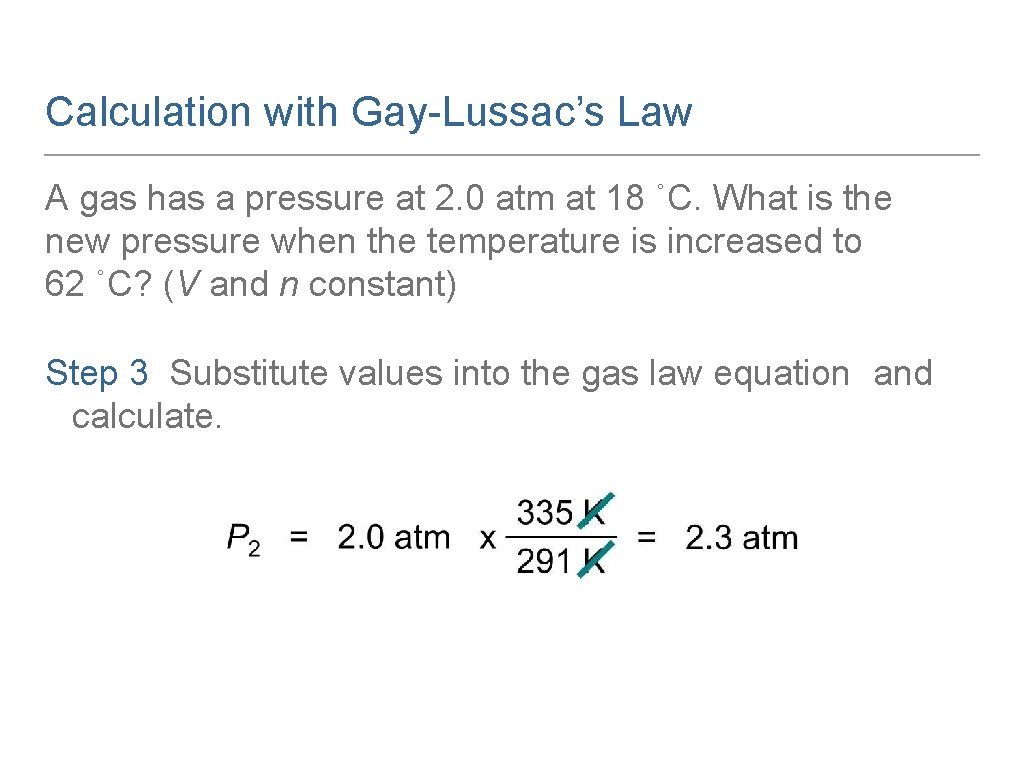






















































- Slides: 95

Properties of Gases

Common Gases Of the elements on the periodic table, some exist as a gas at room temperature, these include ▪ the Noble Gases, Group 8 A (18), ▪ H 2, N 2, O 2, F 2, Cl 2, and ▪ many oxides of nonmetals such as CO, CO 2, NO 2, SO 2, and SO 3.

Gases and Environmental Concerns Some gases are responsible for environmental and health concerns including ▪ methane, CH 4, ▪ chlorofluorocarbons (CFCs), ▪ nitrogen oxides found in smog, and ▪ volatile organic compounds (VOCs), such as compounds found in paint thinners.

Kinetic Theory of Gases A gas consists of small particles that ▪ move randomly with high velocities. ▪ have essentially no attractive (or repulsive) forces toward each other. ▪ have a very small volume compared to the volume of the containers they occupy. ▪ are in constant motion. ▪ have kinetic energies that increase with an increase in temperature.

Properties of Gases are described in terms of four properties: 1. pressure (P), 3. temperature (T), and 2. volume (V), 4. amount (n).

Learning Check Identify the property of a gas (temperature, volume, or pressure) that is described by each of the following: A. increases the kinetic energy of gas particles B. the force of the gas particles hitting the walls of the container C. the space that is occupied by a gas

Solution Identify the property of a gas that is described by each of the following: A. increases the kinetic energy of gas particles temperature B. the force of the gas particles hitting the walls of the container pressure C. the space that is occupied by a gas volume

Gas Pressure Gas pressure ▪ is the force acting on a specific area. ▪ has units of atm, mm. Hg, torr, lb/in. 2, and kilopascals(k. Pa). 1 atm = 760 mm. Hg (exact) 1 atm = 760 torr (exact) 1 atm = 14. 7 lb/in. 2 1 atm = 101. 325 k. Pa

Learning Check 1. What is 475 mm. Hg expressed in atmospheres (atm)? A. 475 atm B. 0. 625 atm C. 3. 61 x 105 atm 2. The pressure in a tire is 2. 00 atm. What is this pressure in mm. Hg? A. 2. 00 mm. Hg B. 1520 mm. Hg C. 22 300 mm. Hg

Solution 1. What is 475 mm. Hg expressed in atmospheres (atm)? B. 0. 625 atm 2. The pressure of a tire is measured as 2. 00 atm. What is this pressure in mm. Hg? B. 1520 mm. Hg

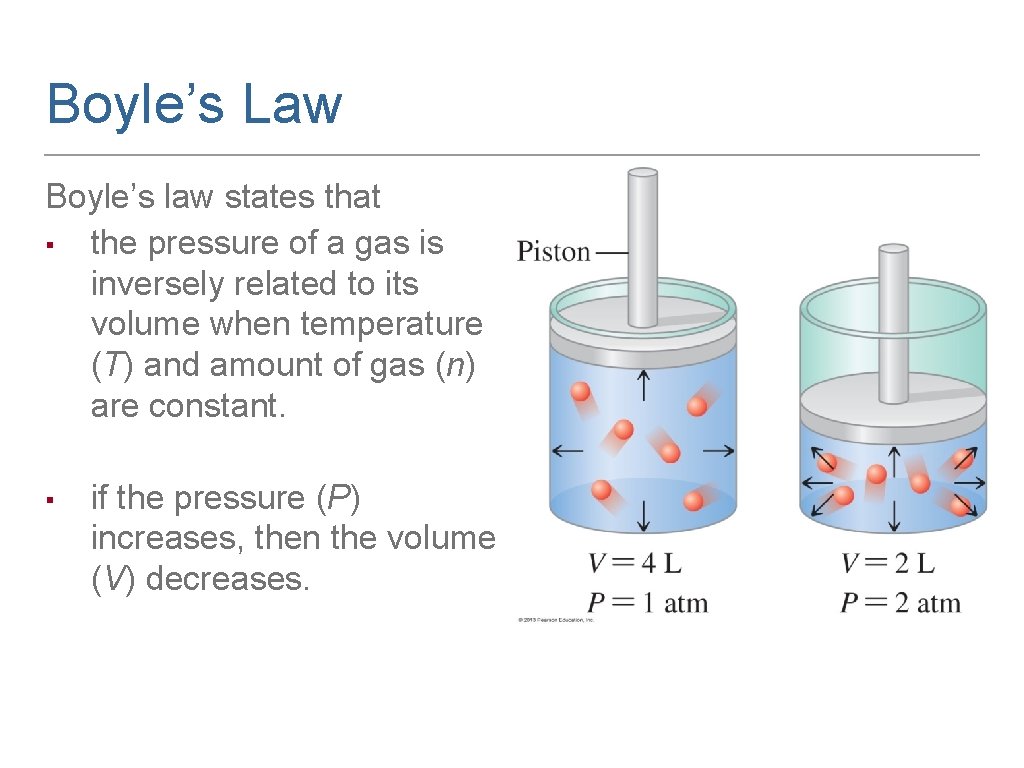
Boyle’s Law Boyle’s law states that ▪ the pressure of a gas is inversely related to its volume when temperature (T) and amount of gas (n) are constant. ▪ if the pressure (P) increases, then the volume (V) decreases.

PV Constant in Boyle’s Law In Boyle’s law, ▪ the product P × V is constant as long as T and n do not change. ▪ ▪ it can be stated that since P × V is a constant. P 1 V 1 = P 2 V 2 (T, n constant)

Guide to Using Gas Laws

Solving for a Gas Law Factor A sample of nitrogen gas (N 2) has a volume of 4. 2 L at 1. 0 atm. If the pressure is decreased to 0. 75 atm with no change in the temperature or amount of gas, what will be the new volume? Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 V 1 = 4. 2 L V 2 = ? P 1 = 1. 0 atm P 2 = 0. 75 atm Know Predict V increases P decreases Factors T and n remain constant.

Solving for a Gas Law Factor A sample of nitrogen gas (N 2) has a volume of 4. 2 L at 1. 0 atm. If the pressure is decreased to 0. 75 atm with no change in the temperature or amount of gas, what will be the new volume? Step 2 Rearrange the gas law equation to solve for the unknown quantity. P 1 V 1 = P 2 V 2 Boyle’s Law To solve for V 2 , divide both sides by P 2.

Solving for a Gas Law Factor A sample of nitrogen gas (N 2) has a volume of 4. 2 L at 1. 0 atm. If the pressure is decreased to 0. 75 atm with no change in the temperature or amount of gas, what will be the new volume? Step 3 Substitute values into the gas law equation and calculate.

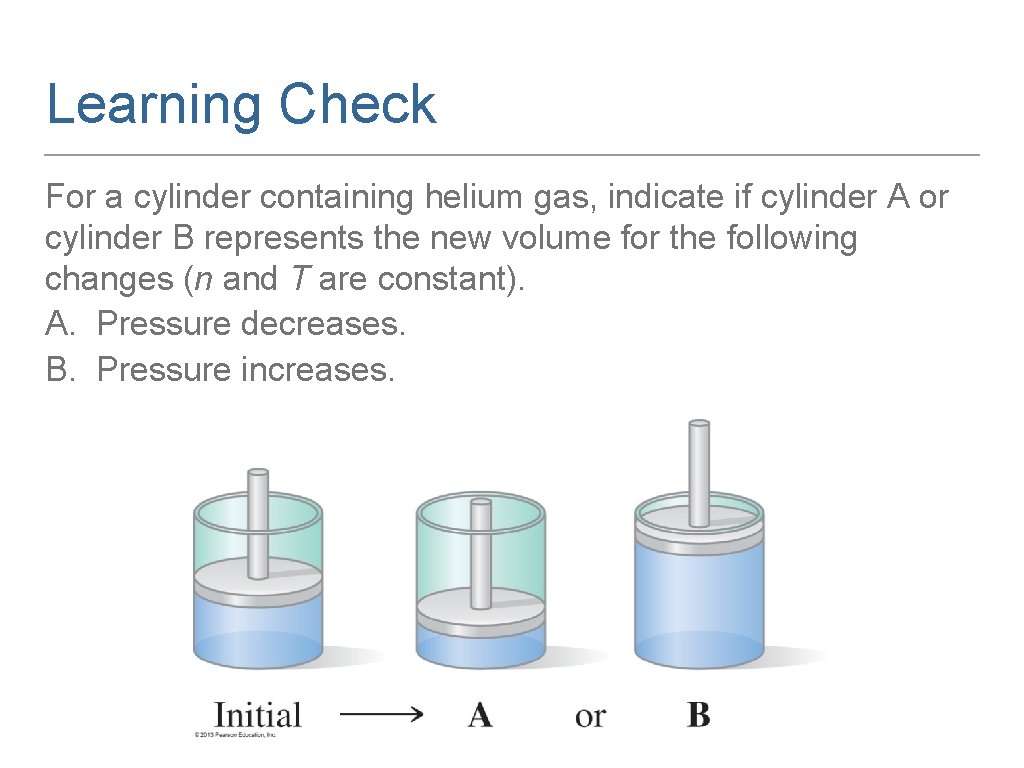
Learning Check For a cylinder containing helium gas, indicate if cylinder A or cylinder B represents the new volume for the following changes (n and T are constant). A. Pressure decreases. B. Pressure increases.

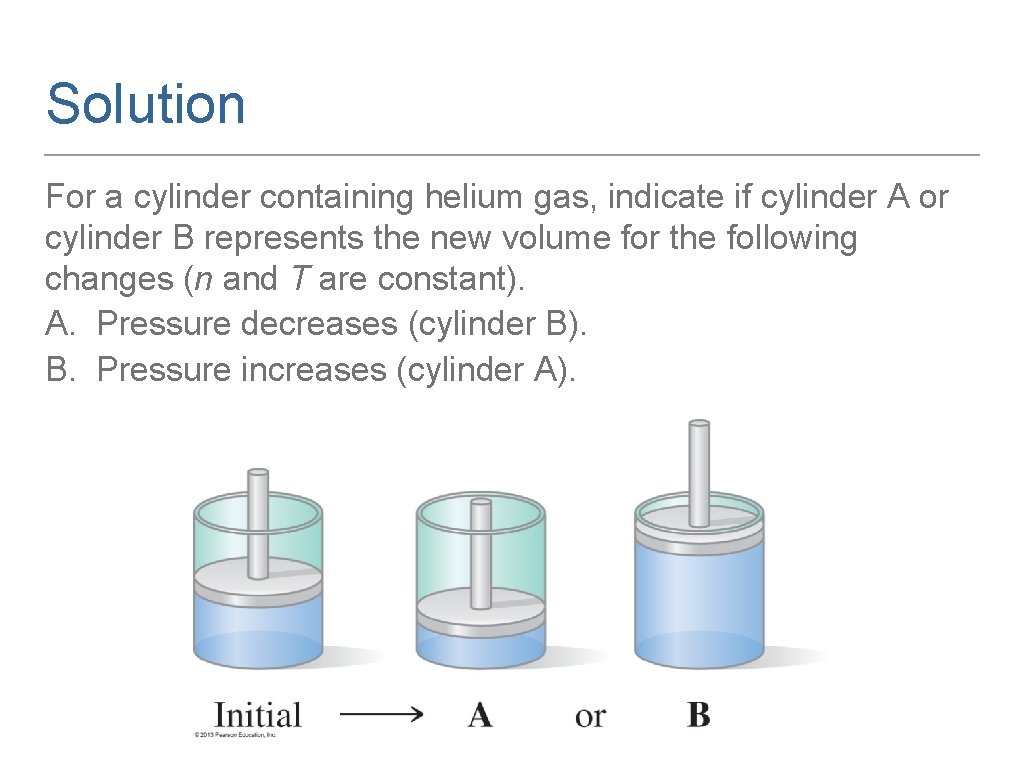
Solution For a cylinder containing helium gas, indicate if cylinder A or cylinder B represents the new volume for the following changes (n and T are constant). A. Pressure decreases (cylinder B). B. Pressure increases (cylinder A).

Charles’s Law Charles’s law states that, ▪ the Kelvin temperature of a gas is directly related to the volume of the gas, ▪ P and n are constant, and ▪ as the temperature of a gas increases, the molecules move faster and its volume increases to maintain constant P.

Charles’s Law: V and T Charles’s law states that when the temperature of a gas increases, making the molecules move faster, the volume of the gas must increase to maintain constant pressure.

Charles’s Law: V and T A sample of neon gas has a volume of 3. 1 L and a temperature of 12 ˚C. Find the new volume (L) of the gas if the temperature rises to 32 ˚C. Assume the pressure and amount of gas remain constant. Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 Know T 1 = 12 °C + 273 = 285 T 1 = 32 °C+ 273 = 305 K T increases V 1 = 3. 1 L V 1 = ? Predict V increases

Charles’s Law: V and T A sample of neon gas has a volume of 3. 1 L and a temperature of 12 ˚C. Find the new volume (L) of the gas if the temperature rises to 32 ˚C. Assume the pressure and amount of gas remain constant. Step 2 Rearrange the gas law equation to solve for the unknown quantity.

Charles’s Law: V and T A sample of neon gas has a volume of 3. 1 L and a temperature of 12 ˚C. Find the new volume (L) of the gas if the temperature rises to 32 ˚C. Assume the pressure and amount of gas remain constant. Step 3 Substitute values into the gas law equation and the table.

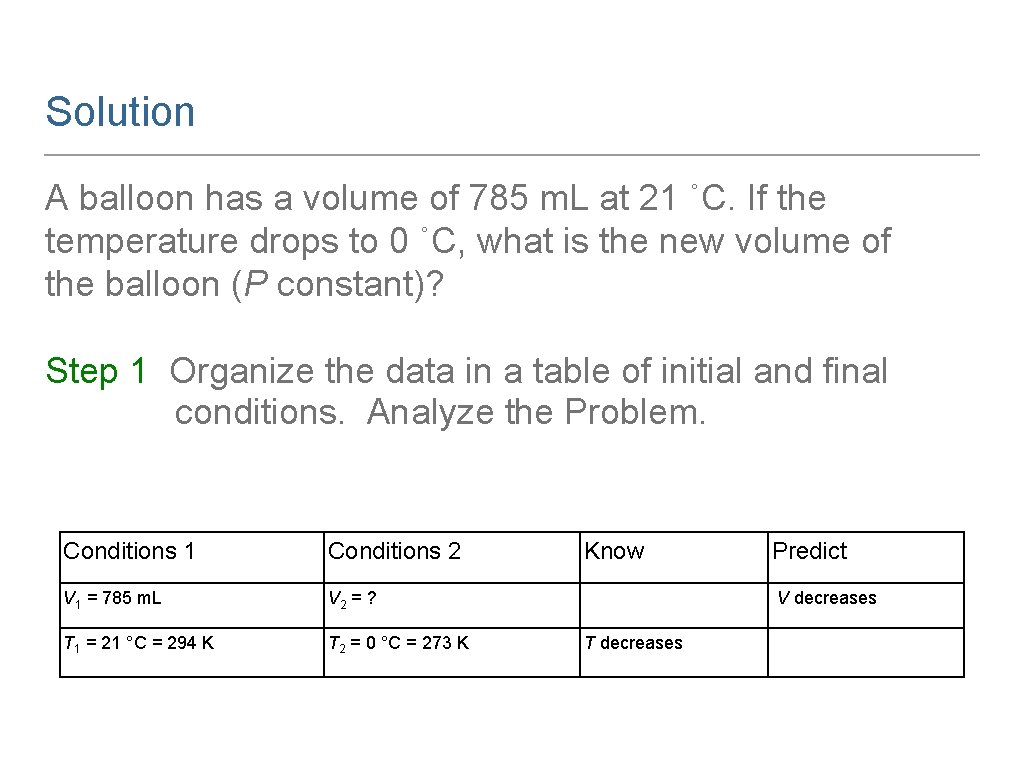
Learning Check A balloon has a volume of 785 m. L at 21 ˚C. If the temperature drops to 0 ˚C, what is the new volume of the balloon (P constant)?

Solution A balloon has a volume of 785 m. L at 21 ˚C. If the temperature drops to 0 ˚C, what is the new volume of the balloon (P constant)? Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 V 1 = 785 m. L V 2 = ? T 1 = 21 °C = 294 K T 2 = 0 °C = 273 K Know Predict V decreases T decreases

Solution A balloon has a volume of 785 m. L at 21 ˚C. If the temperature drops to 0 ˚C, what is the new volume of the balloon (P constant)? Step 2 Rearrange the gas law equation to solve for the unknown quantity. Step 3 Substitute values into the gas law equation and the table.

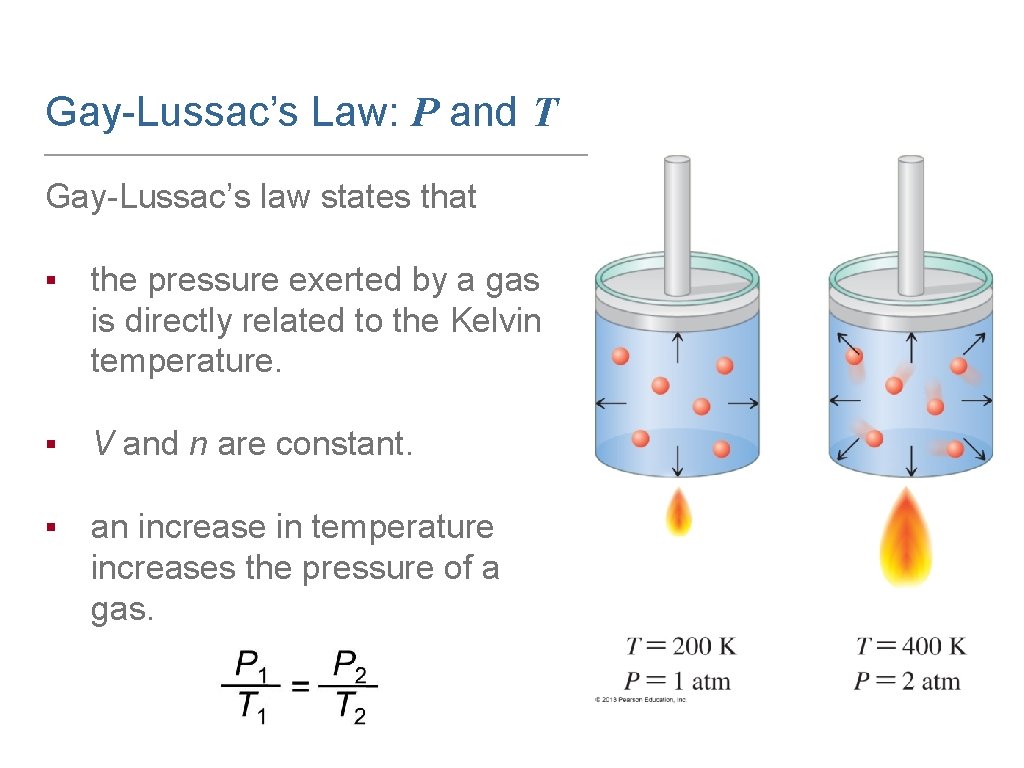
Gay-Lussac’s Law: P and T Gay-Lussac’s law states that ▪ the pressure exerted by a gas is directly related to the Kelvin temperature. ▪ V and n are constant. ▪ an increase in temperature increases the pressure of a gas.

Calculation with Gay-Lussac’s Law A gas has a pressure at 2. 0 atm at 18 ˚C. What is the new pressure when the temperature is increased to 62 ˚C? (V and n constant) Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 P 1 = 2. 0 atm P 2 = ? T 1 = 18 °C + 273 = 291 K T 2 = 62 °C + 273 = 335 K Know Predict P increase T increase
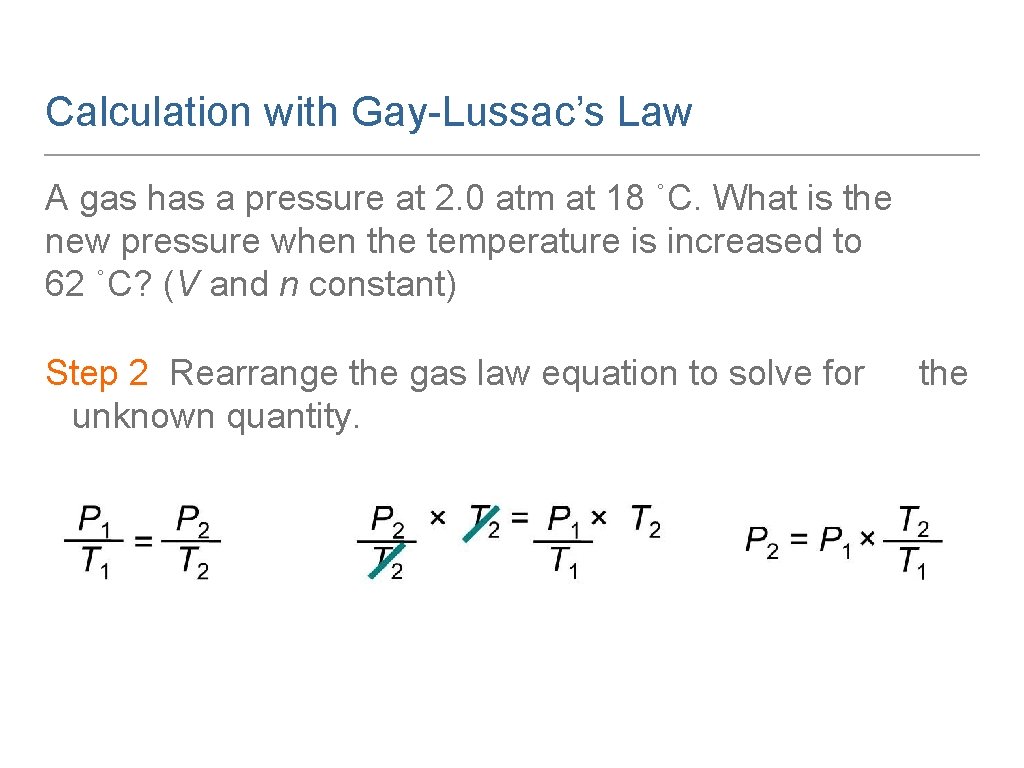
Calculation with Gay-Lussac’s Law A gas has a pressure at 2. 0 atm at 18 ˚C. What is the new pressure when the temperature is increased to 62 ˚C? (V and n constant) Step 2 Rearrange the gas law equation to solve for unknown quantity. the
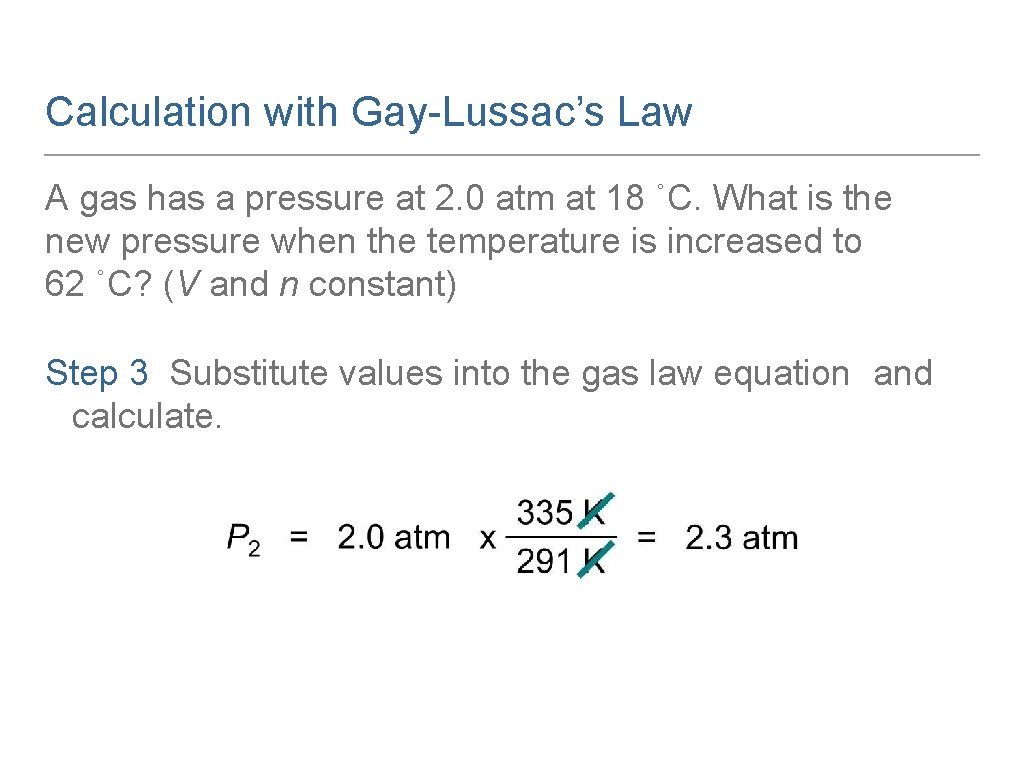
Calculation with Gay-Lussac’s Law A gas has a pressure at 2. 0 atm at 18 ˚C. What is the new pressure when the temperature is increased to 62 ˚C? (V and n constant) Step 3 Substitute values into the gas law equation and calculate.

Learning Check Solve for the final pressure of a gas with an initial pressure of 1. 20 atm at 75 ˚C when cooled to − 22 ˚C. (V and n constant)

Solution Solve for the final pressure of a gas with an initial pressure of 1. 20 atm at 75 ˚C when cooled to − 22 ˚C. Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 P 1 = 1. 20 atm P 2 = ? T 1 = 75 °C + 273 = 348 K T 2 = − 22 °C + 273 = 251 K Know Predict P decrease T decrease

Solution Solve for the final pressure of a gas with an initial pressure of 1. 20 atm at 75 ˚C when cooled to − 22 ˚C. Step 2 Rearrange the gas law equation to solve for the unknown quantity.

Solution Solve for the final pressure of a gas with an initial pressure of 1. 20 atm at 75 ˚C when cooled to − 22 ˚C. Step 3 Substitute values into the gas law equation and calculate.

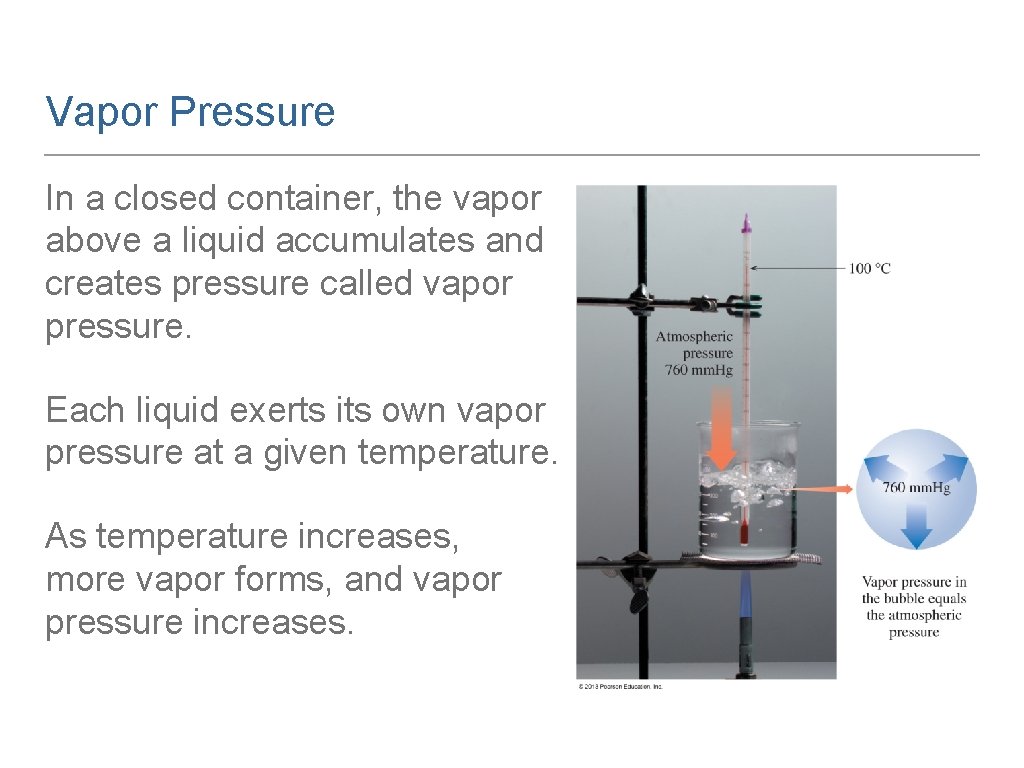
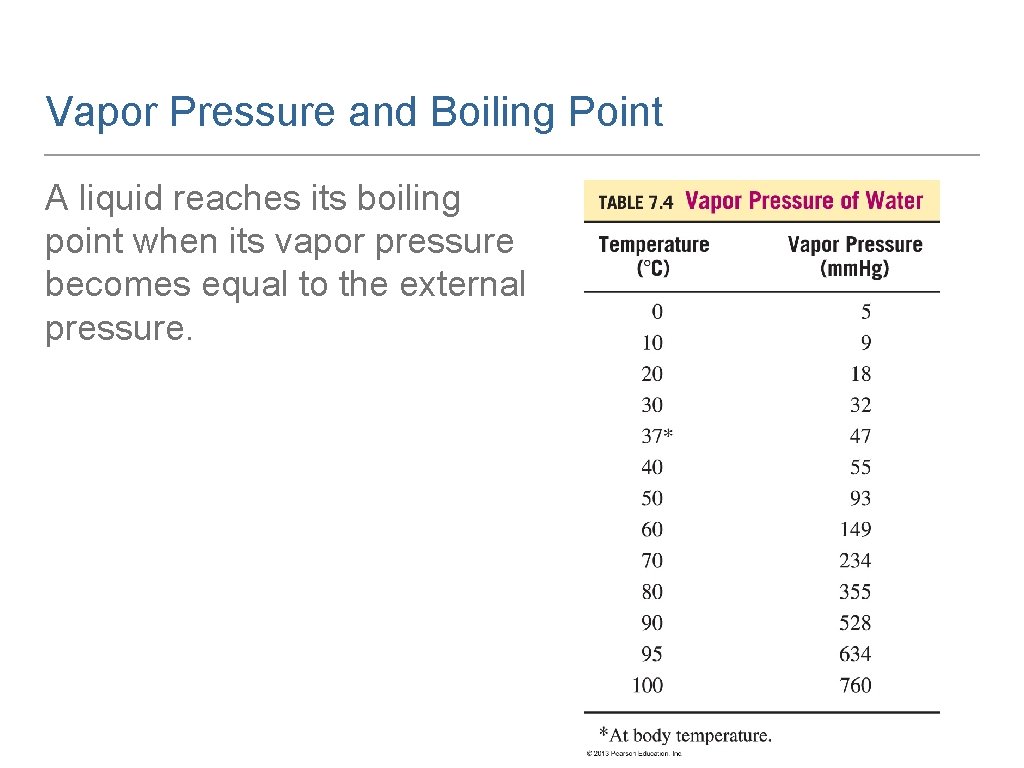
Vapor Pressure In a closed container, the vapor above a liquid accumulates and creates pressure called vapor pressure. Each liquid exerts its own vapor pressure at a given temperature. As temperature increases, more vapor forms, and vapor pressure increases.

Vapor Pressure and Boiling Point A liquid reaches its boiling point when its vapor pressure becomes equal to the external pressure.

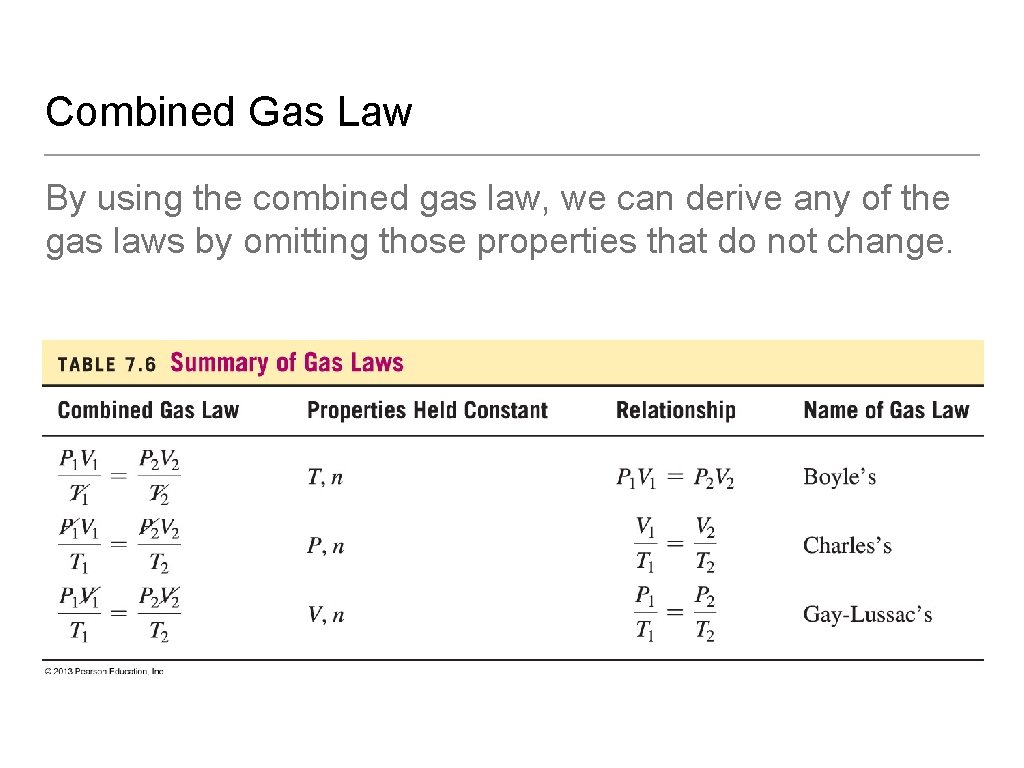
Combined Gas Law The combined gas law uses Boyle’s Law, Charles’s Law, and Gay-Lussac’s Law (n is constant).

Combined Gas Law By using the combined gas law, we can derive any of the gas laws by omitting those properties that do not change.

Using the Combined Gas Law A 0. 180 L sample of gas has a pressure of 0. 800 atm at 29 °C. At what temperature (°C) will the gas have a volume of 90. 0 m. L and a pressure of 3. 20 atm (n constant)? Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 P 1 = 0. 800 atm P 2 = 3. 20 atm V 1 = 0. 180 L V 2 = 90. 0 m. L= 0. 0900 L T 1 = 29 °C + 273 = 302 K T 2 = ?

Using the Combined Gas Law A 0. 180 L sample of gas has a pressure of 0. 800 atm at 29 °C. At what temperature (°C) will the gas have a volume of 90. 0 m. L and a pressure of 3. 20 atm (n constant)? Step 2 Rearrange the gas law equation to solve for the unknown quantity.

Using the Combined Gas Law A 0. 180 L sample of gas has a pressure of 0. 800 atm at 29 °C. At what temperature (°C) will the gas have a volume of 90. 0 m. L and a pressure of 3. 20 atm (n constant)? Step 3 Substitute values to solve for unknown.

Learning Check A gas has a volume of 675 m. L at 35 °C and 0. 850 atm pressure. What is the volume (m. L) of the gas at − 95 °C and a pressure of 802 mm. Hg (n constant)?

Solution A gas has a volume of 675 m. L at 35 °C and 0. 850 atm pressure. What is the volume (m. L) of the gas at − 95 °C and a pressure of 802 mm. Hg (n constant)? Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem.

Solution A gas has a volume of 675 m. L at 35 °C and 0. 850 atm pressure. What is the volume (m. L) of the gas at − 95 °C and a pressure of 802 mm. Hg (n constant)? Step 2 Rearrange the gas law equation to solve for the unknown quantity.

Solution A gas has a volume of 675 m. L at 35 °C and 0. 850 atm pressure. What is the volume (m. L) of the gas at − 95 °C and a pressure of 802 mm. Hg (n constant)? Step 3 Substitute values into the gas law equation and calculate.

Avogadro’s Law: Volume and Moles Avogadro’s law states that • the volume of a gas is directly related to the number of moles (n) of gas. • T and P are constant.

Learning Check If 0. 75 mole of helium gas occupies a volume of 1. 5 L, what volume will 1. 2 moles of helium occupy at the same temperature and pressure? A. 0. 94 L B. 1. 8 L C. 2. 4 L

Solution If 0. 75 mole of helium gas occupies a volume of 1. 5 L, what volume will 1. 2 moles of helium occupy at the same temperature and pressure? Step 1 Organize the data in a table of initial and final conditions. Analyze the Problem. Conditions 1 Conditions 2 V 1 = 1. 5 L V 2 = ? n 1 = 0. 75 mole n 2 = 1. 2 moles Know Predict V increases n increases

Solution If 0. 75 mole of helium gas occupies a volume of 1. 5 L, what volume will 1. 2 moles of helium occupy at the same temperature and pressure? Step 2 Rearrange the gas law equation to solve for the unknown quantity. Step 3 Substitute values into the gas law equation and calculate. Answer is C.

STP To make comparisons between different gases, we use arbitrary conditions called standard temperature (273 K) and standard pressure (1 atm). Standard temperature and pressure is abbreviated as STP. Standard temperature (T) = 0 °C or 273 K Standard pressure (P) = 1 atm (760 mm. Hg)

Molar Volume The molar volume of a gas measured at STP (standard temperature and pressure) is 22. 4 L for 1 mole of any gas. Avogadro’s law indicates that 1 mole of any gas at STP has a volume of 22. 4 L.

Molar Volume as a Conversion Factor The molar volume at STP • has about the same volume as 3 basketballs. • can be used to form 2 conversion factors. and

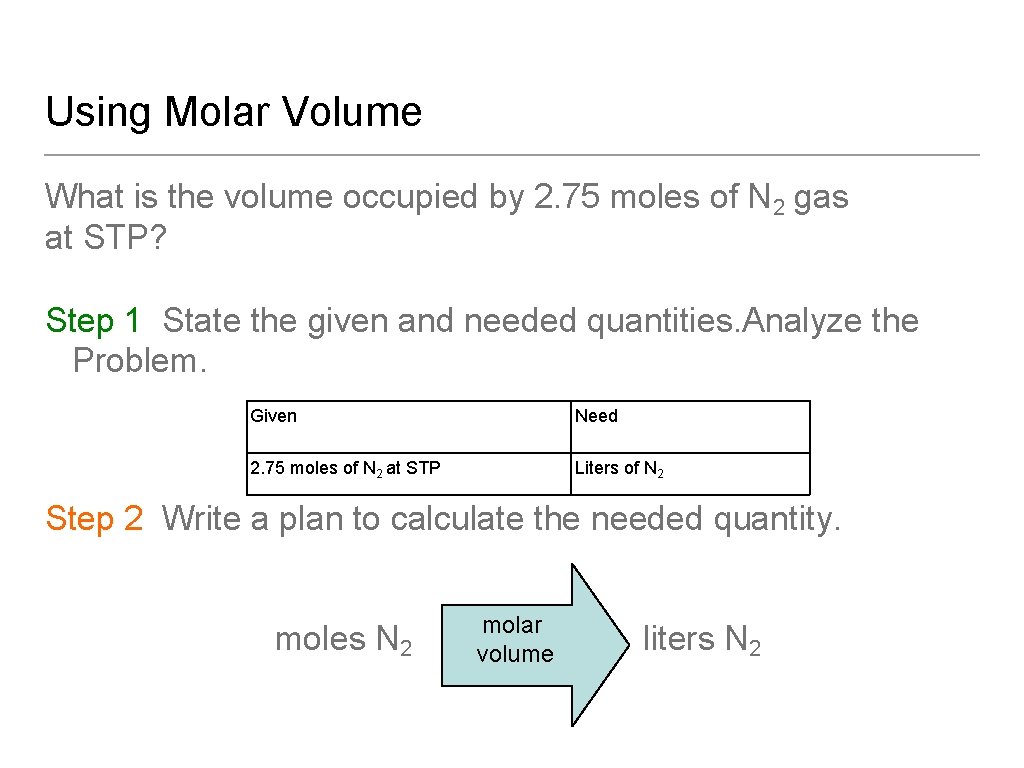
Using Molar Volume What is the volume occupied by 2. 75 moles of N 2 gas at STP? Step 1 State the given and needed quantities. Analyze the Problem. Given Need 2. 75 moles of N 2 at STP Liters of N 2 Step 2 Write a plan to calculate the needed quantity. moles N 2 molar volume liters N 2

Using Molar Volume What is the volume occupied by 2. 75 moles of N 2 gas at STP? Step 3 Write the equalities and conversion factors including 22. 4 L/mole at STP. Step 4 Set up the problem with factors to cancel units.

Learning Check • • What is the volume (L) at STP of 4. 00 g of CH 4? A. 5. 60 L B. 11. 2 L C. 44. 8 L

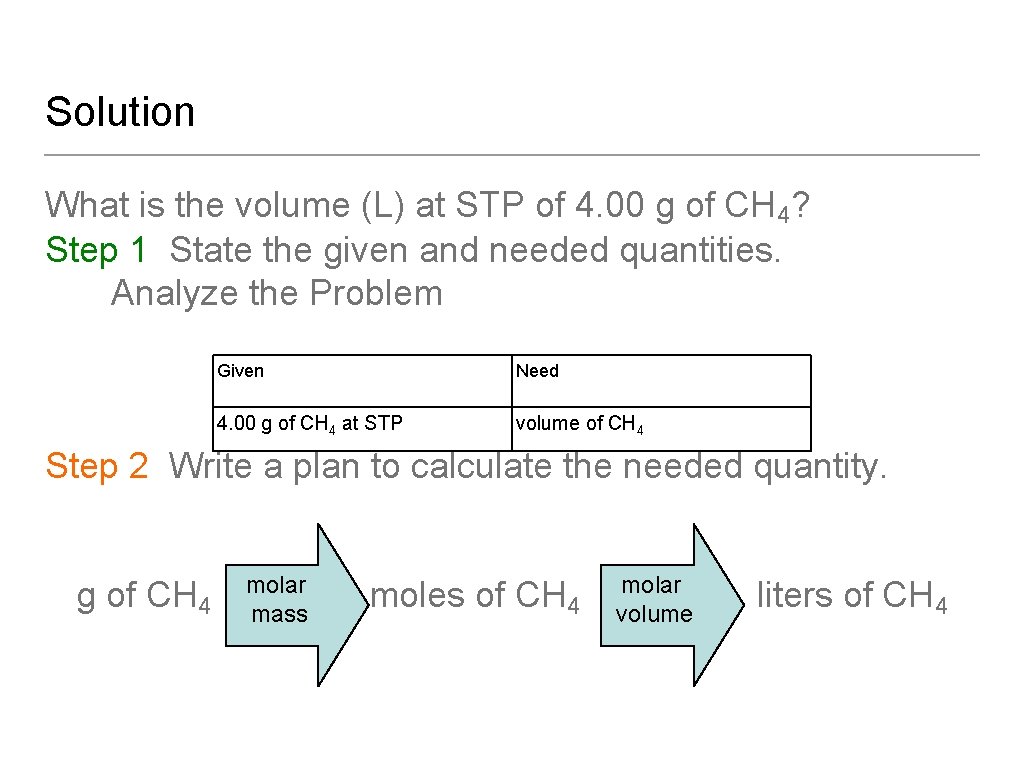
Solution What is the volume (L) at STP of 4. 00 g of CH 4? Step 1 State the given and needed quantities. Analyze the Problem Given Need 4. 00 g of CH 4 at STP volume of CH 4 Step 2 Write a plan to calculate the needed quantity. g of CH 4 molar mass moles of CH 4 molar volume liters of CH 4

Solution What is the volume (L) at STP of 4. 00 g of CH 4? Step 3 Write the equalities and conversion factors including 22. 4 L/mole at STP. Step 4 Set up the problem with factors to cancel Answer: A units.

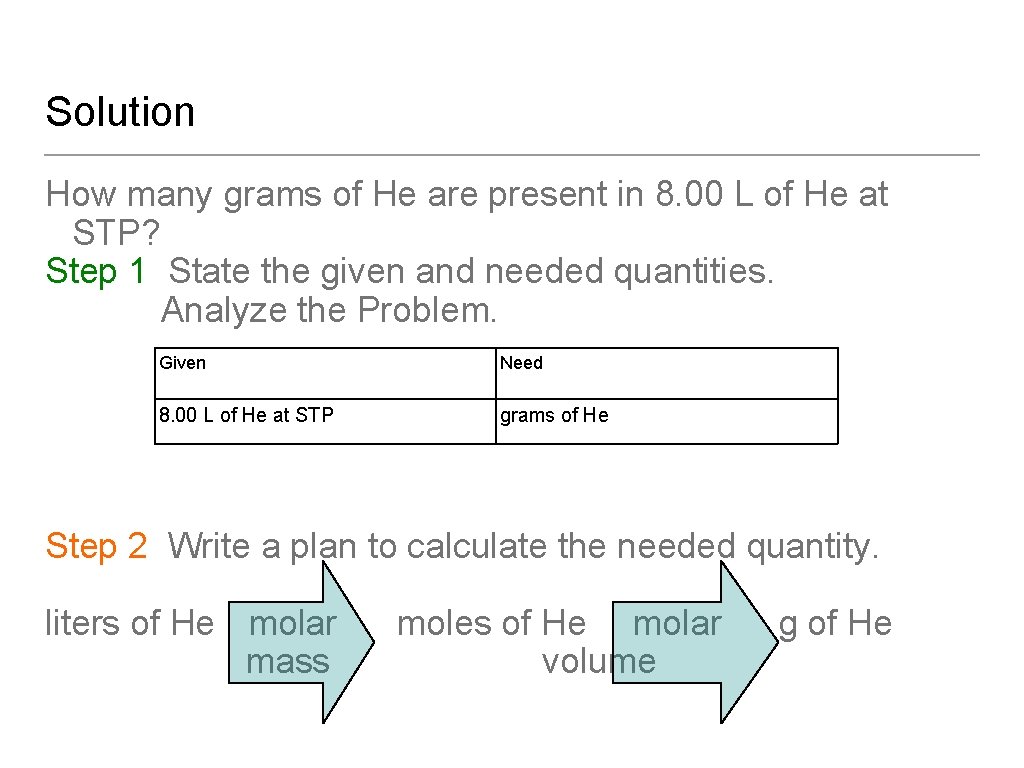
Learning Check How many grams of He are present in 8. 00 L of He at STP? A. 25. 6 g B. 0. 357 g C. 1. 43 g

Solution How many grams of He are present in 8. 00 L of He at STP? Step 1 State the given and needed quantities. Analyze the Problem. Given Need 8. 00 L of He at STP grams of He Step 2 Write a plan to calculate the needed quantity. liters of He molar mass moles of He molar volume g of He

Solution How many grams of He are present in 8. 00 L of He at STP? Step 3 Write the equalities and conversion factors including 22. 4 L/mole at STP. Step 4 Set up the problem with factors to cancel units. Answer: C

Gases in Chemical Reactions The volume or amount of a gas at STP in a chemical reaction can be calculated from • STP conditions. • mole−mole factors from the balanced equation.

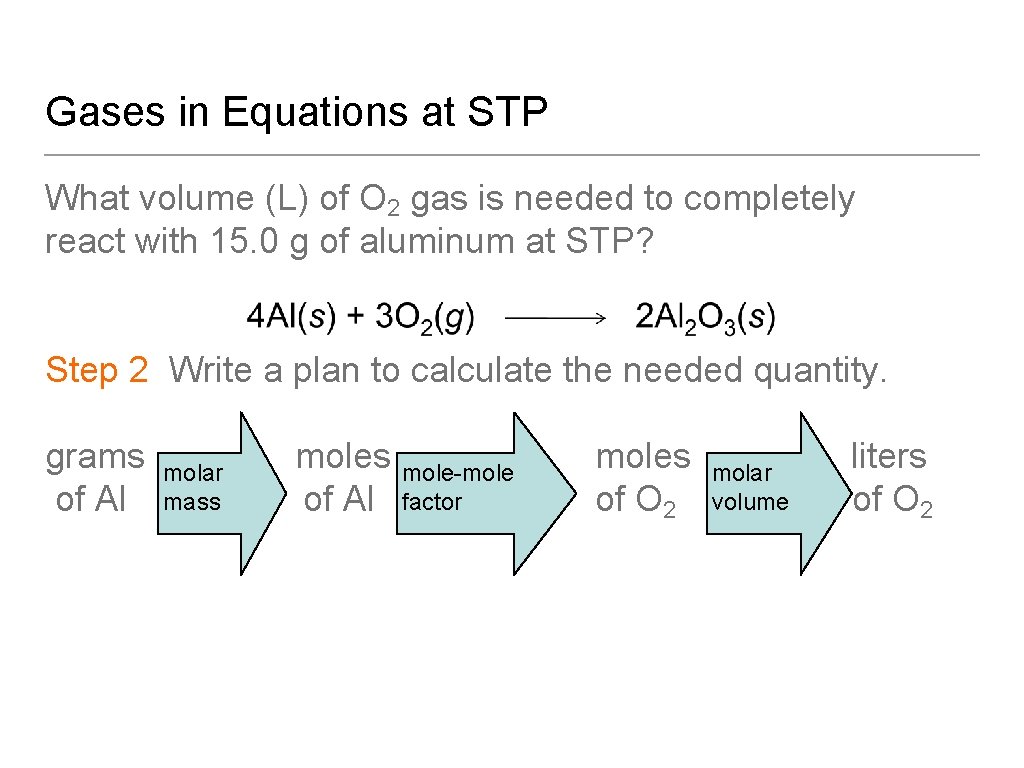
Gases in Equations at STP What volume (L) of O 2 gas is needed to completely react with 15. 0 g of aluminum at STP? Step 1 State the given and needed quantities. Analyze the Problem.

Gases in Equations at STP What volume (L) of O 2 gas is needed to completely react with 15. 0 g of aluminum at STP? Step 2 Write a plan to calculate the needed quantity. grams molar moles mole-mole−moles molar liters volume of Al mass of Al factor of O 2 volume of O 2

Gases in Equations at STP What volume (L) of O 2 gas is needed to completely react with 15. 0 g of aluminum at STP? Step 3 Write the equalities and conversion factors including the molar volume.

Gases in Equations at STP What volume (L) of O 2 gas is needed to completely react with 15. 0 g of aluminum at STP? Step 4 Set up the problem and calculate

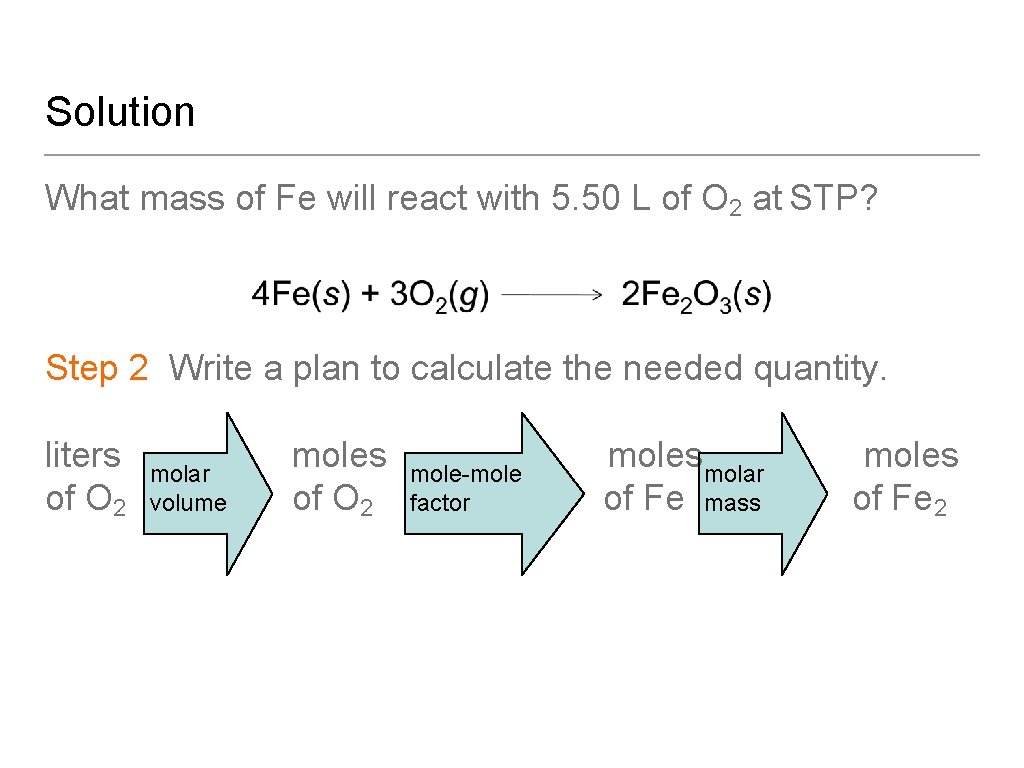
Learning Check What mass of Fe will react with 5. 50 L of O 2 at STP? A. 13. 7 g of Fe B. 18. 3 g of Fe C. 419 g of Fe

Solution What mass of Fe will react with 5. 50 L of O 2 at STP? Step 1 State the given and needed quantities. Analyze the Problem.

Solution What mass of Fe will react with 5. 50 L of O 2 at STP? Step 2 Write a plan to calculate the needed quantity. liters molar moles mole−moles molar moles mole-mole molar of O 2 volume of O 2 factor of Fe mass of Fe 2 volume

Solution What mass of Fe will react with 5. 50 L of O 2 at STP? Step 3 Write the equalities and conversion factors including the molar volume.

Solution What mass of Fe will react with 5. 50 L of O 2 at STP? Step 4 Set up the problem and calculate. Answer is B.

Ideal Gas Law The four properties used in the measurement of a gas, • pressure (P), • volume (V), • temperature (T), and • amount (n), can be combined to give a single expression called the ideal gas law. PV = n. RT

R, Ideal Gas Constant Rearranging the ideal gas law equation shows that the four gas properties equal a constant, R. To calculate the value of R, we substitute the STP conditions for molar volume into the expression:

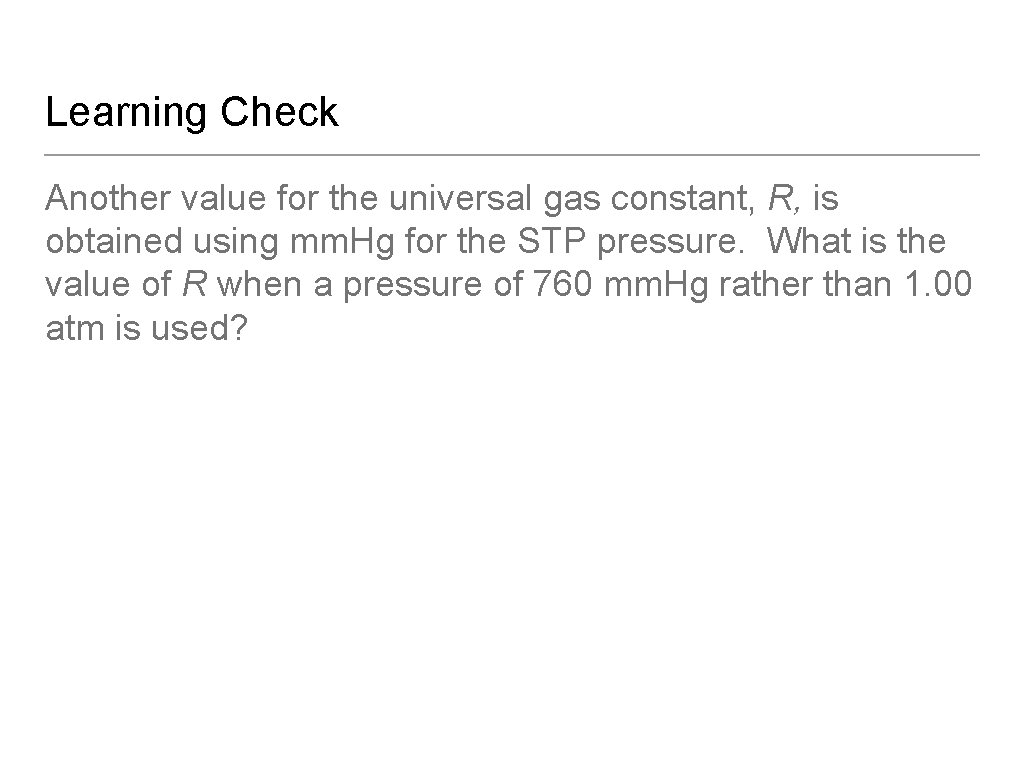
Learning Check Another value for the universal gas constant, R, is obtained using mm. Hg for the STP pressure. What is the value of R when a pressure of 760 mm. Hg rather than 1. 00 atm is used?

Solution Another value for the universal gas constant, R, is obtained using mm. Hg for the STP pressure. What is the value of R when a pressure of 760 mm. Hg rather than 1. 00 atm is used?

Unit Summary for R, the Ideal Gas Constant

Learning Check Dinitrogen oxide (N 2 O), laughing gas, is used by dentists as an anesthetic. If a 20. 0 L tank of laughing gas contains 2. 86 moles of N 2 O at 23 °C, what is the pressure (mm. Hg) in the tank?

Solution If a 20. 0 L tank of laughing gas contains 2. 86 moles of N 2 O at 23 ˚C, what is the pressure (mm. Hg) in the tank? Step 1 State the given and needed quantities. Analyze the Problem.

Solution If a 20. 0 L tank of laughing gas contains 2. 86 moles of N 2 O at 23 ˚C, what is the pressure (mm. Hg) in the tank? Step 2 Rearrange the ideal gas law equation to solve for the needed quantity.

Solution If a 20. 0 L tank of laughing gas contains 2. 86 moles of N 2 O at 23 ˚C, what is the pressure (mm. Hg) in the tank? Step 3 Substitute the gas data into the equation and calculate the needed quantity.

Learning Check A cylinder contains 5. 0 L of an unknown gas at 20. 0 ˚C and 0. 85 atm. If the mass of the gas in the cylinder is 5. 8 g, what is the molar mass of the gas?

Solution A cylinder contains 5. 0 L of an unknown gas at 20. 0 ˚C and 0. 85 atm. If the mass of the gas in the cylinder is 5. 8 g, what is the molar mass of the gas? Step 1 State the given and needed quantities. Analyze the Problem

Solution A cylinder contains 5. 0 L of an unknown gas at 20. 0 ˚C and 0. 85 atm. If the mass of the gas in the cylinder is 5. 8 g, what is the molar mass of the gas? Step 2 Rearrange the ideal gas law equation to solve for the number of moles. Step 3 Obtain the molar mass by dividing the given number of grams by the number of moles.

Chemical Reactions and the Ideal Gas Law

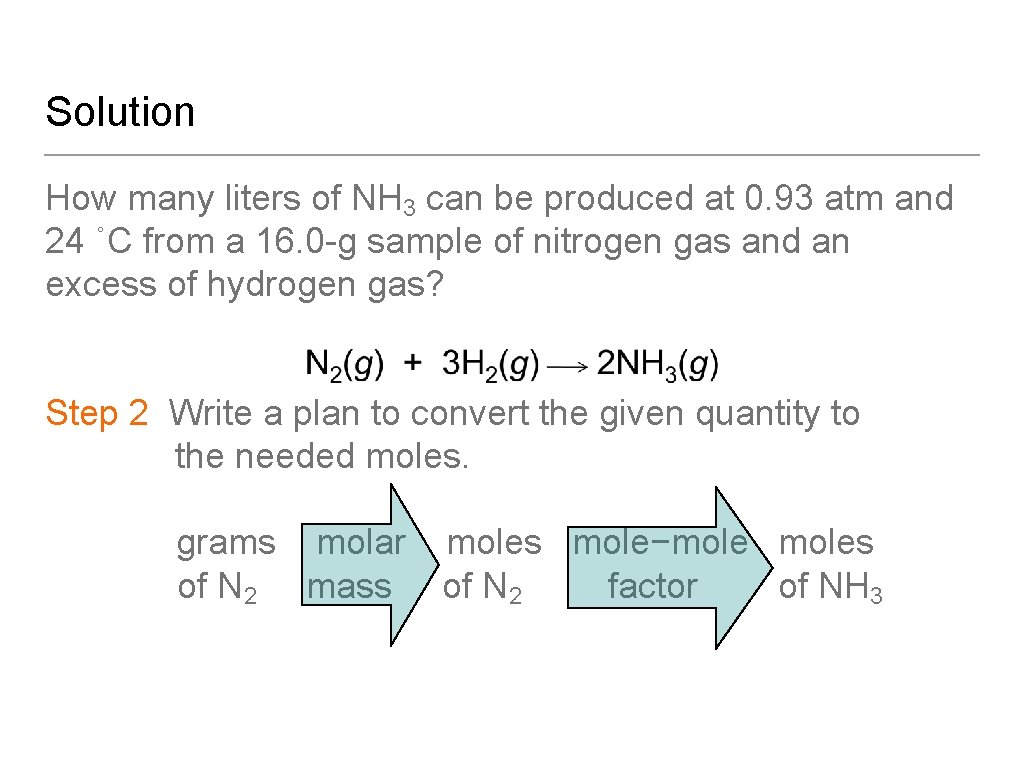
Learning Check Nitrogen gas reacts with hydrogen gas to produce ammonia (NH 3) gas. How many liters of NH 3 can be produced at 0. 93 atm and 24 ˚C from a 16. 0 -g sample of nitrogen gas and an excess of hydrogen gas?

Solution How many liters of NH 3 can be produced at 0. 93 atm and 24 ˚C from a 16. 0 -g sample of nitrogen gas and an excess of hydrogen gas? Step 1 State the given and needed quantities. Analyze the Problem.

Solution How many liters of NH 3 can be produced at 0. 93 atm and 24 ˚C from a 16. 0 -g sample of nitrogen gas and an excess of hydrogen gas? Step 2 Write a plan to convert the given quantity to the needed moles. grams molar of N 2 mass mole−moles of N 2 factor of NH 3

Solution How many liters of NH 3 can be produced at 0. 93 atm and 24 ˚C from a 16. 0 -g sample of nitrogen gas and an excess of hydrogen gas? Step 3 Write the equalities for molar mass and mole– mole factors.

Solution How many liters of NH 3 can be produced at 0. 93 atm and 24 ˚C from a 16. 0 -g sample of nitrogen gas and an excess of hydrogen gas? Step 4 Set up the problem to calculate moles of needed quantity.

Solution How many liters of NH 3 can be produced at 0. 93 atm and 24 ˚C from a 16. 0 -g sample of nitrogen gas and an excess of hydrogen gas? Step 5 Convert the moles of needed to volume using the ideal gas law equation.

Partial Pressure, Dalton’s Law In a gas mixture, each gas exerts its partial pressure, which is the pressure it would exert if it were the only gas in the container. Dalton’s law states that the total pressure of a gas mixture is the sum of the partial pressures of the gases in the mixture.

Dalton’s Law of Partial Pressures indicates that pressure depends on the total number of gas particles.

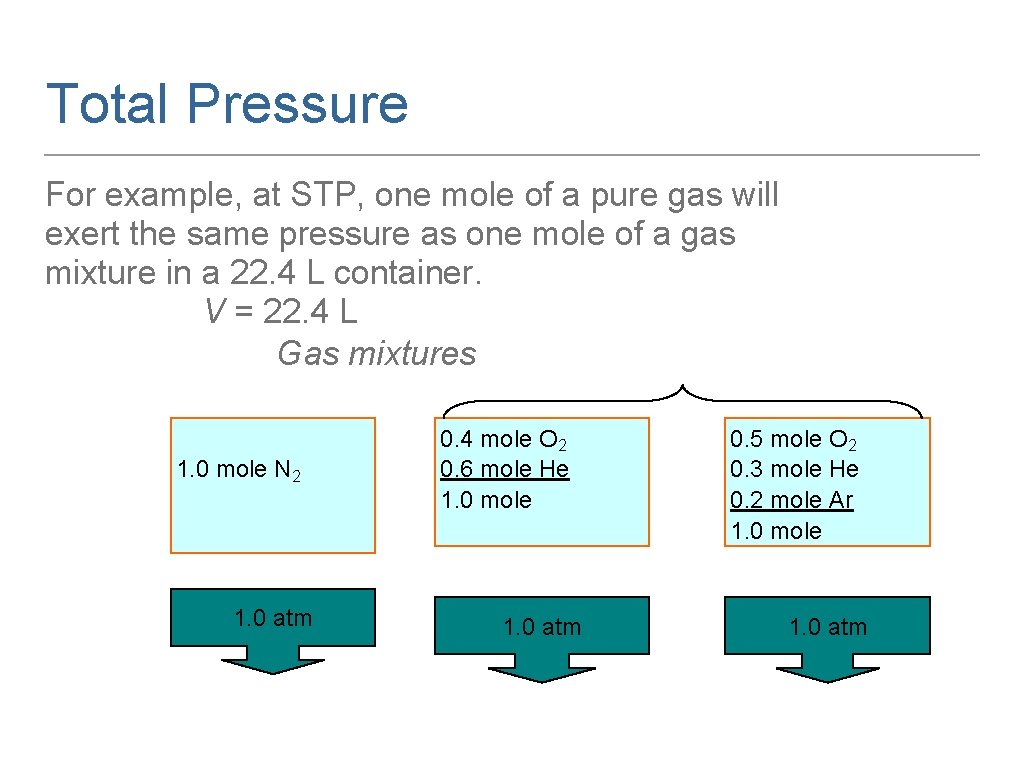
Total Pressure For example, at STP, one mole of a pure gas will exert the same pressure as one mole of a gas mixture in a 22. 4 L container. V = 22. 4 L Gas mixtures 1. 0 mole N 2 1. 0 atm 0. 4 mole O 2 0. 6 mole He 1. 0 mole 1. 0 atm 0. 5 mole O 2 0. 3 mole He 0. 2 mole Ar 1. 0 mole 1. 0 atm

Learning Check A scuba tank contains a mixture of He and O 2 gases at 7. 00 atm. If the pressure of O 2 gas is 1140 mm. Hg, what is the partial pressure of He gas in the tank?

Solution A scuba tank contains a mixture of He and O 2 gases at 7. 00 atm. If the pressure of O 2 gas is 1140 mm. Hg, what is the partial pressure of He gas in the tank? Step 1 Write the equation for partial pressures. Step 2 Rearrange the equation to solve for the unknown pressure.

Solution A scuba tank contains a mixture of He and O 2 gases at 7. 00 atm. If the pressure of O 2 gas is 1140 mm. Hg, what is the partial pressure of He gas in the tank? Step 3 Substitute known pressures into the equation and calculate the unknown partial pressure.